Abstract
Background
The incidence, risk factors, and management of very high defibrillation thresholds (DFTs) during present-day implantable cardioverter defibrillator (ICD) testing are not well known.
Objectives
The purpose of this study was to assess (1) the incidence of very high DFTs and (2) the efficacy/safety of routinely adding a subcutaneous (SQ) array for these patients.
Methods
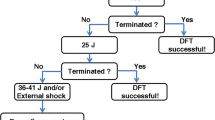
The study evaluated patients undergoing first-time ICD implantation at Southlake Regional Healthcare Centre from January 2006 to December 2007. All implanted ICDs had a maximal output of 35 J. Patients with DFTs greater than a 10-J safety margin from maximum output were considered to have very high readings and underwent SQ array insertion after other attempts at lowering DFT (group I). These patients were compared with the rest of the patients who had acceptable DFTs (group II) using both univariate and multivariate logistic regression analysis. Outcomes of array insertion were also assessed.
Results
A total of 313 patients underwent first-time ICD implantation during the analysis period. Of those, 16 (5.1%) had very high DFTs (group I). By univariate analysis, advanced New York Heart Association class (3 or 4), congestive heart failure hospitalization, non-ischemic cardiomyopathy, amiodarone use, implant of a biventricular device, and highest quartile of left ventricular (LV) chamber enlargement were all significant predictors of very high DFTs (p < 0.05). By multivariate analysis, only amiodarone use [odds ratio (OR) = 10.3, 95% confidence interval (95% CI) = 3.7–32.6] and being in the highest quartile for LV diastolic diameter [OR = 5.4, 95% CI = 1.4–20.8] predicted very high DFT. In all 16 cases, other methods to lower DFT prior to array insertion were attempted but failed for all patients: reversing shock polarity (n = 15), removing the superior vena cava coil (n = 14), reprogramming shock waveform (n = 9), and repositioning right ventricular lead (n = 9). Addition of the array successfully decreased DFT to within safety margin for all patients (33 ± 2 vs 21 ± 5 J, p = 0.02). Complication due to array insertion occurred in one patient (pneumothorax).
Conclusions
Very high DFTs occur in about 5% of patients undergoing ICD implantation and may be predicted by LV dilation and amiodarone use. SQ array insertion reliably corrects this problem over other interventions with a low rate of procedural complication.

Similar content being viewed by others
Abbreviations
- CHF:
-
Congestive heart failure
- DFT:
-
Defibrillation threshold
- EF:
-
Ejection fraction
- ICD:
-
Implantable cardioverter defibrillator
- LV:
-
Left ventricular
- NYHA:
-
New York Heart Association class
- SQ array:
-
Subcutaneous array
- SVC:
-
Superior vena cava
- VF:
-
Ventricular fibrillation
- VT:
-
Ventricular tachycardia
References
Pires, L. A., & Johnson, K. M. (2006). Intraoperative testing of the implantable cardioverter-defibrillator: how much is enough? Journal of Cardiovascular Electrophysiology, 17(2), 140–145.
Russo, A. M., Sauer, W., Gerstenfeld, E. P., et al. (2005). Defibrillation threshold testing: is it really necessary at the time of implantable cardioverter-defibrillator insertion? Heart Rhythm, 2(5), 456–461.
Gold, M. R., Higgins, S., Klein, R., et al. (2002). Efficacy and temporal stability of reduced safety margins for ventricular defibrillation: primary results from the Low Energy Safety Study (LESS). Circulation, 105(17), 2043–2048.
Kuhlkamp, V., Dornberger, V., Khalighi, K., et al. (1998). Effect of a single element subcutaneous array electrode added to a transvenous electrode configuration on the defibrillation field and the defibrillation threshold. Pacing and Clinical Electrophysiology, 21(12), 2596–2605.
Trusty, J. M., Hayes, D. L., Stanton, M. S., et al. (2000). Factors affecting the frequency of subcutaneous lead usage in implantable defibrillators. Pacing and Clinical Electrophysiology, 23(5), 842–846.
