Abstract
Purpose
It has been reported that elevation of asymmetric dimethylarginine (ADMA) concentration is involved in atrial fibrillation (AF) process. The purpose of this study was to determine whether ADMA concentration is related to recurrences of AF after catheter ablation.
Methods
A total of one hundred and thirty-eight patients with persistent AF (49.91 ± 7.23 years), but without known heart disease, who underwent successful mapping and catheter ablation were included. Serum ADMA levels were measured 1 day before the catheter ablation.
Results
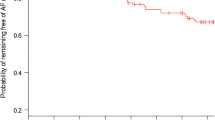
After a mean follow-up period of 285 days (range, 98–651 days), 80 (58%) patients underwent the AF recurrence. Comparing with no AF recurrence group, the serum ADMA concentration, left atrial diameter, and persistent duration of AF in patients with AF recurrence group were significantly increased. Left atrial diameter, persistent duration of AF, and the number of non-pulmonary vein ectopy ablation were increased when serum ADMA concentration was elevated. In addition, AF recurrences were significantly increased when serum ADMA concentration was elevated. A multivariable Cox regression analysis demonstrated that the elevated ADMA concentration [hazard ration (95% CI), 4.59 (1.81–11.62); P = 0.001], the left atrial diameter [1.35 (1.18–1.55), P < 0.001], and the number of non-pulmonary vein ectopy ablation [1.94 (1.18–3.18), P = 0.009] were independent factors associated with the AF recurrence after catheter ablation.
Conclusions
This study demonstrated that (1) serum ADMA concentrations were associated with non-PV ectopies and (2) increased serum ADMA concentrations were associated with higher rate of AF recurrence after catheter ablation in patients with persistent AF, supporting our hypothesis that ADMA might participate in the process of atrial remodeling.

Similar content being viewed by others
References
Calkins, H., Brugada, J., Packer, D. L., Cappato, R., Chen, S. A., Crijns, H. J., et al. (2007). HRS/EHRA/ECAS expert consensus statement on catheter and surgical ablation of atrial fibrillation: recommendations for personnel, policy, procedures and follow-up. Heart Rhythm, 4, 816–861.
Weerasooriya, R., Khairy, P., Litalien, J., Macle, L., Hocini, M., Sacher, F., et al. (2011). Catheter ablation for atrial fibrillation: are results maintained at 5 years of follow-up? Journal of the American College of Cardiology, 57(2), 160–166.
Allessie, M., Ausma, J., & Schotten, U. (2002). Electrical, contractile and structural remodeling during atrial fibrillation. Cardiovascular Research, 54(2), 230–246.
Frustaci, A., Chimenti, C., Bellocci, F., Morgante, E., Russo, M. A., & Maseri, A. (1997). Histological substrate of atrial biopsies in patients with lone atrial fibrillation. Circulation, 96, 1180–1184.
Liu, H., Xiufen, Qu, Liang, Z., Chen, W., Xia, W., & Song, Y. (2008). Variance of DDAH/PRMT/ADMA pathway in atrial fibrillation dogs. Biochemical and Biophysical Research Communications, 377, 884–888.
Xia, W., Qu, X., Yu, Y., Zhang, X., Feng, W., & Song, Y. (2008). Asymmetric dimethylarginine concentration and early recurrence of atrial fibrillation after electrical cardioversion. Pacing and Clinical Electrophysiology, 31, 1036–1040.
Maldonado, G., & Greenland, S. (1993). Simulation study of confounder-selection strategies. American Journal of Epidemiology, 138(11), 923–936.
Vallance, P., Leone, A., Calver, A., Collier, J., & Moncada, S. (1992). Accumulation of an endogenous inhibitor of NO synthesis in chronic renal failure. Lancet, 339, 572–575.
De Gennaro Colonna, V., Bianchi, M., Pascale, V., Ferrario, P., Morelli, F., Pascale, W., et al. (2009). Asymmetric dimethylarginine (ADMA): an endogenous inhibitor of nitric oxide synthase and a novel cardiovascular risk molecule. Medical Science Monitor, 15(4), RA91–RA101.
Cengel, A., Sahinarslan, A., Biberoğlu, G., Hasanoğlu, A., Tavil, Y., Tulmaç, M., et al. (2008). Asymmetrical dimethylarginine level in atrial fibrillation. Acta Cardiologica, 63(1), 33–37.
Lehto, M., Jurkko, R., Parikka, H., Mäntynen, V., Väänänen, H., Montonen, J., et al. (2009). Reversal of atrial remodeling after cardioversion of persistent atrial fibrillation measured with magnetocardiography. Pacing and Clinical Electrophysiology, 32(2), 217–223.
