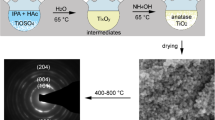
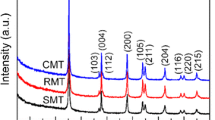
Abstract
Template-free synthesis of TiO2 architectures with controlled morphology evolution has been developed through solvothermal reaction in 1,4-dioxane. By simply varying the molar ratio of the concentrated HCl over Titanium isopropoxide (TTIP) from 0 to 5.0, series of morphologies including nanoparticle-built microspheres, nanoparticle-built microspheres decorated with nanorods, nanorod cauliflowers, and nanorod dendrites have been obtained. The influence of several key factors on the morphology control of TiO2 has been systematically investigated. These parameters include the mass (molar) ratio of HCl/TTIP, solvothermal reaction temperature and time, acid species (concentrated nitric acid), and solvent type (tetrahydrofuran and 1,3-dioxane). The mechanism for the formation of the TiO2 architectures with controlled morphology evolution has been discussed. The application of the TiO2 architectures as water splitting photocatalyst and lithium–ion battery anode has been demonstrated. And the corresponding structure–property correlation has been discussed.


















Similar content being viewed by others
References
Bai J, Zhou B (2014) Titanium dioxide nanomaterials for sensor applications. Chem Rev 114:10131. doi:10.1021/cr400625j
Bai Y, Mora-Sero I, De Angelis F, Bisquert J, Wang P (2014) Titanium dioxide nanomaterials for photovoltaic applications. Chem Rev 114:10095. doi:10.1021/cr400606n
Kapilashrami M, Zhang Y, Liu Y-S, Hagfeldt A, Guo J (2014) Probing the optical property and electronic structure of TiO2 nanomaterials for renewable energy applications. Chem Rev 114:9662. doi:10.1021/cr5000893
Ma Y, Wang X, Jia Y, Chen X, Han H, Li C (2014) Titanium dioxide-based nanomaterials for photocatalytic fuel generations. Chem Rev 114:9987. doi:10.1021/cr500008u
Rajh T, Dimitrijevic NM, Bissonnette M, Koritarov T, Konda V (2014) Titanium dioxide in the service of the biomedical revolution. Chem Rev 114:10177. doi:10.1021/cr500029g
Sanchez C, Belleville P, Popall M, Nicole L (2011) Applications of advanced hybrid organic-inorganic nanomaterials: from laboratory to market. Chem Soc Rev 40:696. doi:10.1039/c0cs00136h
Chen HH, Nanayakkara CE, Grassian VH (2012) Titanium dioxide photocatalysis in atmospheric chemistry. Chem Rev 112:5919. doi:10.1021/cr3002092
Wang J, Xie J, Jiang Y, Zhang J, Wang Y, Zhou Z (2015) Preparation of mesoporous TiO2-B nanowires from titanium glycolate and their application as an anode material for lithium-ion batteries. J Mater Sci 50:6321. doi:10.1007/s10853-015-9172-0
Wen P, Cai C, Zhong H, Hao L, Xu X (2015) A simple way to synthesize anatase with high thermal stability. J Mater Sci 50:5944. doi:10.1007/s10853-015-9117-7
Chen XB, Li C, Gratzel M, Kostecki R, Mao SS (2012) Nanomaterials for renewable energy production and storage. Chem Soc Rev 41:7909. doi:10.1039/c2cs35230c
Froschl T, Hormann U, Kubiak P et al (2012) High surface area crystalline titanium dioxide: potential and limits in electrochemical energy storage and catalysis. Chem Soc Rev 41:5313. doi:10.1039/c2cs35013k
Hu J, Chen M, Fang XS, Wu LW (2011) Fabrication and application of inorganic hollow spheres. Chem Soc Rev 40:5472. doi:10.1039/c1cs15103g
Ren Y, Ma Z, Bruce PG (2012) Ordered mesoporous metal oxides: synthesis and applications. Chem Soc Rev 41:4909. doi:10.1039/c2cs35086f
