Abstract
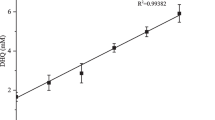
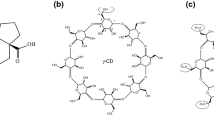
Inclusion complexes of three different lengths of polyamine modified β-cyclodextrins (β-CDs) (hosts 1–3) with baicalein (BC) were prepared by a solution stirring method, and characterized by UV–Vis spectroscopy, proton nuclear magnetic resonance, scanning electron microscopy, thermogravimetric analysis, and X-ray diffractometry. The results demonstrated that 1:1 inclusion complexes of hosts 1–3 with BC are present. The binding ability of hosts 1–3 towards BC increased according to the sequence H1 > H2 > H3, which was in good agreement with the decreasing chain lengths of these hosts. When compared to BC, the inclusion complexes produced 508-, 451- and 323-fold enhancement in water solubility, respectively. At low concentrations, inclusion complexes 1/BC and 3/BC were observed with a better capacity of scavenging when compared to that of free BC. These findings reflected that the complexation strengthened the antioxidant activity of BC. Moreover, the solubility in water and resistance to oxidation of hosts 1–3/BC were satisfactory, making it possible candidates for application in herbal medicine and as healthcare products.











Similar content being viewed by others
References
Shen, Y.C., Chiou, W.F., Chou, Y.C., Chen, C.F.: Mechanisms in mediating the anti-inflammatory effects of baicalin and baicalein in human leukocytes. Eur. J. Pharmacol. 465, 171–181 (2003). https://doi.org/10.1016/S0014-2999(03)01378-5
Wu, J.A., Attele, A.S., Zhang, L., Yuan, C.S.: Anti-HIV activity of medicinal herbs: usage and potential development. Am. J. Chin. Med. 29, 69–81 (2001). https://doi.org/10.1142/S0192415X01000083
Lee, H.Z., Leung, H.W., Lai, M.Y., Wu, C.H.: Baicalein induced cell cycle arrest and apoptosis in human lung squamous carcinoma CH27 cells. Anticancer Res. 25, 959–964 (2005). https://doi.org/10.1097/00006534-200009040-00020
Shimizu, I.: Sho-saiko-to: Japanese herbal medicine for protection against hepatic fibrosis and carcinoma. J. Gastroenterol. Hepatol. 15, 84–90 (2000). https://doi.org/10.1046/j.1440-1746.2000.02138.x
Kimata, M., Shichijo, M., Miura, T., Serizawa, I., Inagaki, N., Nagai, H.: Effects of luteolin, quercetin and baicalein on immunoglobulin E-mediated mediator release from human cultured mast cells. Clin. Exp. Allergy 30, 501–508 (2000). https://doi.org/10.1046/j.1365-2222.2000.00768.x
Kim, D.H., Jeon, S.J., Son, K.H., Jung, J.W., Lee, S., Yoon, B.H., Lee, J.J., Cho, Y.W., Cheong, J.H., Ko, K.H., Ryu, J.H.: The ameliorating effect of oroxylin A on scopolamine-induced memory impairment in mice. Neurobiol. Learn. Mem. 87, 536–546 (2007). https://doi.org/10.1016/j.nlm.2006.11.005
Gao, Z., Huang, K., Yang, X., Xu, H.: Free radical scavenging and antioxidant activities of flavonoids extracted from the radix of Scutellaria baicalensis Georgi. Biochim. Biophys. Acta BBA 1472, 643–650 (1999). https://doi.org/10.1016/S0304-4165(99)00152-X
Shao, Z.H., Vanden Hoek, T.L., Qin, Y., Becker, L.B., Schumacker, P.T., Li, C.Q., Dey, L..,Barth, E., Halpern, H., Rosen, G.M., Yuan, C.S.: Baicalein attenuates oxidant stress in cardiomyocytes. Am. J. Physiol. Heart Circ. Physiol. 282, H999–H1006 (2002). https://doi.org/10.1152/ajpheart.00163.2001
Gao, Z.H., Huang, K.X., Xu, H.B.: Protective effects of flavonoids in the roots of Scutellaria baicalensis Georgi against hydrogen peroxide-induced oxidative stress in HS-SY5Y cells. Pharmacol. Res. 43, 173–178 (2001). https://doi.org/10.1006/phrs.2000.0761
Liu, J., Qiu, L., Gao, J., Jin, Y.: Preparation, characterization and in vivo evaluation of formulation of baicalein with hydroxypropyl-β-cyclodextrin. Int. J. Pharm. 312, 137–143 (2006). https://doi.org/10.1016/j.ijpharm.2006.01.011
Zhou, Y., Dong, W., Ye, J., Hao, H., Zhou, J., Wang, R., Liu, Y.: A novel matrix dispersion based on phospholipid complex for improving oral bioavailability of baicalein: preparation, in vitro and in vivo evaluations. Drug Deliv. 24, 720–728 (2017). https://doi.org/10.1080/10717544.2017.1311968
Misiuk, W.: Investigation of inclusion complex of HP-γ-cyclodextrin with ceftazidime. J. Mol. Liq. 224, 387–392 (2016). https://doi.org/10.1016/j.molliq.2016.10.009
Li, W., Lu, B., Sheng, A., Yang, F., Wang, Z.: Spectroscopic and theoretical study on inclusion complexation of beta-cyclodextrin with permethrin. J. Mol. Struct. 981, 194–203 (2010). https://doi.org/10.1016/j.molstruc.2010.08.008