Ideker, R. E., Epstein, A. E., & Plumb, V. J. (2005). Should shocks still be administered during implantable cardioverter-defibrillator insertion? Heart Rhythm, 2(5), 462–463.
Theuns, D. A., Szili-Torok, T., & Jordaens, L. J. (2005). Defibrillation efficacy testing: long-term follow-up and mortality. Europace, 7(6), 509–515.
Mainigi, S. K., & Callans, D. J. (2006). How to manage the patient with a high defibrillation threshold. Heart Rhythm, 3(4), 492–495.
Day, J. D., Olshansky, B., Moore, S., et al. (2008). High defibrillation energy requirements are encountered rarely with modern dual-chamber implantable cardioverter-defibrillator systems. Europace, 10(3), 347–350.
Hodgson, D. M., Olsovsky, M. R., Shorofsky, S. R., et al. (2002). Clinical predictors of defibrillation thresholds with an active pectoral pulse generator lead system. Pacing and Clinical Electrophysiology, 25(4 Pt 1), 408–413.
Lubinski, A., Lewicka-Nowak, E., Zienciuk, A., et al. (2005). Clinical predictors of defibrillation threshold in patients with implantable cardioverter-defibrillators. Kardiologia Polska, 62(4), 317–328. discussion 329–331.
Shukla, H. H., Flaker, G. C., Jayam, V., et al. (2003). High defibrillation thresholds in transvenous biphasic implantable defibrillators: clinical predictors and prognostic implications. Pacing and Clinical Electrophysiology, 26(1 Pt 1), 44–48.
Birnie, D., Tung, S., Simpson, C., et al. (2008). Complications associated with defibrillation threshold testing: the Canadian experience. Heart Rhythm, 5(3), 387–390.
Hohnloser, S. H., Dorian, P., Roberts, R., et al. (2006). Effect of amiodarone and sotalol on ventricular defibrillation threshold: the optimal pharmacological therapy in cardioverter defibrillator patients (OPTIC) trial. Circulation, 114(2), 104–109.
Bollmann, A., Husser, D., & Cannom, D. S. (2005). Antiarrhythmic drugs in patients with implantable cardioverter-defibrillators. American Journal of Cardiovascular Drugs, 5(6), 371–378.
Schuger, C., Ellenbogen, K. A., Faddis, M., et al. (2006). Defibrillation energy requirements in an ICD population receiving cardiac resynchronization therapy. Journal of Cardiovascular Electrophysiology, 17(3), 247–250.
Kettering, K., Mewis, C., Dornberger, V., et al. (2004). Long-term experience with subcutaneous ICD leads: a comparison among three different types of subcutaneous leads. Pacing and Clinical Electrophysiology, 27(10), 1355–1361.
Zienciuk, A., Lubinski, A., Krolak, T., et al. (2007). Effects of shock polarity reversal on defibrillation threshold in an implantable cardioverter-defibrillator. Kardiologia Polska, 65(5), 495–500. discussion 501–492.
Keane, D., Aweh, N., Hynes, B., et al. (2007). Achieving sufficient safety margins with fixed duration waveforms and the use of multiple time constants. Pacing and Clinical Electrophysiology, 30(5), 596–602.
Blatt, J. A., Poole, J. E., Johnson, G. W., et al. (2008). No benefit from defibrillation threshold testing in the SCD-HeFT (Sudden Cardiac Death in Heart Failure Trial). Journal of the American College of Cardiology, 52(7), 551–556.
Author information
Authors and Affiliations
Corresponding author
Additional information
No financial support was received for this study. No potential conflicts of interest arise from the publication of this manuscript.
Rights and permissions
About this article
Cite this article
Verma, A., Kaplan, A.J., Sarak, B. et al. Incidence of very high defibrillation thresholds (DFT) and efficacy of subcutaneous (SQ) array insertion during implantable cardioverter defibrillator (ICD) implantation. J Interv Card Electrophysiol 29, 127–133 (2010). https://doi.org/10.1007/s10840-010-9511-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10840-010-9511-7