Abstract
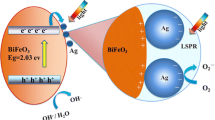
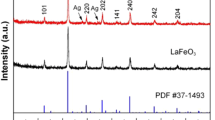
BiFeO3 particles with different morphologies and sizes were synthesized via a hydrothermal process, where the morphology and size was tailored by using different KOH concentrations in precursor solution. The samples prepared at n(KOH) = 3, 4.5, 6, and 7.5 M are composed, respectively, of octahedron-shaped particles (500–600 nm), cube-like particles (200–500 nm), irregular spherical agglomerates (9–16 μm) formed from disk-like grains with diameter of 1.4–2.8 μm and thickness of 0.2 μm, and cuboid-shaped particles with length-to-width ratio of 1.4:1–3.5:1 and width size ranging from 80 to 280 nm. Ag nanoparticles were deposited on the surface of BiFeO3 particles by a chemical reduction method to produce Ag@BiFeO3 nanocomposites. The photocatalytic activity of prepared samples was evaluated by degrading rhodamine B under simulated sunlight irradiation. It is demonstrated that Ag-decorated BiFeO3 particles exhibit an enhanced photocatalytic activity compared to bare BiFeO3 particles. This can be explained by the effective transfer of photogenerated electrons from the conduction band of BiFeO3 to Ag nanoparticles and hence increased availability of holes for the photocatalytic reaction. Hydroxyl radicals were detected by the photoluminescence technique using terephthalic acid as a probe molecule and are found to be produced over the irradiated BiFeO3 and Ag@BiFeO3 photocatalysts; especially, an enhanced yield is observed for the latter.










Similar content being viewed by others
References
A. Mills, R.H. Davies, D. Worsley, Chem. Soc. Rev. 22, 417 (1993)
M.R. Hoffmann, S.T. Martin, W. Choi, D.W. Bahemann, Chem. Rev. 95, 69 (1995)
C. Tabares-Munoz, J.P. Rivera, A. Monnier, H. Schmid, Jpn. J. Appl. Phys. 24, 1051 (1985)
P. Fischer, M. Polomska, I. Sosnowska, M. Szymanski, J. Phys. C 13, 1931 (1980)
J. Wang, J.B. Neaton, H. Zheng, V. Nagarajan, S.B. Ogale, B. Liu, D. Viehland, V. Vaithyanathan, D.G. Schlom, U.V. Waghmare, N.A. Spaldin, K.M. Rabe, M. Wuttig, R. Ramesh, Science 299, 1719 (2003)
T. Soltani, M.H. Entezari, Ultrason. Sonochem. 20, 1245 (2013)
J. Wei, H.Y. Li, S.C. Mao, C. Zhang, Z. Xu, B. Dkhil, J. Mater. Sci. Mater. Electron. 23, 1869 (2012)
J. He, R.Q. Guo, L. Fang, W. Dong, F.G. Zheng, M.R. Shen, Mater. Res. Bull. 48, 3017 (2013)
S. Li, Y.H. Lin, B.P. Zhang, Y. Wang, C.W. Nan, J. Phys. Chem. C 114, 2903 (2010)
F. Gao, X.Y. Chen, K.B. Yin, S.A. Dong, Z.F. Ren, F. Yuan, T. Yu, Z.G. Zou, J.M. Liu, Adv. Mater. 19, 2889 (2007)
S. Li, J.M. Zhang, M.G. Kibria, Z. Mi, M. Chaker, D.L. Ma, R. Nechache, F. Rosei, Chem. Commun. 49, 5856 (2013)
A.P. Zhang, J.Z. Zhang, J. Alloys Compd. 491, 631 (2010)
R. Adhikari, S. Malla, G. Gyawali, T. Sekino, S.W. Lee, Mater. Res. Bull. 48, 3367 (2013)
C.L. Yu, K. Yang, W.Q. Zhou, Q.Z. Fan, L.F. Wei, J.C. Yu, J. Phys. Chem. Solids 74, 1714 (2013)
G.L. Newton, J.R. Milligan, Radiat. Phys. Chem. 75, 473 (2006)
E. Shi, C.R. Cho, M.S. Jang, S.Y. Jeong, H.J. Kim, J. Mater. Res. 9, 2914 (1994)
S.J. Clark, J. Robertson, Appl. Phys. Lett. 90, 132903 (2007)
K. Takahashi, N. Kida, M. Tonouchi, Phys. Rev. Lett. 96, 117402 (2006)
J. Zhang, M.A. Gondal, W. Wei, T.N. Zhang, Q.Y. Xu, K. Shen, J. Alloys Compd. 530, 107 (2012)
J. He, R.Q. Guo, L.A. Fang, W. Dong, F.G. Zheng, M.R. Shen, Mater. Res. Bull. 48, 3017 (2013)
P. Kamat, J. Phys. Chem. C 111, 2834 (2007)
Y.L. Zhang, A.M. Schultz, P.A. Salvador, G.S. Rohrer, J. Mater. Chem. 21, 4168 (2011)
Acknowledgments
This work was supported by the National Natural Science Foundation of China (Grant No. 51262018) and the Hongliu Outstanding Talents Foundation of Lanzhou University of Technology (Grant No. J201205).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Di, L.J., Yang, H., Hu, G. et al. Enhanced photocatalytic activity of BiFeO3 particles by surface decoration with Ag nanoparticles. J Mater Sci: Mater Electron 25, 2463–2469 (2014). https://doi.org/10.1007/s10854-014-1896-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-014-1896-0