Zhou ZY, Tian N, Li JT, Broadwell I, Sun SG (2011) Nanomaterials of high surface energy with exceptional properties in catalysis and energy storage. Chem Soc Rev 40:4167. doi:10.1039/c0cs00176g
Asahi R, Morikawa T, Irie H, Ohwaki T (2014) Nitrogen-doped titanium dioxide as visible-light-sensitive photocatalyst: designs, developments, and prospects. Chem Rev 114:9824. doi:10.1021/cr5000738
Coppens P, Chen Y, Trzop E (2014) Crystallography and properties of polyoxotitanate nanoclusters. Chem Rev 114:9645. doi:10.1021/cr400724e
De Angelis F, Di Valentin C, Fantacci S, Vittadini A, Selloni A (2014) Theoretical studies on anatase and less common TiO2 phases: bulk, surfaces, and nanomaterials. Chem Rev 114:9708. doi:10.1021/cr500055q
Liu G, Yang HG, Pan J, Yang YQ, Lu GQ, Cheng H-M (2014) Titanium dioxide crystals with tailored facets. Chem Rev 114:9559. doi:10.1021/cr400621z
Wang X, Li Z, Shi J, Yu Y (2014) One-dimensional titanium dioxide nanomaterials: nanowires, nanorods, and nanobelts. Chem Rev 114:9346. doi:10.1021/cr400633s
Lee KT, Cho J (2011) Roles of nanosize in lithium reactive nanomaterials for lithium ion batteries. Nano Today 6:28. doi:10.1016/j.nantod.2010.11.002
Rolison DR, Long JW, Lytle JC et al (2009) Multifunctional 3D nanoarchitectures for energy storage and conversion. Chem Soc Rev 38:226. doi:10.1039/b801151f
Lai XY, Halpert JE, Wang D (2012) Recent advances in micro-/nano-structured hollow spheres for energy applications: from simple to complex systems. Energy Environ Sci 5:5604. doi:10.1039/c1ee02426d
Wu D, Zhu F, Li J et al (2012) Monodisperse TiO2 hierarchical hollow spheres assembled by nanospindles for dye-sensitized solar cells. J Mater Chem 22:11665. doi:10.1039/c2jm30786c
Sun Z, Kim JH, Zhao Y et al (2011) Rational design of 3D dendritic TiO2 nanostructures with favorable architectures. J Am Chem Soc 133:19314. doi:10.1021/ja208468d
Li W, Wu ZX, Wang JX, Elzatahry AA, Zhao DY (2014) A perspective on mesoporous TiO2 materials. Chem Mater 26:287. doi:10.1021/cm4014859
Cargnello M, Gordon TR, Murray CB (2014) Solution-phase synthesis of titanium dioxide nanoparticles and nanocrystals. Chem Rev 114:9319. doi:10.1021/cr500170p
Liu YD, Goebl J, Yin YD (2013) Templated synthesis of nanostructured materials. Chem Soc Rev 42:2610. doi:10.1039/c2cs35369e
Liu J, Liu F, Gao K, Wu JS, Xue DF (2009) Recent developments in the chemical synthesis of inorganic porous capsules. J Mater Chem 19:6073. doi:10.1039/b900116f
Cheng YJ, Gutmann JS (2006) Morphology phase diagram of ultrathin anatase TiO2 films templated by a single PS-b-PEO block copolymer. J Am Chem Soc 128:4658. doi:10.1021/ja0562853
Rawolle M, Niedermeier MA, Kaune G et al (2012) Fabrication and characterization of nanostructured titania films with integrated function from inorganic-organic hybrid materials. Chem Soc Rev 41:5131. doi:10.1039/c2cs15321a
Li D, Qin Q, Duan XC, Yang JQ, Guo W, Zheng WJ (2013) General one-pot template-free hydrothermal method to metal oxide hollow spheres and their photocatalytic activities and lithium storage properties. ACS Appl Mater Interfaces 5:9095. doi:10.1021/am4024059
Liao J-Y, Lei B-X, Kuang D-B, Su C-Y (2011) Tri-functional hierarchical TiO2 spheres consisting of anatase nanorods and nanoparticles for high efficiency dye-sensitized solar cells. Energy Environ Sci 4:4079. doi:10.1039/c1ee01574e
