Abstract
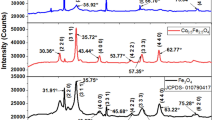
Al3Fe5O12 (AIG) nanopowders were synthesized at different pH using aqueous co-precipitation method. The effect of pH on the phase formation of AIG is characterized using XRD, TEM and TG/DTA. From Scherrer formula the average crystallite sizes were found to be 20, 25, 28 and 32 nm for pH 9, 10, 11 and 12. From TEM micrographs, the particle sizes of the powders were found to be 15, 21, 25 and 30 nm for pH 9, 10, 11 and 12, respectively. It is found that as the pH of the solution increase the particle size also increases. It is clear from the TG/DTA curves that as the pH is increasing the weight losses were found to be small. The obtained nanopowders were further sintered at 900 °C/4 h using conventional sintering method. The phase formation is completed at 800 °C which is correlated with TG/DTA. X-ray photoelectron spectroscopy is used to study the electronic state of the AIG sample. The average grain size of the samples is found to be ~55 nm. Room temperature magnetization measurements established these compounds to be soft magnetic. The room temperature dielectric and magnetic properties (ε′, ε″, µ′ and µ″) of AIG was studied over a wide range of frequency 1 MHz–1.8 GHz. The dielectric constant was found to decrease with increasing frequency. With increase of pH both ε′ and µ′ increased. The Curie temperature was confirmed to be from 560 K (~287 °C) based on the dielectric anomaly observed when these measurements were carried out over a temperature range of 300–600 K. The ferromagnetic resonance linewidth is found to be increasing from 77 to 142 Oe and the effective saturation magnetization (4πMeff) is found to be higher compare to effective saturation magnetization (4πMs) by VSM. This finding provides a new route for AIG materials could be useful for various applications for spintronics.













Similar content being viewed by others
References
M. Wu, A. Hoffmann, Recent Advances in Magnetic Insulators-from Spintronics to Microwave Applications, in Solid State Physics, vol. 64 (Academic Press, Cambridge, 2013)
A. Hoffmann, S.D. Bader, Phys. Rev. Appl. 4, 047001 (2015)
G.J. Long, F. Grandjean, Inorg. Chem. 55, 3413 (2016)
C. Du, H. Wang, P. Chris Hammel, F. Yang, J. Appl. Phys. 117, 172603 (2015)
A. Hamadeh, O. d’Allivy Kelly, C. Hahn, H. Meley, R. Bernard, A.H. Molpeceres, V.V. Naletov, M. Viret, A. Anane, V. Cros, S.O. Demokritov, J.L. Prieto, M. Munoz, G. de Loubens, O. Klein, Phys. Rev. Lett. 113, 197203 (2014)
J. Sklenar, W. Zhang, M.B. Jungfleisch, W. Jiang, H. Chang, J.E. Pearson, M. Wu, J.B. Ketterson, A. Hoffmann, Phys. Rev. B Condens. Matter Mater. Phys. 92, 174406 (2015)
M.B. Jungfleisch, W. Zhang, J. Sklenar, J. Ding, W. Jiang, H. Chang, F.Y. Fradin, J.E. Pearson, J.B. Ketterson, V. Novosad, M. Wu, A. Hoffmann, Phys. Rev. Lett. 116, 057601 (2016)
A.V. Chumak, V.I. Vasyuchka, A.A. Serga, B. Hillebrands, Nat. Phys. 11, 453 (2015)
J. Ding, M. Kostylev, A.O. Adeyeye, Appl. Phys. Lett. 100, 073114 (2012)
V.E. Demidov, S. Urazhdin, H. Ulrichs, V. Tiberkevich, A. Slavin, D. Baither, G. Schmitz, S.O. Demokritov, Nat. Mater. 11, 1028 (2012)
W. Jiang, P. Upadhyaya, W. Zhang, G. Yu, M.B. Jungfleisch, F.Y. Fradin, J.E. Pearson, Y. Tserkovnyak, K.L. Wang, O. Heinonen, S.G.E. te Velthuis, A. Hoffmann, Science 349, 283 (2015)
V. Singh, V.K. Rai, S. Watanabe, T.K. Gundu Rao, I. Ledoux-Rak, H.-Y. Kwak, Appl. Phys. B 101, 631 (2010)
V.D. Murumkar, K.B. Modi, K.M. Jadhav, G.K. Bichile, R.G. Kulkarni, Mater. Lett. 32, 281 (1997)
