Abstract
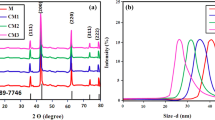
White nanoparticles of calcia-doped ceria were prepared from the precipitate by reacting CeCl3-CaCl2mixed solution with NaOH solution at pH 12 and the oxidation with hydrogen peroxide solution at 40∘C, followed by the calcination at 700∘C for 1 h. The sample before calcination contained significant amount of OH− in the lattice and was yellow, but the powders calcined above 700∘C were white, indicating that cation defect formed by replacing O2 − with OH− played as the color center. It is confirmed that calcia-doped ceria showed much lower photocatalytic activity as well as lower generation of singlet oxygen under UV light irradiation than those with titania and zinc oxide. Calcia-doped ceria particles were coated with amorphous silica by means of sol-gel reaction technique using hydrolysis of tetraethylorthosilicate (TEOS) or acid hydrolysis of sodium silicate. The silica coating by sol-gel reaction with TEOS was much more efficient for the reduction of catalytic activity of ceria for the oxidation of organic materials without loss of UV-shielding ability than that by acid hydrolysis of sodium silicate.
Similar content being viewed by others
References
R. Cai, K. Hashimoto, K. Itoh, Y. Kubota, and A. Fujita, Bull. Chem. Soc. Jpn., 64, 1268 (1991).
Y. Yamamoto, W.C. Dunlap, N. Imai, R. Mashita, R. Konaka, M. Inoue, Y. Hasegawa, and T. Miyoshi, Proceedings of 20th IFSCC Congress Canes, 1, 153 (1998).
S. Yabe and S. Momose, J. Soc. Cosmet. Chem. Jpn., 32, 372 (1998).
P. Chen and I. Chen, J. Am. Ceram. Soc., 76, 1577 (1993).
M. Hirano and E. Kato, J. Am. Ceram. Soc., 79, 777 (1996).
T. Masui, K. Fujikawa, K. Machida, T. Sakata, H. Mori, and G. Adachi, Chem. Mater., 9, 2197 (1997).
X. Yu, F. Li, X. Ye, X. Xin, and Z. Xue, J. Am. Ceram. Soc., 83, 964 (2000).
W. Stöber, A. Fink, and E. Bohn, J. Colloid Interf. Sci., 26, 62 (1968).
Y. Kobayashi, K. Misawa, M. Kobayashi, M. Takeda, M. Konno, M. Satake, Y. Kawazoe, N. Ohuchi, and A. Kasuya, Coll. Surf. A: Physicochem. Eng. Aspects, 242, 47 (2004).
Y. Kobayashi, K. Misawa, M. Takeda, M. Kobayashi, M. Satake, Y. Kawazoe, N. Ohuchi, A. Kasuya, and M. Konno, Coll. Surf. A: Physicochem. Eng. Aspects 251, 197 (2004).
J. Frank, J.V. Geil, and R. Freaso, Food Technol., 36, 71 (1982).
M.W. Laubli and P.A. Bruttel, J. Amer. Oil Chem. Soc., 63, 792 (1986).
C. Kato and K. Makabe, J. Jpn. Oil Chem. Soc., 42, 55 (1993).
A. Ahniyaz, T. Fujiwara, T. Fujino, and M. Yoshimura, J. Nanosci. Nanotechnol., 4, 233 (2004).
A. Ahniyaz, T. Watanabe, and M. Yoshimura, J. Phys. Chem. B, 109, 6136 (2005).
J.R. Kanofsky, Chemico-Biological Interactions, 70, 1 (1989).
T. Miyazawa, K. Fujimoto, M. Kinoshita, and R. Usuki, J. Amer. Oil Chem. Soc., 71, 343 (1994).
Q. Liu, Z. Xu, J.A. Finch, R. Egerton, Chem. Mater., 10, 3936 (1998).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
El-Toni, A.M., Yin, S., Hayasaka, Y. et al. Synthesis and UV-shielding properties of silica-coated calcia-doped ceria nanoparticles via soft solution processes. J Electroceram 17, 9–14 (2006). https://doi.org/10.1007/s10832-006-9928-7
Issue Date:
DOI: https://doi.org/10.1007/s10832-006-9928-7