Abstract
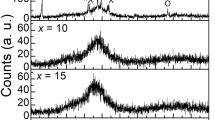
Series of glass based on the (80 − x)TeO2–20ZnO–(x)Er2O3 system (0.5 mol% ≤ x ≤ 2.5 mol%) has successfully been made by melt quenching technique. The optical properties of glass have been investigated by means of IR and Raman spectroscopy. It is observed that as the Er2O3 content is being increased, the sharp IR absorption peaks are consistently shifted from 650 to 672 cm−1 while the Raman shift intensity around 640–670 cm−1 is decreases but increases around 720–740 cm−1. It is found out that both phenomenons are related to the structural changes between the stretching vibration mode of TeO4 tbp and TeO3 tp, and bending vibration mode of Te–O bonds in the glass linkages.


Similar content being viewed by others
References
El-Mallawany R (ed) (2002) Tellurite glasses handbook: physical properties and data. CRC Press LLC
Bǘrger H, Kneipp K, Hobert H, Vogel W (1992) J Non-Cryst Solids 151:134
Sidebottom DL, Hruschka MA, Potter BG, Brow RK (1997) J Non-Cryst Solids 222:282
Sun K (1988) In: Preparation and characterization of rare earth glasses. Thesis, Brown University
Marjanovic S, Toulouse J, Jain H, Sandmann C, Dierolf V, Kortan AR, Kopylov N, Ahrens RG (2003) J Non-Cryst Solids 322:311
Uhlmann DR, Kreidl NJ (eds) (1983) Glass: science and technology, vol. 1. Academics Press, New York
Neindre LL, Jiang S, Hwan BC, Luo T, Watson J, Peyghambarian N (1999) J Non-Cryst Solids 255:97
Charton P, Thomas P, Armand P (2003) J Non-Cryst Solids 321:81
Nukui A, Taniguchi T, Miyata M (2001) J Non-Cryst Solids 293–295:260
Sahar MR, Noordin N (1995) J Non-Cryst Solids 184:137
Sahar MR, Jehbu AK, Karim MM (1997) J Non-Cryst Solids 213&214:164
Aida K, Benino Y, Dimitrov V, Komatsu T, Sato R (2000) J Am Ceram Soc 83(5):1192
Sahar MR, Sulhadi, Rohani MS (2005) In: The Digest of 2005 International Symposium on Glass. Shanghai PRC, 10–14 April 2005, pp SA1–SA4
Liu HS, Chin TS, Yung SW (1997) Mater Chem Phys 50:1
Xia H, Nie Q, Zhang J, Wang J (2003) Mater Lett 4446:1
Hu L, Jiang Z (1996) Phys Chem Glasses 37(1):19
Nazabal V, Todoroki S, Nukui A, Matsumoto T, Suehara S, Hondo T, Araki T, Inoue S, Rivero C, Cardinal T (2003) J Non-Cryst Solids 325:85
Li H, Su Y, Sundaram SK (2001) J Non-Cryst Solids 293–295:402
Jaba N, Mermet A, Duval E, Champagnon B (2005) J Non-Cryst Solids 351:833
Acknowledgements
The authors wish to thank the Ministry of Science, Technology and Innovation for their financial support under Vot 74532. We would also thanks to UTM for their continuous support to this project.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sahar, M.R., Sulhadi, K. & Rohani, M.S. Spectroscopic studies of TeO2–ZnO–Er2O3 glass system. J Mater Sci 42, 824–827 (2007). https://doi.org/10.1007/s10853-006-0095-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-006-0095-7