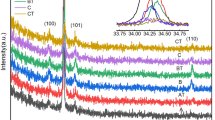
Abstract
The effect of titanium (Ti) doping on the crystal structure, phase, surface/interface chemistry, microstructure and optical band gap of gallium oxide (Ga2O3) (GTO) films is reported. The Ti content was varied from 0 to ~ 5 at% in co-sputtering, using Ga2O3 ceramic and Ti metal targets, deposited GTO films produced. The sputtering power to the Ti target was varied in the range of 0–100 W, while keeping the sputtering power to Ga2O3 constant at 100 W, to produce GTO films with 0–5 at% Ti. The Ti-incorporation-induced effects were significant for the crystal structure, phase, surface/interface chemistry and morphology, which in turn induce changes in the band gap. The high-resolution core-level X-ray photoelectron spectroscopy (XPS) analyses confirm that the Ga ions exist as Ga3+ in both intrinsic Ga oxide and GTO films. However, XPS data reveal the formation of Ga2O3–TiO2 films with the presence of Ti4+ ions with increasing Ti sputtering power, i.e., higher Ti contents in GTO. Evidence for the formation of nanocrystalline Ga2O3–TiO2 films was also found in the structural analyses performed using electron microscopy and grazing incidence X-ray diffraction. Significant band gap reduction (Eg ~ 0.9 eV) occurs in GTO films with increasing Ti dopant concentration from 0 to 5 at%. A correlation between the Ti dopant concentration, surface/interface chemistry, microstructure and band gap of GTO films is established.









Similar content being viewed by others
References
Pearton SJ, Yang J, Cary PH, Ren F, Kim J, Tadjer MJ, Mastro MA (2018) A review of Ga2O3 materials, processing, and devices. Appl Phys Rev 5:011301
Rubio EJ, Mates TE, Manandhar S, Nandasiri M, Shutthanandan V, Ramana CV (2016) Tungsten incorporation into gallium oxide: crystal structure, surface and interface chemistry, thermal stability, and interdiffusion. J Phys Chem C 120:26720–26735
Rubio EJ, Ramana CV (2013) Tungsten-incorporation induced red-shift in the bandgap of gallium oxide thin films. Appl Phys Lett 102:19193
Ogita M, Higo K, Nakanishi Y, Hatanaka Y (2001) Ga2O3 thin film for oxygen sensor at high temperature. Appl Surf Sci 175–176:721–725
Fleischer M, Höllbauer L, Meixner H (1994) Effect of the sensor structure on the stability of Ga2O3 sensors for reducing gases. Sens Actuators B 18:119–124
Guo D, An Y, Cui W, Cui W, Zhi Y, Zhao X, Lei M, Li L, Li P, Wu Z, Tang W (2016) Epitaxial growth and magnetic properties of ultraviolet transparent Ga2O3/(Ga1-xFex)2O3 multilayer thin films. Sci Rep 6:25166
Guo D, Li P, Wu Z, Cui W, Zhao X, Lei M, Li L, Tang W (2016) Inhibition of unintentional extra carriers by Mn valence change for high insulating devices. Sci Rep 6:24190
Suzuki R, Nakagomi S, Kokubun Y, Arai N, Ohira S (2009) Enhancement of responsivity in solar-blind β-Ga2O3 photodiodes with a Au Schottky contact fabricated on single crystal substrates by annealing. Appl Phys Lett 94:222102–222103
Patil SB, Kim IY, Gunjakar JL, Oh SM, Eom T, Kim H, Hwang SJ (2015) Phase tuning of nanostructured gallium oxide via hybridization with reduced graphene oxide for superior anode performance in li-ion battery: an experimental and theoretical study. ACS Appl Mater Interfaces 7:18679–18688
Qian L-X, Wu Z-H, Zhang Y-Y, Lai PT, Liu X-Z, Li Y-R (2017) Ultrahigh-responsivity, rapid-recovery, solar-blind photodetector based on highly nonstoichiometric amorphous gallium oxide. ACS Photonics 4:2203–2211
López I, Lorenz K, Nogales E, Méndez B, Piqueras J, Alves E, García JA (2014) Study of the relationship between crystal structure and luminescence in rare-earth-implanted Ga2O3 nanowires during annealing treatments. J Mater Sci 49:1279–1285. https://doi.org/10.1007/s10853-013-7811-x
