Abstract
Purpose
To determine if the use of ICSI in women of advanced maternal age with non-male factor infertility increases chances of live birth.
Methods
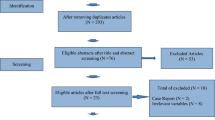
Retrospective data analysis of 10 years of cycle data from a single Australian IVF clinic (Repromed). First cycle patients only of an advanced maternal age (≥ 35 years) with non-male factor infertility utilising standard IVF or ICSI insemination and having at least three oocytes collected at egg pick up were assessed for live birth following transfer of single genetically unscreened blastocyst (N = 577). Subanalysis of clinical pregnancy, miscarriage, fertilisation, embryo utilisation rate and having a blastocyst for transfer were considered. Unadjusted, covariate adjusted and propensity score weighted analysis were performed.
Results
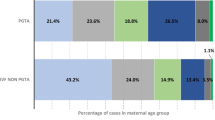
The use of standard IVF insemination in women ≥ 35 years with non-male factor infertility increased the chance of a live birth compared with ICSI insemination (unadjusted OR = 2.72, 95% CI [1.78, 4.17]; adjusted OR = 2.64, 95% CI [1.64, 4.27] and weighted OR = 2.26, 95% CI [1.72, 2.98] 31% vs 14%). All other outcomes (fertilisation rate, embryo utilisation, blastocyst for embryo transfer and miscarriage rate) were unaffected.
Conclusion
In couples with advanced maternal age and non-male factor infertility, standard IVF insemination appears to increase the chance of a live birth compared with ICSI. As such, the results of this study support the use of routine IVF as the preferred insemination technique for older women in non-male factor infertility. However, future randomised controlled trials are still required to assess this policy.


Similar content being viewed by others
Change history
27 February 2021
A Correction to this paper has been published: https://doi.org/10.1007/s10815-021-02076-6
References
Newman J, Fitzgerald O, Paul R, Chambers G. Assisted reproductive technology in Australia and New Zealand 2017. Sydney: National Perinatal epidemiology and Statistics Unit, the University of New South Wales Sydney; 2019.
Rosenwaks Z, Pereira N. The pioneering of intracytoplasmic sperm injection: historical perspectives. Reproduction. 2017;154(6):F71–F7. https://doi.org/10.1530/REP-17-0308.
Taskforce MBSR. Gynaecology Clinical Committee of the Medicare Benefits Schedule Review Taskforce (2018). Australian Government; 2018.
Katz D, Teloken P, Shoshany O. Male infertility – the other side of the equation. Aust Fam Physician. 2017;46:641–6.
Dyer S, Chambers GM, de Mouzon J, Nygren KG, Zegers-Hochschild F, Mansour R, et al. International Committee for Monitoring Assisted Reproductive Technologies world report: assisted reproductive technology 2008, 2009 and 2010. Hum Reprod. 2016;31(7):1588–609. https://doi.org/10.1093/humrep/dew082.
Boulet SL, Mehta A, Kissin DM, Warner L, Kawwass JF, Jamieson DJ. Trends in use of and reproductive outcomes associated with intracytoplasmic sperm injection. Jama. 2015;313(3):255–63. https://doi.org/10.1001/jama.2014.17985.
Abbas AM, Hussein RS, Elsenity MA, Samaha II, El Etriby KA, Abd El-Ghany MF, et al. Higher clinical pregnancy rate with in-vitro fertilization versus intracytoplasmic sperm injection in treatment of non-male factor infertility: systematic review and meta-analysis. J Gynecol Obstet Hum Reprod. 2020;49:101706. https://doi.org/10.1016/j.jogoh.2020.101706.
Li Z, Wang AY, Bowman M, Hammarberg K, Farquhar C, Johnson L, et al. ICSI does not increase the cumulative live birth rate in non-male factor infertility. Hum Reprod. 2018;33(7):1322–30. https://doi.org/10.1093/humrep/dey118.
Khamsi F, Yavas Y, Roberge S, Wong JC, Lacanna IC, Endman M. Intracytoplasmic sperm injection increased fertilization and good-quality embryo formation in patients with non-male factor indications for in vitro fertilization: a prospective randomized study. Fertil Steril. 2001;75(2):342–7. https://doi.org/10.1016/s0015-0282(00)01674-5.
