Abstract
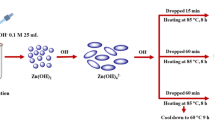
Pure barium stannate (BaSnO3) nanostructures were prepared via a new facile surfactant-free coprecipitation-calcination route by employing Ba(Sal)2 (Sal = salicylidene) and SnCl2·2H2O as precursors. Ba(Sal)2 was employed as precursor to synthesize BaSnO3 nanostructures for the first time. The as-obtained BaSnO3 nanostructures were analyzed by UV–vis diffuse reflectance spectroscopy, field emission scanning electron microscopy, Fourier transform infrared spectroscopy, energy dispersive X-ray microanalysis, transmission electron microscopy, and X-ray diffraction. Based on the morphological investigations of the as-prepared samples, it was found that the particle size and shape of the BaSnO3 depended on the reaction temperature, precipitator and surfactant. BaSnO3 nanostructures with various shapes and particle sizes were successfully prepared. Furthermore, the photocatalytic properties of as-synthesized BaSnO3 were evaluated by degradation of eriochrome black T (anionic dye) as water contaminant.














Similar content being viewed by others
References
M. Sabet, M. Salavati-Niasari, Omid Amiri. Electrochim. Acta 117, 504 (2014)
S. Mortazavi-Derazkola, S. Zinatloo-Ajabshir, M. Salavati-Niasari, Ceram. Int. 41, 9593 (2015)
S. Zinatloo-Ajabshir, M. Salavati-Niasari, Int. J. Appl. Ceram. Technol. 11, 654 (2014)
F. Beshkar, S. Zinatloo-Ajabshir, M. Salavati-Niasari, J. Mater. Sci.: Mater. Electron. 26, 5043 (2015)
S. Zinatloo-Ajabshir, M. Salavati-Niasari, New J. Chem. 39, 3948 (2015)
V.G. Wagner, H. Binder, Z. Anorg. Allg. Chem. 298, 12 (1959)
Z. Zhigang, Z. Gang, Ferroelectrics 101, 43 (1990)
P.H. Borse, J.S. Lee, H.G. Kim, J. Appl. Phys. 100, 124915 (2006)
W. Wang, Sh Liang, K. Ding, J. Bi, J.C. Yu, P. Keung, L. Wong, J. Wu, J. Mater. Sci. 49, 1893 (2014)
S.S. Shin, J.S. Kim, J.H. Suk, K.D. Lee, D.W. Kim, J.H. Park, I.S. Cho, K.S. Hong, J.Y. Kim, ACS Nano 7, 1027 (2013)
J. Cerda, J. Arbiol, G. Dezanneau, R. Díaz, J.R. Morante, Sensors Actuators B 84, 21 (2002)
T. Huang, T. Nakamura, M. Itoh, Y. Inaguma, O. Ishiyama, J. Mater. Sci. 30, 1556 (1995)
W. Zhang, J. Tang, J. Ye, J. Mater. Res. 22, 1859 (2007)
H. Mizoguchi, H.W. Eng, P.M. Woodward, Inorg. Chem. 43, 1667 (2004)
V. Vorgelegt, L. Wensheng, Synthesis of nanosized BaSnO3 powders. Doctoral thesis in Engineering of Natural Sciences, Faculty of Engineering, University of Saarlandes, Saarbrücken—Germany, 2002, pp. 1–2
S. Upadhyay, O. Parkash, D. Kumar, Mater. Lett. 49, 251 (2001)
A.S. Deep, S. Vidya, P.C. Manu, S. Solomon, A. John, J.K. Thomas, J. Alloys Compd. 509, 1830 (2011)
W. Lu, H. Schmidt, J. Sol-Gel. Sci. Technol. 42, 55 (2007)
J. Ahmed, C.K. Blakely, S.R. Bruno, V.V. Poltavets, Mater. Res. Bull. 47, 2282 (2012)
Y.H.O. Muñoz, M. Ponce, J. E. R. Páez Powder Technol. 279, 86 (2015)
F. Beshkar, S. Zinatloo-Ajabshir, M. Salavati-Niasari, Chem. Eng. J. (2015). doi:10.1016/j.cej.2015.05.076
S. Zinatloo-Ajabshir, M. Salavati-Niasari, M. Hamadanian, RSC Adv. 5, 33792 (2015)
S. Mortazavi-Derazkola, S. Zinatloo-Ajabshir, M. Salavati-Niasari, RSC Adv. 5, 56666 (2015)
M. Shakouri-Arania, M. Salavati-Niasari, New J. Chem. 38, 1179 (2014)
M. Ghaed-Amini, M. Bazarganipour, M. Salavati-Niasari, J. Ind. Eng. Chem. 21, 1089 (2015)
G. Pfaff, V.D. Hildenbrand, H. Fuess, J. Mater. Sci. Lett. 17, 1983 (1998)
S. Taoa, F. Gao, X. Liu, O. Toft Sùrensen. Sensors Actuators B 71, 223 (2000)
B. Andriyevsky, A. Patryn, K. Dorywalski, Ch. Cobet, M. Piasecki, I. Kityk, N. Esser, T. Łukasiewicz, J. Dec, Ferroelectrics 426, 194 (2012)
J. Zhong, J. Li, F. Feng, Y. Lu, J. Zeng, W. Hu, Z. Tang, J. Mol. Catal. A: Chem. 357, 101 (2012)
D. Ghanbari, M. Salavati-Niasari, S. Karimzadeh, S. Gholamrezaei, J. NanoStruct. 4, 227 (2014)
G. Nabiyouni, S. Sharifi, D. Ghanbari, M. Salavati-Niasari, J. NanoStruct. 4, 317 (2014)
M. Panahi-Kalamuei, M. Mousavi-Kamazani, M. Salavati-Niasari, J. NanoStruct. 4, 459 (2014)
F. Beshkar, M. Salavati-Niasari, J. NanoStruct. 5, 17 (2015)
M. Goudarzi, D. Ghanbari, M. Salavati-Niasari, J. NanoStruct. 5, 110 (2015)
S. Moshtaghi, M. Salavati-Niasari, D. Ghanbari, J. NanoStruct. 5, 169 (2015)
Acknowledgments
The authors are grateful to University of Kashan for supporting this work by Grant No. 159271/20.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Moshtaghi, S., Zinatloo-Ajabshir, S. & Salavati-Niasari, M. Preparation and characterization of BaSnO3 nanostructures via a new simple surfactant-free route. J Mater Sci: Mater Electron 27, 425–435 (2016). https://doi.org/10.1007/s10854-015-3770-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-015-3770-0