Abstract
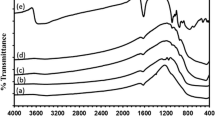
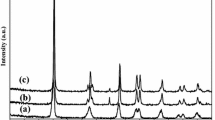
A convenient method for synthesizing highly photocatalytic activity PANI/TiO2–Fe3+ nanocomposite was developed. The effect of calcination temperature on the phase composition of TiO2 nanopowder was investigated. It was found that higher temperature could promote the formation of rutile phase. The nanocomposite was characterized by atomic force microscopy (AFM), transmission electron microscopy (TEM), infrared spectroscopy (IR) and X-ray diffraction (XRD). The results indicated that the nanohybrid was composed of TiO2, Fe3+ and PANI. The photocatalytic property of the nanocomposite was evaluated by the degradation of methyl orange. In the presence of this catalyst, the degradation rate of methyl orange of 95.2% and 70.3% could be obtained under the UV and sunlight irradiation within 30 min, respectively. The apparent rate constant was 5.64 × 10−2 which is better than that of the Degussa P25.









Similar content being viewed by others
References
Fujishima A, Honda K (1972) Nature 37:238
Guo XJ, Yuan L, Qi YL, Li XE, Yang W, Gao JZ (2004) Rare Metals 23:352
Xu ZL, Yang QJ, Xie C, Yan WJ, Du YG, Gao ZM, Zhang JH (2005) J Mater Sci 40:1539, DOI: 10.1007/s10853-005-0599-6
Yu JG (2004) Rare Metals 23:289
Yan XL, He J, Evans DG, Duan X, Zhu YX (2005) Appl Catal B Environ 55:243
Khanna PK, Lonkar SP, Subbarao VVVS, JUN KW (2004) Mater Chem Phys 87:49
Alan GM, Arthur JE (1995) Synth Met 69:85
Somani PR, Marimuthu R, Mulik UP, Sainkar SR, Amalnerkar DP (1999) Synth Met 106:45
Arroyo R, Córdoba G, Padilla J, Lara VH (2002) Mater Lett 54:397
Spurr RA, Myers H (1957) Anal Chem 29:760
Langford JA, Wilson AJC (1978) J Appl Crystallogr 11:102
Klong HP, Alexander LE (1954) In: X-ray diffraction procedures for crystalline and amorphous solids. Wiley, New York, p 491
Sclafani A, Herrman JM (1996) J Phys Chem 100:13655
Li XW, Chen W, Bian CQ, He JB, Xu N, Xue G (2003) Appl Surf Sci 217:16
Yu JG, Zhao XJ (2000) J Catal 21:213 (in Chinese)
Sonawane RS, Kale BB, Dongare MK (2004) Mater Chem Phys 85:52
Piscopo A, Robert D, Weber JV (2001) J Photochem Photobiol A Chem 139:253
Linsebigler AL, Lu GQ, Yates JT (1995) Chem Rev 95:735
Měšt’ánková H, Mailhot G, Jirkovský J, Krýsa J, Bolte M (2005) Appl Catal B Environ 57:257
Vaschetto ME, Monkman AP, Springborg M (1999) J Mol Struct (Theochem) 468:181
Acknowledgements
This work was supported in part by the Combined Project between the Educational Commission and the Economic Commission of Gansu Province (99CX-04, 0310B-08), the Natural Science Foundation of Gansu Province (3ZS041-A25–028), EYTP of MOE. China, and the Invention Project of Science & Technology (KJCXGC-01, NWNU), China.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gao, J., Li, S., Yang, W. et al. Synthesis of PANI/TiO2–Fe3+ nanocomposite and its photocatalytic property. J Mater Sci 42, 3190–3196 (2007). https://doi.org/10.1007/s10853-006-1353-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-006-1353-4