Jenkins JM, Davies DW, Devonport H, Anthony FW, Gadd SC, Watson RH, et al. Comparison of ‘poor’ responders with ‘good’ responders using a standard buserelin/human menopausal gonadotrophin regime for in-vitro fertilization. Hum Reprod. 1991;6(7):918–21. https://doi.org/10.1093/oxfordjournals.humrep.a137459.
Tannus S, Son WY, Gilman A, Younes G, Shavit T, Dahan MH. The role of intracytoplasmic sperm injection in non-male factor infertility in advanced maternal age. Hum Reprod. 2017;32(1):119–24. https://doi.org/10.1093/humrep/dew298.
Gennarelli G, Carosso A, Canosa S, Filippini C, Cesarano S, Scarafia C, et al. ICSI versus conventional IVF in women aged 40 years or more and unexplained infertility: a retrospective evaluation of 685 cycles with propensity score model. J Clin Med. 2019;8(10). https://doi.org/10.3390/jcm8101694.
Practice Committees of the American Society for Reproductive M, the Society for Assisted Reproductive Technology. Electronic address Aao, Practice Committees of the American Society for Reproductive M, the Society for Assisted Reproductive T. The use of preimplantation genetic testing for aneuploidy (PGT-A): a committee opinion. Fertil Steril. 2018;109(3):429–36. https://doi.org/10.1016/j.fertnstert.2018.01.002.
WHO. WHO laboratory manual for the examination and processing of human semen. 5th ed. Cambridge: Cambridge University Press; 2010.
WHO. WHO laboratory manual for the examination of human semen and sperm-cervical mucus interaction. 4th ed. Cambridge: Cambridge University Press; 1999.
Thalluri V, Tremellen KP. Ultrasound diagnosed adenomyosis has a negative impact on successful implantation following GnRH antagonist IVF treatment. Hum Reprod. 2012;27(12):3487–92. https://doi.org/10.1093/humrep/des305.
Tremellen KP, Lane M. Avoidance of weekend oocyte retrievals during GnRH antagonist treatment by simple advancement or delay of hCG administration does not adversely affect IVF live birth outcomes. Hum Reprod. 2010;25(5):1219–24. https://doi.org/10.1093/humrep/deq059.
Trounson AO, Gardner DK. Handbook of in vitro fertilization. 4th Edition: Taylor and Francis Group. Boca Raton: CRC Press; 2017.
Feil D, Henshaw RC, Lane M. Day 4 embryo selection is equal to day 5 using a new embryo scoring system validated in single embryo transfers. Hum Reprod. 2008;23(7):1505–10. https://doi.org/10.1093/humrep/dem419.
Gardner DK, Schoolcraft WB. Culture and transfer of human blastocysts. Curr Opin Obstet Gynecol. 1999;11(3):307–11. https://doi.org/10.1097/00001703-199906000-00013.
Williamson E, Morley R, Lucas A, Carpenter J. Propensity scores: from naive enthusiasm to intuitive understanding. Stat Methods Med Res. 2012;21(3):273–93. https://doi.org/10.1177/0962280210394483.
Saito H, Saito T, Kaneko T, Sasagawa I, Kuramoto T, Hiroi M. Relatively poor oocyte quality is an indication for intracytoplasmic sperm injection. Fertil Steril. 2000;73(3):465–9. https://doi.org/10.1016/s0015-0282(99)00547-6.
Farhi J, Cohen K, Mizrachi Y, Weissman A, Raziel A, Orvieto R. Should ICSI be implemented during IVF to all advanced-age patients with non-male factor subfertility? Reprod Biol Endocrinol. 2019;17(1):30. https://doi.org/10.1186/s12958-019-0474-y.
Luna M, Bigelow C, Duke M, Ruman J, Sandler B, Grunfeld L, et al. Should ICSI be recommended routinely in patients with four or fewer oocytes retrieved? J Assist Reprod Genet. 2011;28(10):911–5. https://doi.org/10.1007/s10815-011-9614-9.
Rydze RT, Bosler J, Schoyer KD. Advanced maternal age and in vitro fertilization: to inject or to inseminate? That is the question. Fertil Steril. 2020;113(2):325. https://doi.org/10.1016/j.fertnstert.2019.11.004.
Tremellen K. Oxidative stress and male infertility--a clinical perspective. Hum Reprod Update. 2008;14(3):243–58. https://doi.org/10.1093/humupd/dmn004.
