Abstract
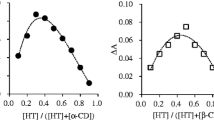
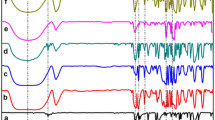
Complexation in solution between danazol and two different cyclodextrins [2-hydroxypropyl-β-cyclodextrin (HP-β-CD) and 2-hydroxypropyl-γ-cyclodextrin (HP-γ-CD)] was studied using phase solubility analysis, and one- and two-dimensional 1H-NMR. The increase of danazol solubility in the aqueous cyclodextrin solutions showed a linear relationship (AL profile). The apparent stability constant, K 1:1, of each complex was calculated and found to be 51.7 × 103 and 7.3 × 103 M−1 for danazol–HP-β-CD and danazol–HP-γ-CD, respectively. 1H-NMR spectroscopic analysis of varying ratios of danazol and the different cyclodextrins in a mixture of EtOD–D2O confirmed the 1:1 stoichiometry. Cross-peaks, from 2D ROESY 1H-NMR spectra, between protons of danazol and H3′ and H5′of cyclodextrins, which stay inside the cyclodextrin cavity, proved the formation of an inclusion complex between danazol and the cyclodextrins. For HP-β-CD, the inclusion complex is formed by entrance of the isooxazole and the A rings of danazol in the cyclodextrin cavity. For HP-γ-CD, two different inclusion structures may exist simultaneously in solution: one with the isooxazole and A ring in the cavity and the other with the C and D ring inside the cavity. DLS showed that self-aggregation of the CD’s was absent in the danazol HP-β-CD system up to a CD concentration of 10% and in the danazol HP-γ-CD system up to a CD concentration of 5%.









Similar content being viewed by others
References
Vercellini, P., Somigliana, E., Vigano, P., Abbiati, A., Barbara, G., Crosignani, P.G.: Endometriosis: current therapies and new pharmacological developments. Drugs 69, 649–675 (2009)
Maia, H., Casoy, J., Valente, J.: Is aromatase expression in the endometrium the cause of endometriosis and related infertility? Gynecol. Endocrinol. 25, 253–257 (2009)
Devalia, H.L., Layer, G.T.: Current concepts in gynaecomastia. Surgeon 7(2), 114–119 (2009)
Frank, M.M.: Hereditary angioedema. J. Allergy Clin. Immunol. 121, S398–S401 (2008)
Araujo, A., Lima, M.A., Syvia, M.T.T.: Human T-lymphotropic virus 1 neurologic disease. Curr. Treat. Options Neurol. 10, 193–200 (2008)
Daou, S., Federici, L., Zimmer, J., Maloisel, F., Serraj, K., Andrès, E.: Idiopathic thrombocytopenic purpura in elderly patients: a study of 47 cases from a single reference center. Eur. J. Intern. Med. 19, 447–451 (2008)
Chen, X., Vaughn, J.M., Yacaman, M.J., Williams III, R.O., Johnston, K.P.: Rapid dissolution of high-potency danazol particles produced by evaporative precipitation into aqueous solution. Pharm. Sci. 93, 1867–1878 (2004)
Badawy, S., Ghorab, M., Adeyeye, C.: Characterization and bioavailability of danazol-hydroxypropyl-β-cyclodextrin coprecipitates. Int. J. Pharm. 128, 45–54 (1996)
Brewster, M., Loftsson, T.: Cyclodextrins as pharmaceutical solubilizers. Adv. Drug Del. Rev. 59, 645–666 (2007)
Szejtli, J.: Introduction and general overview of cyclodextrin chemistry. Chem. Rev. 98, 1743–1754 (1998)
Mosher, G.L., Thompson, D.O.: Complexation cyclodextrin. In: Swarbrick, J. (ed.) Encyclopedia of Pharmaceutical Technology, pp. 671–696. Marcel Dekker, New York (2007)
Carrier, R.L., Miller, L.A., Ahmed, I.: The utility of cyclodextrin for enhancing oral bioavailability. J. Control. Release 123, 78–99 (2007)
Jadhav, G.S., Vavia, P.R.: Physicochemical, insilico and in vivo evaluation of danazol-β-cyclodextrin complex. Int. J. Pharm. 352, 5–16 (2007)