Xu F, Wu Y, Zhang XY, Gao ZY, Jiang K (2012) Controllable synthesis of rutile TiO2 nanorod array, nanoflowers and microspheres directly on fluorine-doped tin oxide for dye-sensitised solar cells. Micro Nano Lett 7:826. doi:10.1049/mnl.2012.0398
Rui Y, Li Y, Zhang Q, Wang H (2013) Size-tunable TiO2 nanorod microspheres synthesised via a one-pot solvothermal method and used as the scattering layer for dye-sensitized solar cells. Nanoscale 5:12574. doi:10.1039/c3nr04462a
Shao F, Sun J, Gao L, Yang S, Luo J (2011) Template-free synthesis of hierarchical TiO2 structures and their application in dye-sensitized solar cells. ACS Appl. Mater. Interfaces 3:2148. doi:10.1021/am200377g
Shen J, Wang H, Zhou Y, Ye N, Li G, Wang L (2012) Anatase/rutile TiO2 nanocomposite microspheres with hierarchically porous structures for high-performance lithium-ion batteries. RSC Adv 2:9173. doi:10.1039/c2ra20962d
Shen J, Wang H, Zhou Y, Ye N, Wang L (2012) Continuous hollow TiO2 structures with three-dimensional interconnected single crystals and large pore mesoporous shells for high-performance lithium-ion batteries. CrystEngComm 14:6215. doi:10.1039/c2ce25833a
Zhou Y, Wang X, Wang H et al (2014) Enhanced dye-sensitized solar cells performance using anatase TiO2 mesocrystals with the Wulff construction of nearly 100% exposed 101 facets as effective light scattering layer. Dalton Trans 43:4711. doi:10.1039/c3dt53010h
Yu X, Wang H, Liu Y et al (2013) One-step ammonia hydrothermal synthesis of single crystal anatase TiO2 nanowires for highly efficient dye-sensitized solar cells. J Mater Chem A 1:2110. doi:10.1039/c2ta00494a
Lai L-L, Wu J-M (2014) A facile synthesis of hierarchical TiO2 for dye adsorption and photocatalysis. RSC Adv 4:36212. doi:10.1039/c4ra04790g
Sarkar D, Ghosh CK, Chattopadhyay KK (2012) Morphology control of rutile TiO2 hierarchical architectures and their excellent field emission properties. Cryst Eng Commun 14:2683. doi:10.1039/c2ce06392a
Sun ZQ, Kim JH, Zhao Y et al (2011) Rational design of 3D dendritic TiO2 nanostructures with favorable architectures. J Am Chem Soc 133:19314. doi:10.1021/ja208468d
Fu X, Wang B, Ren Z, Fan C, Qian G, Wang Z (2014) Controllable synthesis of TiO2 hierarchical nanostructures and their applications in lithium ion batteries. RSC Adv 4:42772. doi:10.1039/c4ra08474h
Wang Y, Wang Q, Zhan X, Wang F, Safdar M, He J (2013) Visible light driven type II heterostructures and their enhanced photocatalysis properties: a review. Nanoscale 5:8326. doi:10.1039/c3nr01577g
Zhang QF, Uchaker E, Candelaria SL, Cao GZ (2013) Nanomaterials for energy conversion and storage. Chem Soc Rev 42:3127. doi:10.1039/c3cs00009e
Acknowledgements
This research is funded by the Natural Science Foundation of China (51103172), the Zhejiang Non-profit Technology Applied Research Program (2013C33190), the open Project of the Beijing National Laboratory for Molecular Science (20140138), and Ningbo Key Laboratory of Polymer Materials. The authors would like to thank Dr. Dingyi Tong for the help with sketch preparation and discussion.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Cheng, T., Zhang, G., Xia, Y. et al. Template-free synthesis of titania architectures with controlled morphology evolution. J Mater Sci 51, 3941–3956 (2016). https://doi.org/10.1007/s10853-015-9713-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-015-9713-6