Abstract
Purpose
To identify the disease-causing genes of Chinese Han women with idiopathic premature ovarian insufficiency (POI).
Methods
Seventy-four Chinese Han women with idiopathic POI were collected to analyze the genetic etiology. Triplet repeat-primed polymerase chain reaction (TP-PCR) was performed to screen the FMR1 (CGG)n premutation, and then 60 POI-related genes were sequenced by targeted next-generation sequencing (NGS) in POI patients with normal FMR1.
Results
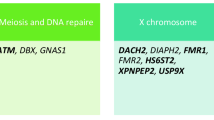
A total of one patient (1/74) with FMR1 premutation was identified. Targeted NGS revealed that 15.07% (11/73) patients had pathogenic or likely pathogenic variants of Mendelian genes (FOXL2, EIF2B2, CYP17A1, CLPP, MCM9, GDF9, MSH5, ERCC6, POLG). Ten novel variants in six Mendelian genes were identified, such as CLPP c.355A>C (p.I119L) and c.688A>C (p.M230L), MCM9 c.1157C>T (p.T386M) and c.1291A>G (p.M431V), GDF9 c. 238C>T (p.Q80X), MSH5 c.604G>C (p.G202R) and c.2063T>C (p.I688T), ERCC6 c.C1769C>T (p.P590L), POLG c.2832G>C (p.E944D), and c.2821A>G (p.I941V).
Conclusion
This study suggested targeted NGS was an efficient etiologic test for idiopathic POI patients without FMR1 premutation and enriched the variant spectrum of POI-related genes.


Similar content being viewed by others
Data availability
The data that supports the findings of this study are available in the supplementary material of this article.
Code availability
The version of each software applications was indicated in the text.
References
POI. tESoHRaEEGGo, Webber L, Davies M, Anderson R, Bartlett J, Braat D, et al. ESHRE Guideline: management of women with premature ovarian insufficiency. Hum Reprod. 2016;31(5):926–37. https://doi.org/10.1093/humrep/dew027.
De Vos M, Devroey P, Fauser BC. Primary ovarian insufficiency. Lancet. 2010;376(9744):911–21. https://doi.org/10.1016/S0140-6736(10)60355-8.
Atabiekov I, Hobeika E, Sheikh U, El Andaloussi A, Al-Hendy A. The role of gene therapy in premature ovarian insufficiency management. Biomedicines. 2018;6(4). https://doi.org/10.3390/biomedicines6040102.
Qin Y, Jiao X, Simpson JL, Chen ZJ. Genetics of primary ovarian insufficiency: new developments and opportunities. Hum Reprod Update. 2015;21(6):787–808. https://doi.org/10.1093/humupd/dmv036.
Cohen N, Fedewa S, Chen AY. Epidemiology and demographics of the head and neck cancer population. Oral Maxillofac Surg Clin North Am. 2018;30(4):381–95. https://doi.org/10.1016/j.coms.2018.06.001.
Jiao X, Ke H, Qin Y, Chen ZJ. Molecular genetics of premature ovarian insufficiency. Trends Endocrinol Metab. 2018;29(11):795–807. https://doi.org/10.1016/j.tem.2018.07.002.
Jiao X, Zhang H, Ke H, Zhang J, Cheng L, Liu Y, et al. Premature ovarian insufficiency: phenotypic characterization within different etiologies. J Clin Endocrinol Metab. 2017;102(7):2281–90. https://doi.org/10.1210/jc.2016-3960.
Barros F, Carvalho F, Barros A, Dória S. Premature ovarian insufficiency: clinical orientations for genetic testing and genetic counseling. Porto Biomed J. 2020;5(3):e62. https://doi.org/10.1097/j.pbj.0000000000000062.
Liao C, Fu F, Yang X, Sun YM, Li DZ. Analysis of Chinese women with primary ovarian insufficiency by high resolution array-comparative genomic hybridization. Chin Med J. 2011;124(11):1739–42.
