Abstract
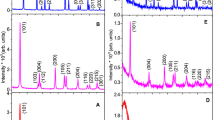
Titania nanoparticles have been prepared using different surfactants such as, acetic acid (AA), oleic acid (OA), oley amine (OM), and a mixture of OA + OM at room temperature by sol–gel method. TiO2 nanoparticles were collected by centrifugation of the precipitate obtained during gel formation. The collected samples were annealed at 550 and 950 °C to study the effect of annealing temperature on the structural and optical properties. The crystal structure and optical properties of titania nanoparticle is investigated by means of X-ray diffraction, Raman spectroscopy, UV–visible spectroscopy, and photoluminescence. After heat treatment at 950 °C, the mixed rutile and anatase phase of TiO2 was revealed for the sample prepared with AA, whereas pure rutile phase was observed for the sample prepared in presence of OA, OM and the mixture of OA + OM. The energy band gap and transmittance of measured for titania nanoparticles was found to be systematically reduced with increase in annealing temperature for each surfactant. The ideality factor decreases with increase in annealing temperature for all surfactants could be related to the voltage dependence of the standard deviation of the distribution of barrier heights.





Similar content being viewed by others
References
T. Lihong, Y. Liqun, D. Kejian, Z. Ling, J. Solid State Chem. 184, 1465 (2011)
P. Kaushik, P.M. Tapas, N. Chirantan, C.D. Subhas, Mol. Struct. 1016, 30 (2012)
R. Mechiakh, N.B. Sedrine, M. Karyaoui, R. Chtourou, Appl. Surf. Sci. 257, 5529 (2011)
N.B. Chaure, A.K. Ray, R. Capan, Semicond. Sci. Technol. 20, 788 (2005)
A.K. Hassan, N.B. Chaure, A.K. Ray, A.V. Nabok, S. Habesch, J. Phys. D Appl. Phys. 36, 1120 (2003)
N.S. Godlisten, E. Gideon, V.Q. Dang, N.K. You, HSh Young, H. Askwar, K. Jong-Kil, T.K. Hee, Powder Technol. 217, 489 (2012)
M.A. Khan, M.S. Akhtar, O.-B. Yang, Sol. Energy 84, 2195 (2010)
T. Madhavi, D. Kanjilal, B. Varsha, Appl. Surf. Sci. 258, 7855 (2012)
M. Vishwasa, K.N. Rao, K.V.A. Gowda, R.P.S. Chakradhar, Spectrochim. Acta Part A 83, 614 (2011)
X. Jiaoxing, L. Li, Y. Yan, H. Wang, X. Wang, X. Fu, G. Li, J. Colloid Interface Sci. 318, 29 (2008)
W. Patladda, C. Sumaeth, S. Thammanoon, Colloids Surf. A Physicochem. Eng. Asp. 384, 519 (2008)
D.L. Liao, B.Q. Liao, J. Photochem. Photobiol. A Chem. 187, 363 (2007)
L. Jing, X. Sun, W. Cai, Z. Xu, Y. Du, H. Fu, J. Phys. Chem. Solids 64, 615 (2003)
F.A. Deorsola, D. Vallauri, Powder Technol. 190, 304 (2009)
W. Changhua, Z. Xintong, Sh Changlu, Zh Yanli, Y. Jikai, S. Panpan, L. Xueping, L. Hong, L. Yichun, X. Tengfeng, W. Dejun, J. Colloid Interface Sci. 363, 157 (2011)
A. Naumenko, I. Gnatiuk, N. Smirnova, A. Eremenko, Thin Solid Films 520, 4541 (2012)
S. Music, M. Gotic, M. Ivanda, S. Popovic, A. Turkovic, R. Trojko, A. Sekulic, K. Furic, Mater. Sci. Eng. B47, 33 (1997)
M. Gartner, R. Scurtu, A. Ghita, M. Zaharescu, M. Modreanu, C. Trapalis, M. Kokkoris, G. Kordas, Thin Solid Films 455, 417 (2004)
M.U. Nizam, S.U.A. Shibly, R. Ovali, S. Islam, M.M.R. Mazumder, M.S. Islam, M.J. Uddin, O. Gulseren, E. Bengu, Photochem. Photobiol. A Chem. 254, 25 (2013)
C.M. Mohan, A.K. Tripathi, M.K. Single, S.P. Gairola, S.N. Pandey, A. Aqrawal, Chem. Phys. Lett. 555, 182 (2013)
S. Schattauer, B. Reinhold, S. Albrecht, Ch. Fahrenson, M. Schubert, S. Janietz, D. Neher, Colloid Polym. Sci. 290, 1843 (2012)
I. Stambolova, V. Blaskov, S. Vassilev, M. Shipochka, A. Loukanov, Bull. Mater. Sci. 35(4), 645 (2012)
C.Y. Jimmy, Y. Jiaguo, H. Wingkei, J. Zitao, Z. Lizhi, Chem. Mater. 14, 3808 (2002)
F.B. Li, X.Z. Li, Chemosphere 48, 1103 (2002)
A. Amtout, R. Leonelli, Solid State Commun. 84(3), 349 (1992)
L.G.J.D. Haart, G. Blasse, J. Solid State Chem. 61, 135 (1986)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dastan, D., Londhe, P.U. & Chaure, N.B. Characterization of TiO2 nanoparticles prepared using different surfactants by sol–gel method. J Mater Sci: Mater Electron 25, 3473–3479 (2014). https://doi.org/10.1007/s10854-014-2041-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-014-2041-9