Abstract
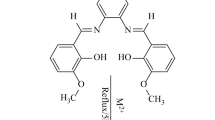
In this study, firstly, two single substitute novel ligands have been synthesized by reacting melamine with 3,4,-dihydroxybenzaldeyhde or 4-carboxybenzaldehyde. Then, eight new mono nuclear single substitute [Salen/Salophen Fe(III) and Cr(III)] complexes have been synthesized by reacting the ligands [2-(3,4-dihydroxybenzimino)-4,6-diamimo-1,3,5-triazine and 2-(4-carboxybenzimino)-4,6-diamimo-1,3,5-triazine)] with tetradentate Schiff bases N,N′-bis(salicylidene)ethylenediamine-(salenH2) or bis(salicylidene)-o-phenylenediamine-(salophen H2). And then, all ligands and complexes have been characterized by means of elementel analysis, FT-IR spectroscopy, 1H NMR, LC–MS, thermal analyses and magnetic suscebtibility measurements. Finally, metal ratios of the prepared complexes were determined using AAS. The complexes have also been characterized as disorted octahedral low-spin Fe(III) and Cr(III) bridged by catechol and COO− groups.







Similar content being viewed by others
References
Carmouna, M., Blanchoud, H., Teil, M.J., Blanchard, M., Chevreuil, M.: Triazines in the Marne and the Seine Rivers (France) longitudinal evolution and flows. Water Air Soil Pollut. 132, 1 (2001). doi:10.1023/A:1012017025060
Klenke, B., Stewart, M., Barrett, M.P., Brun, R., Gilbert, I.H.: Synthesis and biological evaluation of s-triazine substituted polyamines as potential new anti-trypanosomal drugs. J. Med. Chem. 44, 3440 (2001). doi:10.1021/jm010854+
Patel, H.S., Patel, V.C.: Polyimides containing s-triazine ring. Eur. Polym. J. 2263 (2001). doi:10.1016/S0014-3057(01)00107-0
Hoog, P., Gamez, P., Lüken, M., Roubeau, O., Krebs, B., Reedijk, J.: Hexanuclear copper(II) complex of a novel poly(pyridine) ligand exhibiting unique long distance ferromagnetic interactions through a nitrato-O, O′ bridge. Inorg. Chim. Acta 357, 213–218 (2004). doi:10.1016/S0020-1693(03)00428-6
Wang, X., Zhang, Q.: Synthesis, characterization, and cure properties of phosphorus-containing epoxy resins for flame retardance. Eur. Polym. J. 40, 385–395 (2004). doi:10.1016/j.eurpolymj.2003.09.023
Ashmawy, F.M., Ujaimi, A.R., McAuliffe, C.A., Parish, R.V., Pritchard, R.G.: A facile synthesis of [{Fe(salphen)}2O] (salphenH2 = N,N′-o-phenylenebis(salicylidineimine)) and the X-ray crystal structure of [{Fe(salphen)}2O] DMSO. Inorg. Chim. Acta 187, 155–158 (1991). doi:10.1016/s0020-1693(00)90388-8
Coggon, P., McPail, A.T., Gross, P.M., Mabbs, F.E., McLachlan, V.N.: Crystal and molecular structure and magnetic anisotropy of μ-oxo-bis-[N,N-ethylenebis(salicylidene iminato)iron(III)]-dichloromethane. J. Chem. Soc. A. 1014–1019 (1971). doi:10.1039/J19710001014
Dorfman, J.R., Girerd, J.J., Simhon, E.D., Stack, T.D.P., Holm, R.H.: Synthesis, structure, and electronic features of (.mu.-sulfido)bis[(N,N′-ethylenebis-(salicylaldiminato))iron(III)], [Fe(salen)]2S, containing the only authenticated example of the iron(III)–sulfur–iron(III) single bridge. Inorg. Chem. 23, 4407–4412 (1984). doi:10.1021/ic00194a002
Gerloch, M., McKenzie, E.D., Towl, A.D.C.: Crystal and molecular structure of μ-oxo-bis-[N,N′-ethylenebis(salicylideneiminato)iron(III)]-bispyridine. J. Chem. Soc. A. 2850–2858 (1969). doi: 10.1039/J19690002850
Elmali, A., Atakol, O., Svobodaand, I., Fuess, H.Z.: Structure of [N,N′-o-phenylenebis(salicylideneaminato)]iron(III) chloride as a five-coordinate monomer. Kristallographie 203, 273 (1993)
Kamenicek, J., Travnicek, Z., Sindelar, Z., Walla, J.: Redetermination of the structure of μ-oxo-bis[N,N′-o-phenylene-bis(salicylaldiminato) iron(III)]dimethyl sulfoxide solvate. Pol. J. Chem. 70, 854 (1996)
