Abstract
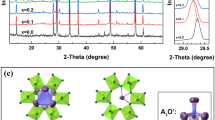
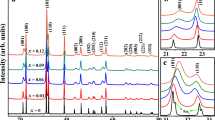
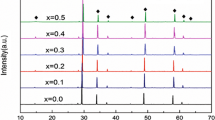
As a potentially attractive candidate of LTCC material, Bi1.5Zn1.0Nb1.5O7 ceramics with substituted Zn2+ were synthesized by a conventional mixed oxide route. For the first time, the effect of the chemical ionic substitution of Sr, Ca and Mn cations on the microstructure, bond energy and dielectric properties of bismuth-based pyrochlores was discussed systematically for Bi1.5Zn1.0Nb1.5O7. Rietveld refinement was carried to analyze the structure variation of the sintered specimens. The solid solubility limits of in the Bi1.5Zn1.0Nb1.5O7 system with Sr, Ca and Mn incorportion were discussed. The samples presented single cubic phase that were used to study the dielectric properties of Bi1.5Zn1.0Nb1.5O7 based on the crystallization and microstructure. The dielectric constant εr was mainly related to relative density not the ionic polarizability. The relative density and the dielectric constant εr showed the same decreased trend as a function of different ionic dopant. And the change of the grain size and grain boundaries had a significant influence on the loss tangent tanδ in this system. The A-site cation bond energy of (Bi1.5Zn0.4M0.1)(Nb1.5Zn0.5)O7 (M = Sr, Ca, Mn, Zn) ceramics was performed to explain the relationship between the temperature coefficient of the dielectric constant τε and the crystal microstructure in a new way. With the decrease of |τε| values as a function of the substitution, the Zn–O bond energy of A-site became strong. This indicated that the (Bi1.5Zn0.4M0.1)(Nb1.5Zn0.5)O7 system tended to be stable.






Similar content being viewed by others
References
L.X. Pang, H. Wang, D. Zhou, X. Yao, Effect of Ag on the sintering and dielectric properties behavior of Bi1.5ZnNb1.5O7 ceramics. J. Alloys Compd. 493, 626–629 (2010)
W. Liu, H. Wang, K. Li, X. Yao, Effect of Ag on the sintering and dielectric properties behavior of Bi1.5ZnNb1.5O7 ceramics. J. Alloys Compd. 491, 218–222 (2010)
C.C. Khawa, K.B. Tan, C.K. Lee, A.R. West, Phase equilibria and electrical properties of pyrochlore and zirconolite phases in the Bi2O3–ZnO–Ta2O5 system. J. Eur. Ceram. Soc. 32, 671–680 (2012)
Y. Liu, R.L. Withers, H.B. Nguyen, K. Elliott, Q.J. Ren, Z.H. Chen, Displacive disorder and dielectric relaxation in the stoichiometric bismuth-containing pyrochlores, Bi2MIIINbO7 (M = In and Sc). J. Solid State Chem. 182, 2748–2755 (2009)
R.L. Withers, T.R. Welberry, A.K. Larsson, Y. Liu, L. Norén, H. Rundlöf, F.J. Brink, Local crystal chemistry, induced strain and short range order in the cubic pyrochlore (Bi1.5−αZn0.5−β)(Zn0.5−γNb1.5−δ)O(7−1.5α−β−γ−2.5δ)(BZN). J. Solid State Chem. 177, 231–244 (2004)
S.M. Zanetti, S.A. Da Silva, G.P. Thim, A chemical route for the synthesis of cubic bismuth zinc niobate pyrochlore nanopowders. J. Solid State Chem. 177, 4546–4551 (2004)
V. Krayzman, I. Levin, J.C. Woicik, Local structure of displacively disordered pyrochlore dielectrics. Chem. Mater. 19, 932–936 (2007)
D.P. Cann, C.A. Randall, T.R. Shrout, Investigation of the dielectric properties of bismuth pyrochlores. Solid State Commun 100, 529–534 (1996)