Bestetti I, Castronovo C, Sironi A, Caslini C, Sala C, Rossetti R, et al. High-resolution array-CGH analysis on 46,XX patients affected by early onset primary ovarian insufficiency discloses new genes involved in ovarian function. Hum Reprod. 2019;34(3):574–83. https://doi.org/10.1093/humrep/dey389.
Lyon E, Laver T, Yu P, Jama M, Young K, Zoccoli M, et al. A simple, high-throughput assay for Fragile X expanded alleles using triple repeat primed PCR and capillary electrophoresis. J Mol Diagn. 2010;12(4):505–11. https://doi.org/10.2353/jmoldx.2010.090229.
Richards S, Aziz N, Bale S, Bick D, Das S, Gastier-Foster J, et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med. 2015;17(5):405–24. https://doi.org/10.1038/gim.2015.30.
Zheng GX, Lau BT, Schnall-Levin M, Jarosz M, Bell JM, Hindson CM, et al. Haplotyping germline and cancer genomes with high-throughput linked-read sequencing. Nat Biotechnol. 2016;34(3):303–11. https://doi.org/10.1038/nbt.3432.
Sullivan AK, Marcus M, Epstein MP, Allen EG, Anido AE, Paquin JJ, et al. Association of FMR1 repeat size with ovarian dysfunction. Hum Reprod. 2005;20(2):402–12. https://doi.org/10.1093/humrep/deh635.
Guo T, Qin Y, Jiao X, Li G, Simpson JL, Chen ZJ. FMR1 premutation is an uncommon explanation for premature ovarian failure in Han Chinese. PLoS One. 2014;9(7):e103316. https://doi.org/10.1371/journal.pone.0103316.
Hodges E, Xuan Z, Balija V, Kramer M, Molla MN, Smith SW, et al. Genome-wide in situ exon capture for selective resequencing. Nat Genet. 2007;39(12):1522–7. https://doi.org/10.1038/ng.2007.42.
Albert TJ, Molla MN, Muzny DM, Nazareth L, Wheeler D, Song X, et al. Direct selection of human genomic loci by microarray hybridization. Nat Methods. 2007;4(11):903–5. https://doi.org/10.1038/nmeth1111.
Yang T, Wei X, Chai Y, Li L, Wu H. Genetic etiology study of the non-syndromic deafness in Chinese Hans by targeted next-generation sequencing. Orphanet J Rare Dis. 2013;8:85. https://doi.org/10.1186/1750-1172-8-85.
Li Z, Huang J, Zhao J, Chen C, Wang H, Ding H, et al. Rapid molecular genetic diagnosis of hypertrophic cardiomyopathy by semiconductor sequencing. J Transl Med. 2014;12:173. https://doi.org/10.1186/1479-5876-12-173.
Fonseca DJ, Patino LC, Suarez YC, de Jesus Rodriguez A, Mateus HE, Jimenez KM, et al. Next generation sequencing in women affected by nonsyndromic premature ovarian failure displays new potential causative genes and mutations. Fertil Steril. 2015;104(1):154–62 e2. https://doi.org/10.1016/j.fertnstert.2015.04.016.
Bouilly J, Beau I, Barraud S, Bernard V, Azibi K, Fagart J, et al. Identification of multiple gene mutations accounts for a new genetic architecture of primary ovarian insufficiency. J Clin Endocrinol Metab. 2016;101(12):4541–50. https://doi.org/10.1210/jc.2016-2152.
Beysen D, De Paepe A, De Baere E. FOXL2 mutations and genomic rearrangements in BPES. Hum Mutat. 2009;30(2):158–69. https://doi.org/10.1002/humu.20807.
Nallathambi J, Neethirajan G, Usha K, Jitendra J, De Baere E, Sundaresan P. FOXL2 mutations in Indian families with blepharophimosis-ptosis-epicanthus inversus syndrome. J Genet. 2007;86(2):165–8.