Djedäine, F., Lin, S.Z., Perly, B., Wouessidjewe, D.: High-field nuclear magnetic resonance techniques for the investigation of a β-cyclodextrin: indomethacin inclusion complex. J. Pharm. Sci. 79, 643–646 (1990)
Salvatierra, D., Jaime, C., Virgili, A., Sánchez-Ferrando, F.: Determination of the inclusion geometry for the β-cyclodextrin/benzoic acid complex by NMR and molecular modeling. J. Org. Chem. 61, 9578–9581 (1996)
Higuchi, T., Connors, K.A.: Phase solubility technique. Adv. Anal. Chem. Inst. 4, 212–217 (1965)
Keller, R.: The Computer Aided Resonance Assignment Tutorial, 1st edn. CANTINA Verlag, Goldau (2004)
Loftsson, T., Brewster, M.: Pharmaceutical applications of cyclodextrins. J. Pharm. Sci. 85, 1017–1025 (1996)
Uekama, K., Fujinaga, T., Otagiri, M., Yamasaki, M.: Inclusion complexations of steroid hormones with cyclodextrins in water and in solid phase. Int. J. Pharm. 10, 1–15 (1982)
Larsen, K.L., Acchmann, F.L., Wimmer, R., Stella, V.J., Kjølner, U.M.: Phase solubility and structure of the inclusion complexes of prednisolone and 6α-methylprednisolone with various cyclodextrin. J. Pharm. Sci. 94, 507–515 (2005)
Martin, A.: Complexation and protein binding. In: Martin, A. (ed.) Physical Pharmacy, 4th edn, pp. 251–283. Lippincott Williams and Wilkins, Baltimore (1993)
C Fernandes, C.M., Carhalvo, R.A., da Costa, S.P., Veiga, F.: Multimodal molecular encapsulation of nicardipine hydrochloride by β-cyclodextrin, hydroxypropyl-β-cyclodextrin and triacetyl-β-cyclodextrin in solution. Structural studies by 1H NMR and ROESY experiments. Eur. J. Pharm. Sci. 18, 285–296 (2003)
Ganza-Gonzalez, A., Vila-Jato, J.L., Anguiano-Igea, S., Otero-Espinar, F.J., Blanco-Méndez, J.: A proton nuclear magnetic resonance study of the inclusion complex of naproxen with β-cyclodextrin. Int. J. Pharm. 106, 179–185 (1994)
Másson, M., Loftsson, T., Jónsdóttir, S., Fridriksdóttir, H., Peterson, D.S.: Stabilisation of ionic drugs through complexation with non-ionic and ionic cyclodextrins. Int. J. Pharm. 164, 45–55 (1998)
Do, T.T., Nauwelaerts, K., Froeyen, M., Baudemprez, L., Van Speybroeck, M., Augustijns, P., Annaert, P., Martens, J., Van Humbeeck, J., Van den Mooter, G.: Comparison of the complexation between methylprednisolone and different cyclodextrins in solution by 1H-NMR and molecular modeling studies. J. Pharm. Sci. 99(9), 3863–3873 (2010)
Chankvetadze, B., Lomsadze, K., Burjanadze, N., Breitkeutz, J., Pinto, G., Chesse, M., Berganthal, K., Blaschke, G.: Comparative enantioseparations with native beta-cyclodextrin, randomly acetylated beta-cyclodextrin and heptakis-(2, 3-di-O-acetyl)-beta-cyclodextrin in capillary electrophoresis. Electrophoresis 24, 1083–1091 (2003)
Messner, M., Kurkov, S.V., Jansook, P., Loftsson, T.: Self-assembled cyclodextrin aggregates and nanoparticles. Int. J. Pharm. 387, 199–208 (2010)
Acknowledgments
A K.U. Leuven Industrial Research Fund (IOF) provided financial support for this study. MVS acknowledges the Institute for the Promotion of Innovation through Science and Technology in Flanders (IWT-Vlaanderen) for a PhD grant.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Thi, T.D., Nauwelaerts, K., Baudemprez, L. et al. Comparison between 2-hydroxypropyl-β-cyclodextrin and 2-hydroxypropyl-γ-cyclodextrin for inclusion complex formation with danazol. J Incl Phenom Macrocycl Chem 71, 137–147 (2011). https://doi.org/10.1007/s10847-010-9917-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-010-9917-y