Higashiwa M, Sasaki K, Kuramata A, Masui T, Amakoshi SY (2014) Development of gallium oxide power devices. Phys Status Solidi A 211:21–26
López I, Nogales E, Méndez B, Piqueras J, Peche A, Ramirez-Castellanos J, Gonzalez-Calet JM (2013) Influence of Sn and Cr doping on morphology and luminescence of thermally grown Ga2O3 nanowires. J Phys Chem C 117:3036–3045
Dakhel AA (2012) Structural, optical, and opto-dielectric properties of W-doped Ga2O3 thin films. J Mater Sci 47:3034–3039. https://doi.org/10.1007/s10853-011-6134-z
Manandhar S, Ramana CV (2017) Direct, functional relationship between structural and optical properties in titanium-incorporated gallium oxide nanocrystalline thin films. Appl Phys Lett 110:061902
Battu AK, Manandhar S, Shutthanandan V, Ramana CV (2017) Controlled optical properties via chemical composition tuning in molybdenum-incorporated β-Ga2O3 nanocrystalline films. Chem Phys Lett 684:363–367
Kumar SS, Rubio EJ, Noor-A-Alam M, Martinez G, Manandhar S, Shuthanandan V (2013) Structure, morphology, and optical properties of amorphous and nanocrystalline gallium oxide thin films. J Phys Chem C 117:4194–4200
Tang X, Huang X, Huang Y, Gou Y, Pastore J, Yang Y, Xiong Y, Qian J, Brock JD, Lu J, Abruna HD, Zhuan L (2018) High-performance Ga2O3 anode for lithium ion batteries. ACS Appl Mater Interfaces 10:5519–5526
Wang X, Zeng X, Cao D (2018) Biomass-derived nitrogen-doped porous carbons (NPC) and NPC/polyaniline composites as high performance supercapacitor materials. Eng Sci 1:55–63
Du W, Wang X, Zhan J, Sun X, Kang L, Jiang F, Zhang X, Shao Q, Dong M, Liu H, Murugadoss V, Guo Z (2019) Biological cell template synthesis of nitrogen-doped porous hollow carbon spheres/MnO2 composites for high-performance asymmetric supercapacitors. Electrochim Acta 296:907–915
Idrees M, Batool S, Kong J, Zhuang Q, Liu H, Shao Q, Lu N, Feng Y, Wujcik EK, Gao Q, Ding T, Wei R, Guo Z (2019) Polyborosilazane derived ceramics-nitrogen sulfur dual doped graphene nanocomposite anode for enhanced lithium ion batteries. Electrochim Acta 296:925–937
Qi H, Teng M, Liu M, Liu S, Li J, Ya H, Teng C, Huang Z, Liu H, Shao Q, Umar A, Ding T, Gao Q, Guo Z (2019) Biomass-derived nitrogen-doped carbon quantum dots: highly selective fluorescent probe for detecting Fe3+ ions and tetracyclines. J Colloid Interface Sci 539:332–341
Deng W, Kang T, Liu H, Zhang J, Wang N, La N, Ma Y, Umar A, Guo Z (2018) Potassium hydroxide activated and nitrogen doped graphene with enhanced supercapacitive behavior. Sci Adv Mater 10:937–949
Shindume L, Zhao Z, Wang N, Lu H, Umar A, Zhang J, Wu T, Guo Z (2019) Enhanced photocatalytic activity of B, N-codoped TiO2 by a new molten nitrate process. J Nanosci Nanotechnol 19:839–849
Huang J, Li Y, Cao Y, Peng F, Cao Y, Liu H, Guo Z (2018) Hexavalent chromium removal over magnetic carbon nanoadsorbents: synergistic effect of fluorine and nitrogen co-doping. J Mater Chem A 6:13062–13074
Mayer M, SIMNRA (1999) A simulation program for the analysis of NRA, RBS and ERDA. In: AIP Conf. Proc. AIP, pp 541–544
Ghosh SC, Biesinger MC, LaPierre RR, Kruse P (2007) X-ray photoelectron spectroscopic study of the formation of catalytic gold nanoparticles on ultraviolet-ozone oxidized GaAs(100) substrates. J Appl Phys 101:114322
Naumkin AV, Kraut-Vass A, Gaarenstroom SW, Powell CJ (2012) NIST Standard Reference database 20
Hollinger G, Skheyta-Kabbani R, Gendry M (1994) Oxides on GaAs and InAs surfaces: an x-ray-photoelectron-spectroscopy study of reference compounds and thin oxide layers. Phys Rev B 49:11159