Matsukawa T, Wang X, Liu R, Wortham NC, Onuki Y, Kubota A, et al. Adult-onset leukoencephalopathies with vanishing white matter with novel missense mutations in EIF2B2, EIF2B3, and EIF2B5. Neurogenetics. 2011;12(3):259–61. https://doi.org/10.1007/s10048-011-0284-7.
Marsh CA, Auchus RJ. Fertility in patients with genetic deficiencies of cytochrome P450c17 (CYP17A1): combined 17-hydroxylase/17,20-lyase deficiency and isolated 17,20-lyase deficiency. Fertil Steril. 2014;101(2):317–22. https://doi.org/10.1016/j.fertnstert.2013.11.011.
Labauge P, Horzinski L, Ayrignac X, Blanc P, Vukusic S, Rodriguez D, et al. Natural history of adult-onset eIF2B-related disorders: a multi-centric survey of 16 cases. Brain. 2009;132(Pt 8):2161–9. https://doi.org/10.1093/brain/awp171.
Carvalho LC, Brito VN, Martin RM, Zamboni AM, Gomes LG, Inacio M, et al. Clinical, hormonal, ovarian, and genetic aspects of 46,XX patients with congenital adrenal hyperplasia due to CYP17A1 defects. Fertil Steril. 2016;105(6):1612–9. https://doi.org/10.1016/j.fertnstert.2016.02.008.
Jenkinson EM, Rehman AU, Walsh T, Clayton-Smith J, Lee K, Morell RJ, et al. Perrault syndrome is caused by recessive mutations in CLPP, encoding a mitochondrial ATP-dependent chambered protease. Am J Hum Genet. 2013;92(4):605–13. https://doi.org/10.1016/j.ajhg.2013.02.013.
Marlin S, Lacombe D, Jonard L, Leboulanger N, Bonneau D, Goizet C, et al. Perrault syndrome: report of four new cases, review and exclusion of candidate genes. Am J Med Genet A. 2008;146A(5):661–4. https://doi.org/10.1002/ajmg.a.32180.
Kim MJ, Kim SJ, Kim J, Chae H, Kim M, Kim Y. Genotype and phenotype heterogeneity in perrault syndrome. J Pediatr Adolesc Gynecol. 2013;26(1):e25–7. https://doi.org/10.1016/j.jpag.2012.10.008.
Brodie EJ, Zhan H, Saiyed T, Truscott KN, Dougan DA. Perrault syndrome type 3 caused by diverse molecular defects in CLPP. Sci Rep. 2018;8(1):12862. https://doi.org/10.1038/s41598-018-30311-1.
Wood-Trageser MA, Gurbuz F, Yatsenko SA, Jeffries EP, Kotan LD, Surti U, et al. MCM9 mutations are associated with ovarian failure, short stature, and chromosomal instability. Am J Hum Genet. 2014;95(6):754–62. https://doi.org/10.1016/j.ajhg.2014.11.002.
Fauchereau F, Shalev S, Chervinsky E, Beck-Fruchter R, Legois B, Fellous M, et al. A non-sense MCM9 mutation in a familial case of primary ovarian insufficiency. Clin Genet. 2016;89(5):603–7. https://doi.org/10.1111/cge.12736.
Elvin JA, Yan C, Matzuk MM. Oocyte-expressed TGF-beta superfamily members in female fertility. Mol Cell Endocrinol. 2000;159(1-2):1–5.
Carabatsos MJ, Elvin J, Matzuk MM, Albertini DF. Characterization of oocyte and follicle development in growth differentiation factor-9-deficient mice. Dev Biol. 1998;204(2):373–84. https://doi.org/10.1006/dbio.1998.9087.
Dong J, Albertini DF, Nishimori K, Kumar TR, Lu N, Matzuk MM. Growth differentiation factor-9 is required during early ovarian folliculogenesis. Nature. 1996;383(6600):531–5. https://doi.org/10.1038/383531a0.
Dixit H, Rao LK, Padmalatha V, Kanakavalli M, Deenadayal M, Gupta N, et al. Mutational screening of the coding region of growth differentiation factor 9 gene in Indian women with ovarian failure. Menopause. 2005;12(6):749–54. https://doi.org/10.1097/01.gme.0000184424.96437.7a.
