Abstract
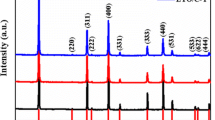
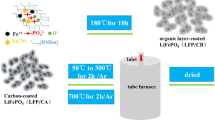
In order to obtain LiFePO4 (LFP) with the fine particle size and high electronic conductivity, the secondary carbon-coated LFP-C composite was synthesized by a facile two-step method with polyethylene glycol (PEG) as a grain growth inhibitor and sucrose as a main carbon source. For comparison, commercial LFP and LFP prepared without PEG and sucrose were also studied. Li-ion capacitors (LICs) using environmentally friendly, safe and low-cost LiNO3 aqueous electrolyte were assembled with LFP samples as the positive electrode. Results show that the secondary carbon-coated LiFePO4-C composite with nanometer-sized particles was synthesized at 550 °C successfully and the carbon content was about 23.4 wt%. The lamellar carbon coating, with much graphitic nature, wraps and connects LFP particles, ensuring the good electronic connection between LFP particles. The results of cyclic voltammetry, galvanostatic charge/discharge and electrochemical impedance show that the electrochemical performances of secondary carbon-coated LFP dramatically increase due to the decrease of particle size through adding PEG and the increased electronic conductivity by the introduction of carbon coating. It exhibits the C pe of 845.2 F g−1 at the scanning rate of 5 mV s−1. The LIC delivers a C ps of 59.3 F g−1 and E p of 8.2 Wh kg−1 at the current density of 2 mA cm−2. The LIC exhibits an excellent cycling performance and it maintains 98 % of its initial C ps after 500 cycles. The secondary carbon-coated LiFePO4-C composite is the suitable positive electrode material for LICs with neutral aqueous electrolytes.










Similar content being viewed by others
References
H. Kang, G. Wang, H. Guo, M. Chen, C. Luo, K. Yan, Eng. Chem. Res. 51, 7923 (2012)
Z. Chang, Y. Yang, M. Li, X. Wang, Y. Wu, J. Mater. Chem. A 2, 10739 (2014)
D. Puthusseri, V. Aravindan, S. Madhavi, S. Ogale, Electrochim. Acta 130, 766 (2014)
V. Aravindan, W. Chuiling, M.V. Reddy, G.V. Rao, B.V. Chowdari, S. Madhavi, Phys. Chem. Chem. Phys. 14, 5808 (2012)
W.J. Cao, J.P. Zheng, J. Electrochem. Soc. 160, A1572 (2013)
M.D. Stoller, S. Murali, N. Quarles, Y. Zhu, J.R. Potts, X. Zhu, H.W. Ha, R.S. Ruoff, Phys. Chem. Chem. Phys. 14, 3388 (2012)
G.G. Amatucci, F. Badway, A. Du Pasquier, T. Zheng, J. Electrochem. Soc. 148, A930 (2001)
L.M. Chen, Q.Y. Lai, Y.J. Hao, J.H. Huang, X.Y. Ji, Ionics 14, 441 (2008)
T. Zhang, F. Zhang, L. Zhang, Y. Lu, Y. Zhang, X. Yang, Y. Ma, Y. Huang, Carbon 92, 106 (2015)
Q. Zhu, S. Zheng, X. Lu, Y. Wan, Q. Chen, J. Yang, L.Z. Zhang, Z. Lu, J. Alloys Compd. 654, 384 (2016)
W. Tang, L.L. Liu, S. Tian, L. Li, L.L. Li, Y.B. Yue, Y. Bai, Y.P. Wu, K. Zhu, R. Holze, Electrochem. Commun. 13, 1159 (2011)
F.X. Wang, S.Y. Xiao, Y. Shi, L.L. Liu, Y.S. Zhu, Y.P. Wu, J.Z. Wang, R. Holze, Electrochim. Acta 93, 301 (2013)
X. Zeng, Q. Liu, M. Chen, L. Leng, T. Shu, L. Du, H. Song, S. Liao, Electrochim. Acta 177, 277 (2015)
A.K. Padhi, K.S. Nanjundaswamy, J.B. Goodenough, J. Electrochem. Soc. 144, 1188 (1994)
R. Sehrawat, A. Sil, J. Mater. Sci. Mater. Electron. 26, 5175 (2015)
D. Shao, J. Wang, X. Dong, W. Yu, G. Liu, F. Zhang, L. Wang, J. Mate. Sci. Mater. Electron 25, 1040 (2013)