Syed N, Zavabeti A, Mohiuddin M, Zhang Y Wang, Datta RS, Atkin P, Carey J, Tan C, van Emdes J (2017) Sonication-assisted synthesis of gallium oxide suspensions featuring trap state absorption: test of photochemistry. Adv Funct Mater 27:1702295
Ramana CV, Rubio E, Barraza C, Miranda Gallardo A, McPeak S, Kotru S, Grant J (2014) Chemical bonding, optical constants, and electrical resistivity of sputter-deposited gallium oxide thin films. Appl Phys 115:043508
Trinchi A, Kaciulis S, Pandolfi L, Ghantasala MK, Li YX, Wlodarski W, Viticoli S, Comini E, Sberveglieri G (2004) Characterization of Ga2O3 based MRISiC hydrogen gas sensors. Sens Actuators B 103:129–135
Ou S-L, Wuu D-S, Fu Y-C, Liu S-P, Horng R-H, Liu L, Feng Z-C (2012) Growth and etching characteristics of gallium oxide thin films by pulsed laser deposition. Mater Chem Phys 133:700–705
Bharti B, Kumar S, Lee H-N, Kumar R (2016) Formation of oxygen vacancies and Ti3+ state in TiO2 thin film and enhanced optical properties by air plasma treatment. Sci Rep 6:32355
Xu N, Liu L, Sun X, Liu X, Han D, Wang Y, Han R, Kang J, Yu B (2008) Characteristics and mechanism of conduction/set process in TiN/ZnO/Pt resistance switching random-access memories. Appl Phys Lett 92:232112
Hussain H, Tocci G, Woolcot T, Torrelles X, Pang CL, Humphery DS, Yim CM, Grinter DC, Cabailh G, ikondoa O, Lindsay R, Zegenhang J, Michaelidas A, Thornton G (2016) Structure of a model TiO2 photocatalytic interface. Nat Mater 16:461
González-Elipe A, Munuera G, Espinos J, Sanz J (1989) Compositional changes induced by 3.5 keV Ar + ion bombardment in Ni-Ti oxide systems: a comparative study. Surf Sci 220:368–380
Massaro CC, Rotolo P, De Riccardis F, Milella E, Napoli A, Wieland M, Textor M, Spencer N (2002) Comparative investigation of the surface properties of commercial titanium dental implants. Part I: chemical composition. J Mater Sci Mater Med 13:535–548
Tao J, Luttrell T, Batzill M (2011) A two-dimensional phase of TiO2 with a reduced bandgap. Nat Chem 3:296–300
Gupta R, Sen S (1975) Calculation of multiplet structure of core p-vacancy levels II. Phys Rev B 12:15
Leedy KD, Chabak KD, Vasilyev V, Look DC, Boeckl JJ, Brown JL, Tetlak SE, Green AJ (2017) Highly conductive homoepitaxial Si-doped Ga2O3 films on (010) β-Ga2O3 by pulsed laser deposition. Appl Phys Lett 111:012103
Ramana CV, Baghmar G, Rubio EJ, Hernandez MJ (2013) Optical constants of amorphous, transparent titanium-doped tungsten oxide thin films. ACS Appl Mater Interfaces 5:4659–4666
Kalidindi N, Manciu F, Ramana CV (2011) Crystal structure, phase, and electrical conductivity of nanocrystalline W0.95Ti0.05O3 thin films. ACS Appl Mater Interfaces 3:863–868
Geller S (1960) Crystal structure of -Ga2O3. J Chem Phys 33:676–684
Egerton RF (2005) Physical principles of electron microscopy. Springer, Berlin
Battu AK, Manandhar S, Ramana CV (2018) Nanomechanical characterization of titanium incorporated gallium oxide nanocrystalline thin films. Mater Today Nano 2:7–14
Acknowledgements
The authors acknowledge, with pleasure, the support from the National Science Foundation (NSF) with Grant No. ECCS-1509653. CVR also acknowledges the NSF-PREM Grant #DMR-1827745.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Manandhar, S., Battu, A.K., Tan, S. et al. Effect of Ti doping on the crystallography, phase, surface/interface structure and optical band gap of Ga2O3 thin films. J Mater Sci 54, 11526–11537 (2019). https://doi.org/10.1007/s10853-019-03663-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-019-03663-w