Abstract
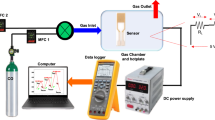
The gas sensing behavior of praseodymium doped cerium oxide (Pr x Ce1 − xO2 or PCO) has been examined for 0–1000 ppm CO or H2 in a 10% O2 atmosphere at temperatures ranging from 250–350_∘C. Total conductivity as a function of temperature suggests that oxygen diffusion kinetics are slow below approximately 350_∘C. Devices with x = 0.05 and 0.10 show stable, n-type gas sensing response, while those with x = 0.20 exhibit significant drift in sensor output, presumably due to bulk oxygen migration. The response to CO is significantly stronger than that to H2 at 300_∘C, and at 350_∘C the response to H2 is nearly zero, resulting in a CO-selective gas sensing element. Suggestions for the source of selectivity in PCO are presented.
Similar content being viewed by others
References
S.R. Morrison, Sensors and Actuators, 12, 425 (1987).
S. Semancik et al., Sensors and Actuators B-Chemical, 77, 579 (2001).
M.S. Nayak, R. Dwivedi, and S.K. Srivastava, Microelectronics Journal, 25, 17 (1994).
Y. He, Y. Li, J. Yu, and Y. Qian, Materials Letters, 40, 23 (1999).
F. Lu and S.-Y. Chen, in Preparation of Catalysts VI: Scientific Bases for the Preparation of Heterogeneous Catalysts, edited by G. Poncelet, et al. (Elsevier Science B. V., 1995), p. 489.
G. Sberveglieri, Sensors and Actuators B, 6, 239 (1992).
T.S. Stefanik and H.L. Tuller, in Materials Research Society Symposium Proceedings: Solid State Ionics—2002, edited by P. Knauth, J.-M. Tarascon, E. Traversa, H.L. Tuller, (Materials Research Society, PA Warrendale, 2002), vol. 756, p. 163.
A.E.C. Palmqvist, E.M. Johansson, S.G. Järas, and M. Muhammed, Catalysis Letters, 56, 69 (1998).
S. Sugiyama, Y. Matsumura, and J.B. Moffat, Journal of Catalysis, 139, 338 (1993).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Stefanik, T.S., Tuller, H.L. Praseodymium-Cerium Oxide as a Surface-Effect Gas Sensor. J Electroceram 13, 771–774 (2004). https://doi.org/10.1007/s10832-004-5190-z
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10832-004-5190-z