Marek, J., Smekal, Z., Travnicek, Z.: Structure of (μ-oxo)-bis[N,N′-ethylene-bis(salicylaldiminato)iron(III)]. Acta Univ. Palacki Olomuc., Fac. Rerum Nat. 35, 33 (1996)
Kopel, P., Sindelar, Z., Klicka, R.: Complexes of iron(III) salen and saloph Schiff bases with bridging dicarboxylic and tricarboxylic acids. Transit. Met. Chem. 23, 139 (1998). doi:10.1023/A:1006990925318
Wollmann, R.G., Hendrickson, D.N.: Reaction of.mu.-oxo-bridged iron(III) complexes with organic acids: a characterization of the products. Inorg. Chem. 17(4), 926–930 (1978). doi:10.1021/ic50182a026
Koç, Z.E., Uçan, H.İ.: Complexes of iron(III) salen and saloph Schiff bases with bridging 2,4,6-tris(2,5-dicarboxyphenylimino-4-formylphenoxy)-1,3,5-triazine and 2,4,6-tris(4-carboxyphenylimino-4′-formylphenoxy)-1,3,5-triazine. Transit. Met. Chem. 32, 597–602 (2007). doi:10.1007/s11243-007-0213-7
Uysal, Ş., Uçan, H.İ.: The synthesis and characterization of melamine based Schiff bases and its trinuclear[salen/salophenFe(III)] and [salen/salophenCr(III)] capped complexes. J. Incl. Phenom. Macrocycl. Chem. 65(3), 299–304 (2009). doi:10.1007/s10847-009-9581-2
Uysal, Ş., Uçan, H.İ.: The synthesis and characterization of 2,4,6-tris(3,4-dihydroxybenzimino)-1,3,5-triazine and its [salen/salophenFe(III)] and [salen/salophenCr(III)] capped complexes. J. Incl. Phenom. Macrocycl. Chem. 65(3), 403–409 (2009). doi:10.1007/s10847-009-9598-6
Barone, G., Silvestri, A., La Manna, G.: DFT computational study on FeIII-N,N′-ethylene-bis(salicylideneiminato) derivatives. J. Mol. Struct.: THEOCHEM. 715, 79–83 (2005)
Woldemariam, G.A., Mandal, S.S.: Iron(III)-salen damages DNA and induces apoptosis in human cell via mitochondrial pathway. J. Inorg. Biochem. 102, 740–747 (2008)
Juturu, V., Komorowski, J.R.: Letter to the editor. Am. J. Clin. Nutr. 78, 192–193 (2003)
Ryan, G.J., Wanko, N.S., Redman, A.R., Cook, C.B.: Chromium as adjunctive treatment for type 2 diabetes. Ann. Pharmacother. 37, 876–885 (2003). doi:10.1345/aph.1C304
Govindaraju, K., Ramasami, T., Ramaswamy, D.: Chymotrypsin-catalyzed hydrolysis of chromium(III) derivatives of insulin: evidence for stabilization of the protein through interactions with metal ions. J. Inorg. Biochem. 35, 127 (1989). doi:10.1016/0162-0134(89)80005-4
Shrivastava, H.Y., Devaraj, S.N., Nair, B.U.: A Schiff base complex of chromium(III): an efficient inhibitor for the pathogenic and invasive potential of Shigella dysenteriae. J. Inorg. Biochem. 98, 387–392 (2004). doi:10.1016/j.jinorgbio.2003.11.012
Canali, L., Sherrington, D.C.: Utilisation of homogeneous and supported chiral metal(salen) complexes in asymmetric catalysis. Chem. Soc. Rev. 28, 85–93 (1999). doi:10.1039/a806483k
Yoon, T.P., Jacobsen, E.N.: Privileged chiral catalysts. Science. 299, 1691–1693 (2003). doi:10.1126/science.1083622
Cozzi, P.G.: Metal–Salen Schiff base complexes in catalysis: practical aspects. Chem. Soc. Rev. 33, 410–421 (2004). doi:10.1039/b307853c
Larrow, J.F., Jacobsen, E.N.: Asymmetric processes catalyzed by chiral (Salen)metal complexes. Top. Organomet. Chem. 6, 123–152 (2004). doi:10.1007/b11772
Yamashita, Y., Katsuki, T.: Asymmetric Diels-Alder reaction using oxo(salen)manganese(V) complex as a Lewis acid catalyst. Synlett. 8, 829–830 (1995). doi:10.1055/s-1995-5106
Srinivasan, K., Michaud, P., Kochi, J.K.: Epoxidation of olefins with cationic (salen)manganese(III) complexes. The modulation of catalytic activity by substituents. J. Am. Chem. Soc. 108, 2309–2320 (1986). doi:10.1021/ja00269a029
Samsel, E.G., Srinivasan, K., Kochi, J.K.: Mechanism of the chromium-catalyzed epoxidation of olefins. Role of oxochromium(V) cations. J. Am. Chem. Soc. 107, 7606–7617 (1985). doi:10.1021/ja00311a064