Laissue P, Christin-Maitre S, Touraine P, Kuttenn F, Ritvos O, Aittomaki K, et al. Mutations and sequence variants in GDF9 and BMP15 in patients with premature ovarian failure. Eur J Endocrinol. 2006;154(5):739–44. https://doi.org/10.1530/eje.1.02135.
Kovanci E, Rohozinski J, Simpson JL, Heard MJ, Bishop CE, Carson SA. Growth differentiating factor-9 mutations may be associated with premature ovarian failure. Fertil Steril. 2007;87(1):143–6. https://doi.org/10.1016/j.fertnstert.2006.05.079.
Mandon-Pepin B, Touraine P, Kuttenn F, Derbois C, Rouxel A, Matsuda F, et al. Genetic investigation of four meiotic genes in women with premature ovarian failure. Eur J Endocrinol. 2008;158(1):107–15. https://doi.org/10.1530/EJE-07-0400.
Guo T, Zhao S, Zhao S, Chen M, Li G, Jiao X, et al. Mutations in MSH5 in primary ovarian insufficiency. Hum Mol Genet. 2017;26(8):1452–7. https://doi.org/10.1093/hmg/ddx044.
Troelstra C, van Gool A, de Wit J, Vermeulen W, Bootsma D, Hoeijmakers JH. ERCC6, a member of a subfamily of putative helicases, is involved in Cockayne’s syndrome and preferential repair of active genes. Cell. 1992;71(6):939–53.
Qin Y, Guo T, Li G, Tang TS, Zhao S, Jiao X, et al. CSB-PGBD3 Mutations cause premature ovarian failure. PLoS Genet. 2015;11(7):e1005419. https://doi.org/10.1371/journal.pgen.1005419.
Lamantea E, Tiranti V, Bordoni A, Toscano A, Bono F, Servidei S, et al. Mutations of mitochondrial DNA polymerase gammaA are a frequent cause of autosomal dominant or recessive progressive external ophthalmoplegia. Ann Neurol. 2002;52(2):211–9. https://doi.org/10.1002/ana.10278.
Cohen BH, Naviaux RK. The clinical diagnosis of POLG disease and other mitochondrial DNA depletion disorders. Methods. 2010;51(4):364–73. https://doi.org/10.1016/j.ymeth.2010.05.008.
Steuerwald N, Barritt JA, Adler R, Malter H, Schimmel T, Cohen J, et al. Quantification of mtDNA in single oocytes, polar bodies and subcellular components by real-time rapid cycle fluorescence monitored PCR. Zygote. 2000;8(3):209–15.
Santos TA, El Shourbagy S, St John JC. Mitochondrial content reflects oocyte variability and fertilization outcome. Fertil Steril. 2006;85(3):584–91. https://doi.org/10.1016/j.fertnstert.2005.09.017.
Pagnamenta AT, Taanman JW, Wilson CJ, Anderson NE, Marotta R, Duncan AJ, et al. Dominant inheritance of premature ovarian failure associated with mutant mitochondrial DNA polymerase gamma. Hum Reprod. 2006;21(10):2467–73. https://doi.org/10.1093/humrep/del076.
Luoma P, Melberg A, Rinne JO, Kaukonen JA, Nupponen NN, Chalmers RM, et al. Parkinsonism, premature menopause, and mitochondrial DNA polymerase gamma mutations: clinical and molecular genetic study. Lancet. 2004;364(9437):875–82. https://doi.org/10.1016/S0140-6736(04)16983-3.
Ledig S, Ropke A, Wieacker P. Copy number variants in premature ovarian failure and ovarian dysgenesis. Sex Dev. 2010;4(4-5):225–32. https://doi.org/10.1159/000314958.
