Abstract
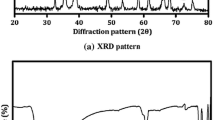
The CuS nanocrystal powders were prepared by a simple chemical route at room temperature and the effect of the solution concentration on properties of CuS nanocrystal were investigated. These CuS nanocrystals were prepared from the solution in which the ratios of copper to thiosulphate were varied from 1:1 to 1:5. X-ray diffraction patterns suggests that the prepared CuS nanocrystals of ratios 1:3–1:5 were very close to the covellite form of copper sulphide. The optical spectra reveal that the powder 1:3, 1:4 and 1:5 shows high absorbance in the visible region. Raman spectra confirmed the covellite form of the copper sulphide nanocrystal. Scanning electron microscopy revealed the formation of agglomerated CuS hexagonal and flex with significant morphological deviation through sulphur concentration. Photoluminescence spectra indicate the concentrations of sulphur increases then the PL emission intensity is also increased. From TEM images, the morphology of the Copper sulphide powder get changes with increasing volumetric ratio of Cu/S. Mechanism of the CuS nanocrystal formation and phase transition is also discussed.









Similar content being viewed by others
References
M. Law, L.E. Greene, J.C. Johnson, R. Saykally, P.D. Yang, Nat. Mater. 4, 455 (2005)
M. Grätzel, J. Photochem. Photobiol. A 164, 3 (2004)
J.B. Baxter, E.S. Aydil, Appl. Phys. Lett. 053114, 86 (2005)
I. Gur, N.A. Fromer, M.L. Geier, A.P. Alivisatos, Science 310, 462 (2005)
K.S. Leschkies, R. Divakar, J. Basu, E. Enache-Pommer, J.E. Boercker, C.B. Carter, U.R. Kortshagen, D.J. Norris, E.S. Aydil, Nano Lett. 7, 1793 (2007)
B. Tian, X. Zheng, T.J. Kempa, Y. Fang, N. Yu, G. Yu, J. Huang, C. Lieber, Nat. 449, 885 (2007)
W.U. Huynh, J.J. Dittmer, A.P. Alivisatos, Science 295, 24–25 (2002)
I. Gur, N.A. Fromer, C. Chen, A.G. Kanaras, A.P. Alivisatos, Nano Lett. 7, 409 (2007)
D.H. Cui, J. Xu, T. Zhu, G. Paradee, S. Ashok, M. Gerhold, Appl. Phys. Lett. 88, 183111 (2006)
W.J.E. Beek, M.M. Wienk, R.A. Janssen, J. AdV. Mater. 16, 1009 (2004)
I. Grozdanov, M. Najdoski, J. Solid State Chem. 114, 469–475 (1995)
Q. Xu, B. Huang, Y. Zhao, Y. Yan, R. Noufi, S. Wei, Appl. Phys. Lett. 100, 061906 (2012)
L. Isac, A. Duta, A. Kriza, S. Manolache, M. Nanu, Thin Solid Films 515, 5755–5758 (2007)
Y. Lu, Y. Hou, Y. Wang, Z. Feng, X. Liu, Y. Lü, Synth. Met. 161, 906–910 (2011)
S.V. Bagul, S.D. Chavhan, R. Sharma, J. Phys. Chem. Solids 68, 1623–1629 (2007)
Y. Lu, G. Yi, J. Jia, Y. Liang, Appl. Surf. Sci. 256, 7316–7322 (2010)
P. Zhang, L. Gao, J. Mater. Chem. 13, 2007–2010 (2003)
M.T.S. Nair, L. Guerrero, P.K. Nair, Semicond. Sci. Technol. 13, 1164–1169 (1998)
S.S. Dhasade, J.S. Patil, S.H. Han, M.C. Rath, V.J. Fulari, Mater. Lett. 90, 138–141 (2013)