Yoon, H., Burrows, C.J.: Catalysis of alkene oxidation by nickel salen complexes using sodium hypochlorite under phase-transfer conditions. J. Am. Chem. Soc. 110, 4087–4089 (1988). doi:10.1021/ja00220a086
Luts, T., Frank, R., Suprun, W., Fritzsche, S., Hey-Hawkins, E., Papp, H.: Epoxidation of olefins catalyzed by novel Mn(III) and Mo(IV)-Salen complexes immobilized on mesoporous silica gel Part II: study of the catalytic epoxidation of olefins. J. Mol. Catal. A Chem. 273, 250–258 (2007). doi:10.1016/j.molcata.2007.04.010
Kopel, P., Sindelar, Z., Klicka, R.: Complexes of iron(III) salen and saloph Schiff bases with bridging dicarboxylic and tricarboxylic acids. Transit. Met. Chem. 23, 139 (1998). doi:10.1023/A:1006990925318
Kopel, P., Sindelar, Z., Biler, M., Klicka, R.: Complexes of iron(III) salen and saloph Schiff bases bridged by dicarboxylic acids. Pol. J. Chem. 72(9), 2060–2066 (1998)
Gembicky, M., Boca, R., Renz, F.: A heptanuclear Fe(II)–Fe(III)6 system with twelve unpaired electrons. Inorg. Chem. Commun. 3, 662–665 (2000). doi:10.1016/S1387-7003(00)00160-X
Tahmassebi, D.C., Sasaki, T.: Synthesis of a new trialdehyde template for molecular imprinting. J. Org. Chem. 59, 679–681 (1994). doi:10.1021/jo00082a034
Yang, G.M., Liao, D.Z., Jiang, Z.H., Yan, S.P., Wang, G.L.: Novel oxalate-bridged trinuclear FeIII–MII–FeIII (M = Cu and VO) complexes: synthesis and magnetism. Transit. Met. Chem. 23, 313–315 (1998). doi:10.1023/A:1015777304222
Uysal, Ş., Coşkun, A., Koç, Z.E., Uçan, H.İ.: Synthesis and characterization of a new dioxime and its heterotrinuclear BF2 + capped complexes. J. Macromol. Scı. A. 45(9), 727–732 (2008). doi:10.1080/10601320802222616
Khalil, S.M., Emara, A.A.: Asymmetric Schiff base (N2O3) complexes as ligands towards Mn(II), Fe(III) and Co(II), synthesis and characterization. J. Coord. Chem. 55, 17–32 (2002). doi:10.1080/00958970211871
Karatas, I., Ucan, H.I.: The synthesis of biphenylglyoxime and bis(phenylglyoxime) and their complexes with Cu(II), Ni(II) and Co(II). Synth. React. Inorg. Met.-Org. Chem. 28, 383–391 (1998). doi:10.1080/00945719809349362
Ucan, H.I., Karatas, I., Irez, G., Deveci, M.A., Mercimek, B.: The synthesis of four new Schiff bases and some of their transition metal complexes. Synth. React. Inorg. Met.-Org. Chem. 28(3), 331–338 (1998). doi:10.1080/00945719809349358
Koç, Z.E., Uçan, H.İ.: Complexes of iron(III) and chrom(III) salen and saloph Schiff bases with bridging 2,4,6-tris(4-nitrophenylimino-4′-formylphenoxy)-1,3,5-triazine. J. Macromol. Scı. A. 45(12), 1072–1077 (2008). doi:10.1080/10601320802458087
Dotson, D.L.: Part 1. The mesomorphic properties of aryloxy-s-triazines and their analogs, Part 2. The synthesis and polymerization behavior of α-aminonitriles and related compounds. PhD. Dissertation, Virginia Polytechnic Institute and State University, Virginia (1996)
El-Metwally, N.M., Gabr, I.M., El-Asmy, A.A.: Spectral, magnetic, electrical and thermal studies on malonyl bis(thiosemicarbazide) complexes. Transit. Met. Chem. 31, 71–78 (2006). doi:10.1007/s11243-005-6347-6
Brzyska, W., Krol, A.: Properties and thermal decomposition in air atmosphere of Co(II), Ni(II), Cu(II) and Zn(II) benzene-1,2-dioxyacetates. Thermochim. Acta 223, 241–249 (1993). doi:10.1016/0040-6031(93)80140-6
Acknowledgment
The authors would like to acknowledge the Scientific Research Projects (BAP) of Selcuk University for supporting this study through a grant: 2005/5201005.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Uysal, Ş., Uçan, H.İ. The synthesis and characterization of single substitute melamine cored Schiff bases and their [Fe(III) and Cr(III)] complexes. J Incl Phenom Macrocycl Chem 68, 165–173 (2010). https://doi.org/10.1007/s10847-010-9762-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-010-9762-z