Abstract
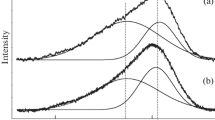
The secondary electron emission (SEE) properties of sulfur-incorporated nanocomposite carbon (n-C) films were studied. Maximum SEE yield (δ max) values obtained ranged from 3.27 to 6.98, which are between those for graphite (δ max ~ 1) and high purity diamond films in their as-grown condition (δ max ~ 9), and are consistent with the composite nature of the films. It was found that δ max values of n-C films are mostly determined by the atomic oxygen concentration on the surface of the films, which appears to control the probability of escape of the secondary electrons from the surface of the films, as inferred by employing Ascarelli’s model for SEE (J Appl Phys 89:689, 2001). Also, mean escape depth values for the secondary electrons were obtained using this model, and their significance as bulk parameters for the films is discussed.





Similar content being viewed by others
References
D.M. Trucchi, C. Scilletta, E. Cappelli, P.G. Merli, S. Zoffoli, G. Mattei, P. Ascarelli, Diam. Relat. Mater. 15, 827 (2006). doi:10.1016/j.diamond.2005.12.038
A. Shih, J. Yater, P. Pehrsson, J. Butler, C. Hor, R. Abrams, J. Appl. Phys. 82, 1860 (1997). doi:10.1063/1.365990
J.E. Yater, A. Shih, J. Appl. Phys. 87, 8103 (2000). doi:10.1063/1.373505
H.J. Hopman, J. Verhoeven, P.K. Bachmann, Diam. Relat. Mater. 9, 1238 (2000). doi:10.1016/S0925-9635(99)00238-1
G. Morell, A. González-Berríos, B.R. Weiner, S. Gupta, J. Mater. Sci. Mater. Electron. 17, 443 (2006). doi:10.1007/s10854-006-8090-y
S. Gupta, B.R. Weiner, G. Morell, J. Mater. Res. 18, 363 (2003). doi:10.1557/JMR.2003.0047
D.W. Marquardt, J. Soc. Ind. Appl. Math. 11, 431 (1963). doi:10.1137/0111030
R.J. Nemanich, J.T. Glass, G. Luckovsky, R.E. Schroder, J. Vac. Sci. Technol. A 6, 1783 (1988). doi:10.1116/1.575297
P. Ascarelli, E. Cappelli, F. Pinzari, M.C. Rossi, S. Salvatori, P.G. Merli, A. Migliori, J. Appl. Phys. 89, 689 (2001). doi:10.1063/1.1326854
S. Ruben, Handbook of the Elements (Open Court Publishing Company, La Salle, Illinois, 1985), p. 17
A. Shih, J.E. Yater, in Properties, Growth and Applications of Diamond, ed. by M.H. Nazaré, A.J. Neves (Inspec, London, 2001), pp. 82–91
Acknowledgments
Research supported in part by NASA Training Grant NNG05GG78H (PR Space Grant) and NASA Cooperative Agreements NNX07AO30A (PR NASA EPSCoR Core), and NASA Grant No. NNX08BA48A (NASA CNM URC). The authors would like to acknowledge Mr. Oscar Resto for his help with the SEE system, Dr. Esteban Fachini for XPS measurements, Dr. Ram Katiyar for Raman spectroscopy, and Dr. Carlos Cabrera for AFM.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
González-Berríos, A., Makarov, V.I., Goenaga-Vázquez, Y. et al. Secondary electron emission from nanocomposite carbon films. J Mater Sci: Mater Electron 20, 996–1000 (2009). https://doi.org/10.1007/s10854-008-9822-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-008-9822-y