Novo, G., Guttilla, D., Fazio, G., Cooper, D., & Novo, S. (2009). The role of the renin-angiotensin system in atrial fibrillation and the therapeutic effects of ACE-Is and ARBS. British Journal of Clinical Pharmacology, 66(3), 345–351.
Li, J., Solus, J., Chen, Q., Rho, Y. H., Milne, G., Stein, C. M., et al. (2010). Role of inflammation and oxidative stress in atrial fibrillation. Heart Rhythm, 7(4), 438–444.
Van Wagoner, D. R. (2008). Oxidative stress and inflammation in atrial fibrillation: role in pathogenesis and potential as a therapeutic target. Journal of Cardiovascular Pharmacology, 52(4), 306–313.
Van Gelder, I. C., Crijns, H. J., Van Gilst, W. H., Verwer, R., & Lie, K. I. (1991). Prediction of uneventful cardioversion and maintenance of sinus rhythm from direct-current electrical cardioversion of chronic atrial fibrillation and flutter. The American Journal of Cardiology, 68(1), 41–46.
Berruezo, A., Tamborero, D., Mont, L., Benito, B., Tolosana, J. M., Sitges, M., et al. (2007). Pre-procedural predictors of atrial fibrillation recurrence after circumferential pulmonary vein ablation. European Heart Journal, 28(7), 836–841.
Skalidis, E. I., Zacharis, E. A., Tsetis, D. K., Pagonidis, K., Chlouverakis, G., Yarmenitis, S., et al. (2007). Endothelial cell function during atrial fibrillation and after restoration of sinus rhythm. The American Journal of Cardiology, 1, 99(9), 1258–1262.
Aldhoon, B., Melenovský, V., Peichl, P., & Kautzner, J. (2010). New insights into mechanisms of atrial fibrillation. Physiological Research, 59(1), 1–12.
Kourliouros, A., Savelieva, I., Kiotsekoglou, A., Jahangiri, M., & Camm, J. (2009). Current concepts in the pathogenesis of atrial fibrillation. American Heart Journal, 157(2), 243–252.
Yamada, T., Murakami, Y., Okada, T., Yoshida, N., Toyama, J., Yoshida, Y., et al. (2007). Plasma brain natriuretic peptide level after radiofrequency catheter ablation of paroxysmal, persistent, and permanent atrial fibrillation. Europace, 9, 770–774.
Kallergis, E. M., Manios, E. G., Kanoupakis, E. M., Mavrakis, H. E., Kolyvaki, S. G., Lyrarakis, G. M., et al. (2008). The role of the post-cardioversion time course of hs-CRP levels in clarifying the relationship between inflammation and persistence of atrial fibrillation. Heart, 94(2), 133–134.
Trocha, M., Szuba, A., Merwid-Lad, A., & Sozański, T. (2010). Effect of selected drugs on plasma asymmetric dimethylarginine (ADMA) levels. Die Pharmazie, 65(8), 562–571.
Schneider, M. P., Hua, T. A., Böhm, M., Wachtell, K., Kjeldsen, S. E., & Schmieder, R. E. (2010). Prevention of atrial fibrillation by renin-angiotensin system inhibition a meta-analysis. Journal of the American College of Cardiology, 55(21), 2299–2307.
Xia, W., Yin, Z., Li, J., Song, Y., & Qu, X. (2009). Effects of rosuvastatin on asymmetric dimethylarginine levels and early atrial fibrillation recurrence after electrical cardioversion. Pacing and Clinical Electrophysiology, 32(12), 1562–1566.
Lai, J., Zhou, D., Xia, S., Shang, Y., Want, L., Zheng, L., et al. (2009). Reduced testosterone levels in males with lone atrial fibrillation. Clinical Cardiology, 32(1), 43–46.
Korantzopoulos, P., Kolettis, T. M., Kountouris, E., Dimitroula, V., Karanikis, P., Pappa, E., et al. (2005). Oral vitamin C administration reduces early recurrence rates after electrical cardioversion of persistent atrial fibrillation and attenuates associated inflammation. International Journal of Cardiology, 10, 102(2), 321–326.
Kubes, P., & SuzukiM, G. D. N. (1991). Nitric oxide: an endogenous modulator of leukocyte adhesion. Proceedings of the National Academy of Sciences, 88, 4651–4655.
Sarkar, R., Meinberg, E. G., Stanley, J. C., Gordon, D., & Webb, R. C. (1996). Nitric oxide reversibly inhibits themigration of cultured vascular smooth muscle cells. Circulation Research, 78, 225–230.