H.I. Won, H.H. Nersisyan, C.W. Won, K.H. Lee, Mater. Chem. Phys. 129, 955 (2011)
S. Geller, M.A. Gilleo, J. Phys. Chem. Solids 3, 30 (1957)
K. Praveena, S. Srinath, J. Magn. Magn. Mater. 349, 45 (2014)
K. Sadhana, S.R. Murthy, K. Praveena, Mater. Sci. Semicond. Process. 34, 305 (2015)
D. Vandormael, F. Grandjean, D. Hautot, G.J. Long, J. Phys. Condens. Matter 13, 1759 (2001)
X. Guo, A.H. Tavakoli, S. Sutton, R.K. Kukkadapu, L. Qi, A. Lanzirotti, M. Newville, M. Asta, A. Navrotsky, Chem. Mater. 26, 1133 (2014)
B.E. Warren, X-ray Diffraction (Addison-Wesley, Reading, 1969)
K. Praveena, K. Sadhana, S. Srinath, S. Ramana Murthy, in AIP Conference Proceedings, vol. 1447 (2012), p. 291
K. Sadhana, S.R. Murthy, K. Praveena, J. Mater. Sci. Mater. Electron. 25, 5130 (2014)
P. Ayub, V.R. Palkar, S. Chatopadhyay, M. Multani, Phys. Rev. B 51, 6135 (1995)
N. Yahya, R.A.H. Masoud, H. Daud, A.A. Aziz, H.M. Zaid, Am. J. Eng. Appl. Sci. 2, 76 (2009)
P. Ayub, M. Multani, M. Barma, V.R. Palkar, R. Vijayaraghavan, J. Phys. C 21, 2229 (1988)
L. Xifeng, H. Wang, Y. Yang, T. Liu, J. Mater. Sci. Technol. 27, 245 (2011)
G.C. Allen, K.R. Hallam, Appl. Surf. Sci. 93, 25 (1996)
T. Yamashita, P. Hayes, Appl. Surf. Sci. 254, 2441 (2008)
P. Vaqueiro, M.P. Crosnier-Lopez, M.A. Lopez-Quintela, J. Solid State Chem. 126, 161 (1996)
A. Potdevin, G. Chadeyron, D. Boyer, R. Mahiou, J. Non-Cryst, Solids 352, 2510 (2006)
R.A. Mc Currie, Ferromagnetic Materials: Structure and Properties (Academic Press, University of Michigan, Cambridge, 1954), p. 352
Y.F. Chen, K.T. Wu, Y.D. Yao, C.H. Peng, K.L. You, W.S. Tse, Microelectron. Eng. 81, 329 (2005)
T. Kim, S. Nasu, M. Shima, J. Nanopart. Res. 9, 737 (2007)
R.D. Sanchez, J. Rivas, P. Vaqueiro, J. Magn. Magn. Mater. 247, 92 (2002)
X.Z. Guo, B.G. Ravi, P.S. Devi, J.C. Hanson, J. Margolies, R.J. Gambino, J.B. Parise, S. Sampath, J. Magn. Magn. Mater. 295, 145 (2005)
R. Metselaar, M.A.H. Huyberts, J. Phys. Chem. Solids 34, 2257 (1973)
J. Smit, H.P.J. Wijn, Ferrites (Wiley, New York, 1959)
J. Smit, H.P.J. Wijn, Les Ferrites (Dunod, Paris, 1961)
A.K. Singh, T.C. Goel, R.G. Mendiratta, J. Appl. Phys. 91, 6626 (2002)
N. Rezlescu, E. Rezlescu, Phys. Status Solidi A 23, 575 (1974)
J.T.S. Irvine, A. Huanosta, R. Velenzuela, A.R. West, J. Am. Ceram. Soc. 73, 729 (1990)
C.G. Koops, Phys. Rev. 83, 121 (1951)
L.L. Hench, J.K. West, Principles of Electronic Ceramics (Wiley, New York, 1990), p. 346
F.G. Brockman, R.P. White, J. Am. Ceram. Soc. 54, 183 (1971)
L.G. Uitert, Proc. IRE 44, 1294 (1956)
K. Ishino, Y. Narumiya, Am. Ceram. Bull. 66, 1469 (1987)
D.A. Dimitrov, G.M. Wysin, Phys. Rev. B 51, 11947 (1995)
V.P. Shilov, J.C. Bacri, F. Gazeau, F. Gendron, R. Perzynski, Y.L. Raikher, J. Appl. Phys. 85, 6642 (1999)
A. Verma, R. Chatterjee, J. Magn. Magn. Mater. 306, 313 (2006)
L.T. Rabinkin, Z.I. Novikova, Ferrites, Izv. Acad. Nauk (USSR, Minsk, 1960), p. 146
B. Bhoi, N. Venkataramani, R.P.R.C. Aiyar, S. Prasad, IEEE Trans. Magn. 49, 990 (2013)
Acknowledgements
Dr K. Praveena acknowledges the Ministry of Science and Technology of Republic of China under Grant Nos. MOST 105-2811-M-003-018 and MOST 105-2112-M-003-013-MY3 for financial support.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Praveena, K., Matteppanavar, S., Liu, HL. et al. Effect of pH on electrical and magnetic properties of Al3Fe5O12 nanoparticles. J Mater Sci: Mater Electron 28, 4179–4191 (2017). https://doi.org/10.1007/s10854-016-6038-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-016-6038-4