R. Muruganantham, M. Sivakumar, R. Subadevi, N.L. Wu, J. Mater. Sci. Mater. Electron. 26, 2095 (2015)
Y. Wang, R. Mei, X. Yang, Ceram. Int. 40, 8439 (2014)
Z. Wang, H. Guo, P. Yan, Ceram. Int. 40, 15801 (2014)
M. Kuzmanović, D. Jugović, M. Mitrić, B. Jokić, N. Cvjetićanin, D. Uskoković, Ceram. Int. 41, 6753 (2015)
A.V. Murugan, T. Muraliganth, A. Manthiram, Electrochem. Commun. 10, 903 (2008)
H. Shu, X. Wang, Q. Wu, B. Hu, X. Yang, Q. Wei, B. Hu, X. Yang, Q. Wei, Q. Liang, Y. Bai, M. Zhou, C. Wu, M. Chen, A. Wang, L. Jiang, J. Power Sources 237, 149 (2013)
C. Qin, X. Lu, G. Yin, Z. Jin, Q. Tan, X. Bai, Mater. Chem. Phys. 126, 453 (2011)
L.N. Wang, X.C. Zhan, Z.G. Zhang, K.L. Zhang, J. Alloys Compd. 456, 461 (2008)
A. Churikov, A. Gribov, A. Bobyl, A. Kamzin, E. Terukov, Ionics 20, 1 (2013)
Y.L. Cao, L.H. Yu, T. Li, X.P. Ai, H.X. Yang, J. Power Sources 172, 913 (2007)
D. Choi, P.N. Kumta, J. Power Sources 163, 1064 (2007)
X. Li, W. Wang, C. Shi, H. Wang, Y. Xing, J. Solid State Electrochem. 13, 921 (2008)
Z. Xu, L. Xu, Q. Lai, X. Ji, Mater. Res. Bull. 42, 883 (2007)
J. Yang, X. Han, X. Zhang, F. Cheng, J. Chen, Nano Res. 6, 679 (2013)
Z.G. Lu, M.F. Lo, C.Y. Chung, J. Phys. Chem. C 112, 7069 (2008)
Y.D. Cho, G.T.K. Fey, H.M. Kao, J. Power Sources 189, 256 (2009)
O. Toprakci, H.A. Toprakci, L. Ji, G. Xu, Z. Lin, X. Zhang, A.C.S. Appl, Mater. Interfaces 4, 1273 (2012)
A. Naik, J. Zhou, C. Gao, L. Wang, Electrochim. Acta 142, 215 (2014)
F.Y. Kang, J. Ma, B.H. Li, New Carbon Mater. 26, 161 (2011)
J. He, H. Wang, C. Gu, S. Liu, J. Alloys Compd. 604, 239 (2014)
B. Wang, D. Wang, Q. Wang, T. Liu, C. Guo, X. Zhao, J. Mater. Chem. A 1, 135 (2013)
Y. Zhang, W. Wang, P. Li, Y. Fu, X. Ma, J. Power Sources 210, 47 (2012)
D. Hulicova, M. Kodama, H. hatori. Chem. Mater. 18, 2318 (2006)
Y. Liu, X. Li, H. Guo, Z. Wang, W. Peng, Y. Yang, R. Liang, J. Power Sources 184, 522 (2008)
M. Zhou, L. Liu, L. Yi, Z. Yang, S. Mao, Y. Zhou, T. Hu, Y. Yang, B. Shen, X. Wang, J. Power Sources 234, 292 (2013)
L. Wang, G.C. Liang, X.Q. Ou, X.K. Zhi, J.P. Zhang, J.Y. Cui, J. Power Sources 189, 423 (2009)
J. Zhu, K. Yoo, I. El-Halees, D. Kisailus, A.C.S. Appl, Mater. Interfaces 6, 21550 (2014)
F. Zhang, T. Zhang, X. Yang, L. Zhang, K. Leng, Y. Huang, Y. Chen, Energy Environ. Sci. 6, 1623 (2013)
R. Dominko, M. Bele, M. Gaberscek, M. Remskar, D. Hanzel, S. Pejovnik, J. Jamnik, J. Electrochem. Soc. 152, A607 (2005)
J. Wang, X. Sun, Energy Environ. Sci. 5, 5163 (2012)
Acknowledgments
The authors gratefully acknowledge the financial assistance of Natural Science Foundation Committee of China (Grant Number 21206034), Outstanding Youth Science Foundation Committee of Heilongjiang University (Grant Number JCL201202) and Graduate Student Innovation Research Project Committee of Heilongjiang Province(Grant Number YJSCX2012-318HLJ).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Qiu, S., Zhang, X., Li, Y. et al. Facile synthesis and electrochemical performances of secondary carbon-coated LiFePO4-C composite for Li-ion capacitors based on neutral aqueous electrolytes. J Mater Sci: Mater Electron 27, 7255–7264 (2016). https://doi.org/10.1007/s10854-016-4692-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-016-4692-1