Feng, Q., Song, W., Lu, X., Hamilton, J. A., Lei, M., Peng, T., et al. (2002). Development of heart failure and congenital septal defects in mice lacking endothelial nitric oxide synthase. Circulation, 106, 873–879.
Van Wagoner, D. R. (2003). Electrophysiological remodeling in human atrial fibrillation. Pacing and Clinical Electrophysiology, 26, 1572–1575.
Cooke, J. P. (2004). Asymmetrical dimethylarginine: the uber marker? Circulation, 109(15), 1813–1818.
Suda, O., Tsutsui, M., Morishita, T., Tasaki, H., Ueno, S., Nakata, S., et al. (2004). Asymmetric dimethylarginine produces vascular lesions in endothelial nitric oxide synthase-deficient mice: involvement of renin-angiotensin system and oxidative stress. Arteriosclerosis, Thrombosis, and Vascular Biology, 24, 1682–1688.
Natale, A., Pisanó, E., Beheiry, S., Richey, M., Leonelli, F., Fanelli, R., et al. (2000). Ablation of right and left atrial premature beats following cardioversion in patients with chronic atrial fibrillation refractory to antiarrhythmic drugs. The American Journal of Cardiology, 1,85(11), 1372–1375.
Schmitt, C., Ndrepepa, G., Weber, S., Schmieder, S., Weyerbrock, S., Schneider, M., et al. (2002). Biatrial multisite mapping of atrial premature complexes triggering onset of atrial fibrillation. The American Journal of Cardiology, 15,89(12), 1381–1387.
Lin, W. S., Tai, C. T., Hsieh, M. H., Tsai, C. F., Lin, Y. K., Tsao, H. M., et al. (2003). Catheter ablation of paroxysmal atrial fibrillation initiated by non-pulmonary vein ectopy. Circulation, 1,107(25), 3176–3183.
Stühlinger, M. (2007). Asymmetric dimethylarginine (ADMA): a novel cardiovascular risk factor? Wiener Medizinische Wochenschrift, 157(3–4), 57–60.
Szuba, A., & Podgorski, M. (2006). Asymmetric dimethylarginine (ADMA) a novel cardiovascular risk factor—evidence from epidemiological and prospective clinical trials. Pharmacological Reports, 58 Suppl, 16–20.
Allessie, M. A., Boyden, P. A., Camm, A. J., Kleber, A. G., Lab, M. J., Legato, M. J., et al. (2001). Pathophysiology and prevention of atrial fibrillation. Circulation, 103(5), 769–777.
Engelmann, M. D., & Svendsen, J. H. (2005). Inflammation in the genesis and perpetuation of atrial fibrillation. European Heart Journal, 26, 2083–2092.
Boos, C. J., Anderson, R. A., & Lip, G. Y. (2006). Is atrial fibrillation an inflammatory disorder? European Heart Journal, 27, 136–149.
Kurotobi, T., Iwakura, K., Inoue, K., Kimura, R., Okamura, A., Koyama, Y., et al. (2010). A pre-existent elevated C-reactive protein is associated with the recurrence of atrial tachyarrhythmias after catheter ablation in patients with atrial fibrillation. Europace, 12(9), 1213–1218.
Osmancik, P., Peroutka, Z., Budera, P., Herman, D., Stros, P., & Straka, Z. (2010). Changes in cytokine concentrations following successful ablation of atrial fibrillation. European Cytokine Network, 1, 21(4), 278–284.
Luan, Y., Guo, Y., Li, S., Yu, B., Zhu, S., Li, S., et al. (2010). Interleukin-18 among atrial fibrillation patients in the absence of structural heart disease. Europace, 12(12), 1713–1718.
Degener, S., Pattberg, S. V., Feuersenger, H., Bansmann, P. M., Shin, D. I., Krummenauer, F., et al. (2007). Plasma natriuretic peptide concentrations as a predictor for successful catheter ablation in patients with drug-refractory atrial fibrillation. Circulation Journal, 71(3), 313–320.
Shimano, M., Shibata, R., Inden, Y., Yoshida, N., Uchikawa, T., Tsuji, Y., et al. (2009). Reactive oxidative metabolites are associated with atrial conduction disturbance in patients with atrial fibrillation. Heart Rhythm, 6(7), 935–940.
Acknowledgments
This study was fully supported by the Key Science and Technology Program of Heilongjiang Province, China (no. GC10C305-1).
Conflict of interest
None declared
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, L., Xiufen, Q., Shuqin, S. et al. Asymmetric dimethylarginine concentration and recurrence of atrial tachyarrythmias after catheter ablation in patients with persistent atrial fibrillation. J Interv Card Electrophysiol 32, 147–154 (2011). https://doi.org/10.1007/s10840-011-9588-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10840-011-9588-7