Abstract
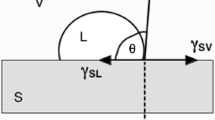
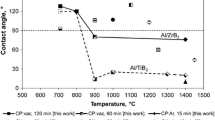
The wetting of (La0.6Sr0.4)(Co0.2Fe0.8)O3−δ substrates by Ag-CuO was investigated using the standard sessile drop technique, followed by metallographic examination of the quenched specimens. The addition of CuO substantially improves the wetting of these substrates by silver. The largest improvements in contact angle are observed at low CuO-content, apparently through the formation of a homogeneous silver-copper oxide liquid at the area of sessile drop/substrate contact. At a critical composition of x CuO~ 8 mol% the mechanism of wetting changes, undergoing a transition initiated by the formation of two immiscible liquids within the molten sessile drop.
Similar content being viewed by others
References
G. R. DOUGHTY and H. HIND, Key Eng. Mater. 122–124 (1996) 145.
J. W. STEVENSON, T. R. ARMSTRONG, R. D. CARNEIM, L. R. PEDERSON and W. J. WEBER, J. Electrochem. Soc. 143 (1996) 2722.
Y.-S. CHOU, J. W. STEVENSON, T. R. ARMSTRONG, J. S. HARDY, K. HASINSKA and L. R. PEDERSON, J. Mater. Res. 15 (2000) 1505.
C. C. SHÜLER, A. STUCK, N. BECK, H. KESER and U. TÅCK, J. Mater. Sci.: Mater. in Elec. 11 (2000) 389.
K. M. ERSKINE, A. M. MEIER and S. M. PILGRIM, J. Mater. Sci. 37 (2002) 1705.
K. S. WEIL and D. M. PAXTON, in Proceedings of the 26th Annual Conference on Composites, Advanced Ceramics, Materials, and Structures: A, edited by H.-T. Lin and M. Singh (The American Ceramic Society, Westerville, OH, 2002) p. 263.
R. S. ROTH, J. R. DENNIS and H. F. McMURDIE (eds.), “Phase Diagrams for Ceramists” (The American Ceramic Society, Westerville, OH, 1987) Vol. VI, p. 197.
A. PETRIC, P. HUANG and F. TIETZ, Sol. St. Ion. 135 (2000) 719.
D. A. MORTIMER and M. G. NICHOLAS, J. Mater. Sci. 8 (1973) 640.
Z. B. SHAO, K. R. LIU, L. Q. LIU, H. K. LIU and S. DOU, J. Am. Ceram. Soc. 76 (1993) 2663.
C. MOURE, D. GUTIERREZ, O. PENA and P. DURAN, J. Sol. St. Chem. 163 (2002) 377.
H. W. HSU, Y. H. CHANG, G. J. CHEN and K. J. LIN, Mater. Sci. and Eng. B B64 (1999) 180.
R. S. ROTH, J. R. DENNIS and H. F. McMURDIE (eds.), “Phase Diagrams for Ceramists” (The American Ceramic Society, Westerville, OH, 1987), Vol. V, p. 345.
J. W. CAHN, J. Chem. Phys. 66 (1977) 3667.
R. N. GRUGEL and A. HELLAWELL, Met. Trans. 12A (1981) 669.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Weil, K.S., Kim, J.Y. & Hardy, J.S. Interfacial analysis of (La0.6Sr0.4)(Co0.2Fe0.8)O3− δ substrates wetted by Ag-CuO. J Mater Sci 40, 2341–2348 (2005). https://doi.org/10.1007/s10853-005-1956-1
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10853-005-1956-1