Norling A, Hirschberg AL, Rodriguez-Wallberg KA, Iwarsson E, Wedell A, Barbaro M. Identification of a duplication within the GDF9 gene and novel candidate genes for primary ovarian insufficiency (POI) by a customized high-resolution array comparative genomic hybridization platform. Hum Reprod. 2014;29(8):1818–27. https://doi.org/10.1093/humrep/deu149.
Wang B, Li L, Ni F, Song J, Wang J, Mu Y, et al. Mutational analysis of SAL-Like 4 (SALL4) in Han Chinese women with premature ovarian failure. Mol Hum Reprod. 2009;15(9):557–62. https://doi.org/10.1093/molehr/gap046.
Nishimura-Tadaki A, Wada T, Bano G, Gough K, Warner J, Kosho T, et al. Breakpoint determination of X;autosome balanced translocations in four patients with premature ovarian failure. J Hum Genet. 2011;56(2):156–60. https://doi.org/10.1038/jhg.2010.155.
Qin C, Yuan Z, Yao J, Zhu W, Wu W, Xie J. AMH and AMHR2 genetic variants in Chinese women with primary ovarian insufficiency and normal age at natural menopause. Reprod BioMed Online. 2014;29(3):311–8. https://doi.org/10.1016/j.rbmo.2014.05.003.
Fouquet B, Pawlikowska P, Caburet S, Guigon C, Makinen M, Tanner L, et al. A homozygous FANCM mutation underlies a familial case of non-syndromic primary ovarian insufficiency. Elife. 2017;6. https://doi.org/10.7554/eLife.30490.
Demain LA, Urquhart JE, O’Sullivan J, Williams SG, Bhaskar SS, Jenkinson EM, et al. Expanding the genotypic spectrum of Perrault syndrome. Clin Genet. 2017;91(2):302–12. https://doi.org/10.1111/cge.12776.
Weinberg-Shukron A, Renbaum P, Kalifa R, Zeligson S, Ben-Neriah Z, Dreifuss A, et al. A mutation in the nucleoporin-107 gene causes XX gonadal dysgenesis. J Clin Invest. 2015;125(11):4295–304. https://doi.org/10.1172/JCI83553.
Chen A, Tiosano D, Guran T, Baris HN, Bayram Y, Mory A, et al. Mutations in the mitochondrial ribosomal protein MRPS22 lead to primary ovarian insufficiency. Hum Mol Genet. 2018;27(11):1913–26. https://doi.org/10.1093/hmg/ddy098.
Zhang YX, Li HY, He WB, Tu C, Du J, Li W, et al. XRCC2 mutation causes premature ovarian insufficiency as well as non-obstructive azoospermia in humans. Clin Genet. 2018;95:442–3. https://doi.org/10.1111/cge.13475.
Zhang D, Liu Y, Zhang Z, Lv P, Liu Y, Li J, et al. Basonuclin 1 deficiency is a cause of primary ovarian insufficiency. Hum Mol Genet. 2018;27(21):3787–800. https://doi.org/10.1093/hmg/ddy261.
Qin Y, Zhang F, Chen ZJ. BRCA2 in Ovarian Development and Function. N Engl J Med. 2019;380(11):1086–7. https://doi.org/10.1056/NEJMc1813800.
Funding
This study was supported by the National Key Research and Development Program of China (2016YFC1000200, 2017YFC1001602) and the National Natural Science Foundation of China (81471429, 81730041) and Natural Science Foundation of Jiangsu Province (BK20201488).
Author information
Authors and Affiliations
Contributions
JS, JL, and FD conceived and designed the study. JS, YG, and XM collected samples. DY, FS, JX, and XS performed the experiments. JS, DW, and YC analyzed the data. JS wrote the manuscript. JL and FD revised the manuscript. All authors read and approved the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shen, J., Qu, D., Gao, Y. et al. Genetic etiologic analysis in 74 Chinese Han women with idiopathic premature ovarian insufficiency by combined molecular genetic testing. J Assist Reprod Genet 38, 965–978 (2021). https://doi.org/10.1007/s10815-021-02083-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-021-02083-7