Li, J., Han, L., Chao, J.: Preparation and characterization of the inclusion complex of baicalein with γ-cyclodextrin: an antioxidant ability study. J. Incl. Phenom. Macrocycl. Chem. 73, 247–254 (2012). https://doi.org/10.1007/s10847-011-0048-x
Zhou, Q.N., Wei, X.H., Dou, W., Chou, G.X., Wang, Z.H.: Preparation and characterization of inclusion complexes formed between baicalein and cyclodextrins. Carbohyd. Polym. 95, 733–739 (2013). https://doi.org/10.1016/j.carbpol.2013.02.038
Gu, J., Chi, S.M., Zhao, Y., Zheng, P., Ruan, Q., Zhu, H.Y.: Inclusion complexes of coenzyme Q10 with polyamine-modified β-cyclodextrins: characterization, solubilization, and inclusion mode. Helv. Chim. Acta 94, 1608–1617 (2011). https://doi.org/10.1002/hlca.201100026
Liu, Y., Chen, G.S., Zhang, H.Y., Cao, D.X., Yuan, Y.J.: Inclusion complexation and solubilization of paclitaxel by bridged bis(β-cyclodextrin)s containing a tetraethylenepentaamino spacer. J. Med. Chem. 46, 4634–4637 (2003). https://doi.org/10.1021/jm034148f
Meo, P.L., D’Anna, F., Gruttadauria, M., Riela, S., Noto, R.: Synthesis and characterization of new polyamino-cyclodextrin materials. Carbohydr. Res. 347, 32–39 (2012). https://doi.org/10.1016/j.carres.2011.10.029
Liu, Y., Fan, X., Gao, L.: Synthesis and characterization of β-cyclodextrin based functional monomers and its copolymers with N-isopropylacrylamide. Macromol. Biosci. 3, 715–719 (2003). https://doi.org/10.1002/mabi.200300052
Tabushi, I., Shimizu, N., Sugimoto, T., Shiozuka, M., Yamamura, K.: Cyclodextrin flexibly capped with metal ion. J. Am. Chem. Soc. 99, 7100–7102 (1977). https://doi.org/10.1021/ja00463a073
Landy, D., Tetart, F., Truant, E., Surpateanu, G.: Development of a competitive continuous variation plot for the determination of inclusion compounds stoichiometry. J. Incl. Phenom. Macrocycl. Chem. 57(1–4), 409–413 (2007). https://doi.org/10.1007/s10847-006-9226-7
Yang, L.J., Wang, S.H., Zhou, S.Y., Zhao, F., Chang, Q., Li, M.Y., Chen, W., Yang, X.D.: Supramolecular system of podophyllotoxin and hydroxypropyl-β-cyclodextrin: characterization, inclusion mode, docking calculation, solubilization, stability and cytotoxic activity. Mater. Sci. Eng. C 76, 1136 (2017). https://doi.org/10.1016/j.msec.2017.03.197
Wang, L., Li, S., Tang, P., Yan, J., Xu, K., Li, H.: Characterization and evaluation of synthetic riluzole with β-cyclodextrin and 2,6-di-O-methyl-β-cyclodextrin inclusion complexes. Carbohydr. Polym. 129, 9–16 (2015). https://doi.org/10.1016/j.carbpol.2015.04.046
Xu, W.T., Zhang, F.F., Luo, Y.B., Ma, L.Y., Kou, X.H., Huang, K.L.: Antioxidant activity of a water-soluble polysaccharide purified from Pteridium aquilinum. Carbohydr. Res. 344, 217–222 (2009). https://doi.org/10.1016/j.carres.2008.10.021
Guo, M., Perez, C., Wei, Y.B., Rapoza, E., Su, G., Bou-Abdallahb, F., Chasteenb, N.D.: Iron-binding properties of plant phenolics and cranberry’s bio-effects. Dalton Trans. 43(43), 4951 (2007)
Benesi, H.A., Hidebrand, J.H.: A spectrophotometric investigation of the interaction of iodine with aromatic hydrocarbons. J. Am. Chem. Soc. 71, 2703–2707 (1949). https://doi.org/10.1021/ja01176a030
Tangm, P.X., Li, S.S., Wang, L.L., Yang, H.Q., Yan, J., Li, H.: Inclusion complexes of chlorzoxazone with β- and hydroxypropyl-β-cyclodextrin: characterization, dissolution, and cytotoxicity. Carbohydr. Polym. 131, 297–305 (2015). https://doi.org/10.1016/j.carbpol.2015.05.055
Ren, S.H., Zhang, J.Q., Yan, H.H., Zheng, X., Zhu, H.Y., Jin, Y., Lin, J.: Preparation, characterization, molecular docking and in vitro evaluation of two novel morin hydrate/CD inclusion complexes. J. Incl. Phenom. Macrocycl. Chem. 85, 317–328 (2016). https://doi.org/10.1007/s10847-016-0631-2
Aytac, Z., Ipek, S., Durgun, E., Uyar, T.: Antioxidant electrospun zein nanofibrous web encapsulating quercetin/cyclodextrin inclusion complex. J. Mater. Sci. 53, 1527–1539 (2018). https://doi.org/10.1007/s10853-017-1580-x
Acknowledgements
This work was supported by the National Natural Science Foundations (Nos. 21362046 and 21062030), which are gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Du, J., Zhao, L., Yang, S. et al. Preparation, characterization, solubilization and antioxidant activity of polyamine modified β-cyclodextrins with baicalein inclusion complexes. J Incl Phenom Macrocycl Chem 93, 203–213 (2019). https://doi.org/10.1007/s10847-018-0871-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-018-0871-4