J. Banys, S. Rudys, M. Ivanov, J. Li, H. Wang, Dielectric properties of cubic bismuth based pyrochlores containing lithium and fluorine. J. Eur. Ceram. Soc. 30, 385–388 (2010)
K.B. Tan, C.C. Khaw, C.K. Lee, Z. Zainal, G.C. Miles, Structures and solid solution mechanisms of pyrochlore phases in the systems Bi2O3–ZnO–(Nb, Ta)2O5. J. Alloys Compd. 508, 457–462 (2010)
M.C. Wu, Y.C. Huang, W.F. Su, Silver cofirable Bi1.5Zn0.92Nb1.5O6.92 microwave ceramics containing CuO-based dopants. Mater. Chem. Phys. 100, 391–394 (2006)
J. Petzelt, S. Kamba, Submillimetre and infrared response of microwave materials: extrapolation to microwave properties. Mater. Chem. Phys. 79, 175–180 (2003)
K. Sudheendran, K.C.J. Raju, M.V. Jacob, Microwave dielectric properties of Ti-substituted Bi2(Zn2/3Nb4/3)O7 pyrochlores at cryogenic temperatures. J. Am. Ceram. Soc. 92, 1268–1271 (2009)
H.L. Du, X. Yao, Dielectric relaxation characteristics of bismuth zinc niobate pyrochlores containing titanium. Phys. B Condens. Matter 324, 121–126 (2002)
C.J. Nino, T.L. Michael, A.R. Clive, Dielectric relaxation in Bi2O3–ZnO–Nb2O5 cubic pyrochlore. J. Appl. Phys. 89, 4512–4516 (2001)
R.T. Shannon, Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides. Acta Crystallogr. Sect. A Cryst. Phys. Diffr. Theor. Gen. Crystallogr. 32, 751–767 (1976)
B.J. Wuensch, K.W. Eberman, C. Heremans, E.M. Ku, P. Onnerud, E.M. Yeo, J.D. Jorgensen, Connection between oxygen-ion conductivity of pyrochlore fuel-cell materials and structural change with composition and temperature. Solid State Ion. 129, 111–133 (2000)
Y.X. Pang, X. Bao, Influence of temperature, ripening time and calcination on the morphology and crystallinity of hydroxyapatite nanoparticles. J. Eur. Ceram. Soc. 23, 1697–1704 (2003)
R. Jenkins, R.L. Snyder, Introduction to X-Ray Power Diffractometry (Wiley, New York, 1996)
D. Shihua, Y. Xi, M. Yu, L. Puling, Microwave dielectric properties of (Bi1−xRx)NbO4 ceramics (R = Ce, Nd, Dy, Er). J. Eur. Ceram. Soc. 26, 2003–2005 (2006)
R.D. SruNNoN, G.R. RossvraN, Dielectric constants of silicate garnets and the oxide additivity rule. Am. Mineral. 77, 94–100 (1992)
Robert D. Shannon, Dielectric polarizabilities of ions in oxides and fluorides. J. Appl. Phys. 73(1), 348–366 (1993)
A.J. Moulson, J.M. Herbert, Electroceramics: Materials, Properties, Applications (Wiley, New York, 2003)
R.T. Sanderson, Multiple and single bond energies in inorganic molecules. J. Inorg. Nucl. Chem. 30, 375–393 (1968)
R.T. Sanderson, Chemical Bonds and Energy (Academic Press, New York, 1971)
R.T. Sanderson, Electronegativity and bond energy. J. Am. Ceram. Soc. 105, 2259–2261 (1983)
Y.R. Luo, Comprehensive Handbook of Chemical Bond Energies (CRC Press, Boca Raton, 2007)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, L.X., Zhang, S. & Lv, X.S. Crystal chemistry and dielectric properties of (Bi1.5Zn0.4M0.1)(Nb1.5Zn0.5)O7 (M = Sr, Ca, Mn, Zn) pyrochlore oxides. J Mater Sci: Mater Electron 28, 4388–4395 (2017). https://doi.org/10.1007/s10854-016-6066-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-016-6066-0