S.K. Maji, N. Mukherjee, A.K. Dutta, D.N. Srivastava, P. Paul, B. Karmakar, A. Mondal, B. Adhikary, Mater. Chem. Phys. 130, 392–397 (2011)
J. Zou, J. Jiang, L. Huang, H. Jiang, K. Huang, Solid State Sci. 13, 1261–1267 (2011)
H.M. Pathan, J.D. Desai, C.D. Lokhande, Appl. Surf. Sci. 202, 47–56 (2002)
S.D. Sartale, C.D. Lokhande, Mater. Chem. Phys. 65, 63–67 (2000)
S. Thanikaikarasan, T. Mahalingam, A. Kathalingam, H. Moon, Y.D. Kim, J. New Mater. Electrochem. Syst. 13, 29–33 (2010)
M. Xin, K.W. Li, H. Wang, Appl. Surf. Sci. 256, 1436–1442 (2009)
W.Y. Kim, B.M. Palve, H.M. Pathan, O.-S. Joo, Mater. Chem. Phys. 131, 525–528 (2011)
L. Chen, W. Yu, Y. Li, Powder Technol. 191, 52–54 (2009)
Y. Cui, G. Chen, J. Ren, J. Solid State Chem. 172, 17 (2003)
B. Li, Y. Xie, J. Huang, Inorg. Chem. 39, 2061 (2000)
C. Tan, Y. Zhu, R. Lu, Mater. Chem. Phys. 91, 44 (2005)
P. Roy, S.K. Srivastava, Cryst. Growth Des. 6, 1921 (2006)
F.E. Ghodsi, F.Z. Tepehan, G.G. Tepehan, Surf. Sci. 601, 4497 (2007)
E. Guneri, A. Kariper, J. Alloys Compd. 516, 20 (2012)
M.A. Mahadik, S.S. Shinde, Y.M. Hunge, V.S. Mohite, S.S. Kumbhar, A.V. Moholkar, K.Y. Rajpure, C.H. Bhosale, J. Alloys Compd. 611, 446 (2014)
M.A. Butler, J. Appl. Phys. 48, 1914–1920 (1977)
K.M. Gadave, C.D. Lokhande, Thin Solid Films 229, 1–4 (1993)
S.D. Sartale, C.D. Lokhande, Mater. Chem. Phys. 65, 63–67 (2000)
S. Erokhina, V. Erokhin, C. Nicolini, Langmuir 19, 766–771 (2003)
S. Erokhina, V. Erokhin, C. Nicolini, Colloid Surf. A 198–200, 645–650 (2002)
B. Minceva-Sukarova, M. Najdoski, I. Grozdanov, C.J. Chunnilall, J. Mol. Struct. 410, 267 (1997)
S.Y. Wang, W. Wang, Z.H. Lu, Mater. Sci. Eng. B 103, 184 (2003)
M. Ishii, K. Shibata, H. Nozaki, J. Solid State Chem. 105, 504–511 (1993)
A.G. Milekhin, N.A. Yeryukov, L.L. Sveshnikova, T.A. Duda, E.E. Rodyakina, V.A. Gridchin, E.S. Sheremet, D.R.T. Zahn, Beilstein J. Nanotechnol. 6, 749–754 (2015)
M. Saranya, R. Ramachandran, E.J.J. Samuel, S.K. Jeong, A.N. Grac, Powder Technol. 279, 209–220 (2015)
J. Liu, D. Xue, J. Mater. Chem. 21, 223 (2011)
Y. Wu, C. Wadia, W.L. Ma, B. Sadtler, A.P. Alivisatos, Nano Lett. 8, 2551–2555 (2008)
Y.P. Du, B. Xu, T. Fu, M. Cai, F. Li, Y. Zhang, Q.B. Wang, J. Am. Chem. Soc. 132, 1470–1471 (2010)
G.M. Liu, T. Schulmeyer, J. Brotz, A. Klein, W. Jaegerman, Thin Solid Films 431, 477–482 (2003)
Acknowledgments
Authors are thankful to Department Research Development Programme, Department of Physics, Savitribai Phule Pune University India for their financial support for research work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Palve, B.M., Jadkar, S.R. & Pathan, H.M. A simple chemical route to synthesis the CuS nanocrystal powder at room temperature and phase transition. J Mater Sci: Mater Electron 27, 11783–11789 (2016). https://doi.org/10.1007/s10854-016-5318-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-016-5318-3