Zorn B, Vidmar G, Meden-Vrtovec H. Seminal reactive oxygen species as predictors of fertilization, embryo quality and pregnancy rates after conventional in vitro fertilization and intracytoplasmic sperm injection. Int J Androl. 2003;26(5):279–85.
Aitken RJ. Reactive oxygen species as mediators of sperm capacitation and pathological damage. Mol Reprod Dev. 2017;84(10):1039–52. https://doi.org/10.1002/mrd.22871.
Sakkas D, Ramalingam M, Garrido N, Barratt CL. Sperm selection in natural conception: what can we learn from Mother Nature to improve assisted reproduction outcomes? Hum Reprod Update. 2015;21(6):711–26. https://doi.org/10.1093/humupd/dmv042.
Agarwal A, Sharma RK, Nallella KP, Thomas AJ Jr, Alvarez JG, Sikka SC. Reactive oxygen species as an independent marker of male factor infertility. Fertil Steril. 2006;86(4):878–85. https://doi.org/10.1016/j.fertnstert.2006.02.111.
Lord T, Aitken RJ. Fertilization stimulates 8-hydroxy-2′-deoxyguanosine repair and antioxidant activity to prevent mutagenesis in the embryo. Dev Biol. 2015;406(1):1–13. https://doi.org/10.1016/j.ydbio.2015.07.024.
Gawecka JE, Marh J, Ortega M, Yamauchi Y, Ward MA, Ward WS. Mouse zygotes respond to severe sperm DNA damage by delaying paternal DNA replication and embryonic development. PLoS One. 2013;8(2):e56385. https://doi.org/10.1371/journal.pone.0056385.
Devjak R, Burnik Papler T, Verdenik I, Fon Tacer K, Vrtacnik Bokal E. Embryo quality predictive models based on cumulus cells gene expression. Balkan J Med Genet. 2016;19(1):5–12. https://doi.org/10.1515/bjmg-2016-0001.
Gebhardt KM, Feil DK, Dunning KR, Lane M, Russell DL. Human cumulus cell gene expression as a biomarker of pregnancy outcome after single embryo transfer. Fertil Steril. 2011;96(1):47–52.e2. https://doi.org/10.1016/j.fertnstert.2011.04.033.
Carvalho M, Leal F, Mota S, Aguiar A, Sousa S, Nunes J, et al. The effect of denudation and injection timing in the reproductive outcomes of ICSI cycles: new insights into the risk of in vitro oocyte ageing. Hum Reprod. 2020;35(10):2226–36. https://doi.org/10.1093/humrep/deaa211.
McReynolds S, Dzieciatkowska M, McCallie BR, Mitchell SD, Stevens J, Hansen K, et al. Impact of maternal aging on the molecular signature of human cumulus cells. Fertil Steril. 2012;98(6):1574–80.e5. https://doi.org/10.1016/j.fertnstert.2012.08.012.
Patassini C, Garolla A, Bottacin A, Menegazzo M, Speltra E, Foresta C, et al. Molecular karyotyping of human single sperm by array-comparative genomic hybridization. PLoS One. 2013;8(4):e60922. https://doi.org/10.1371/journal.pone.0060922.
Garolla A, Sartini B, Cosci I, Pizzol D, Ghezzi M, Bertoldo A, et al. Molecular karyotyping of single sperm with nuclear vacuoles identifies more chromosomal abnormalities in patients with testiculopathy than fertile controls: implications for ICSI. Hum Reprod. 2015;30(11):2493–500. https://doi.org/10.1093/humrep/dev202.
De Munck N, El Khatib I, Abdala A, El-Damen A, Bayram A, Arnanz A, et al. Intracytoplasmic sperm injection is not superior to conventional IVF in couples with non-male factor infertility and preimplantation genetic testing for aneuploidies (PGT-A). Hum Reprod. 2020;35(2):317–27. https://doi.org/10.1093/humrep/deaa002.
Funding
NOM is the recipient of the NHMRC Peter Doherty Early Career Fellowship.
Author information
Authors and Affiliations
Contributions
Nicole McPherson: conceptualisation, data curation, writing—original draft, approval of final manuscript. Andrew Vincent: formal analysis, writing—review and editing, approval of final manuscript. Leanne Pacella-Ince: conceptualisation and writing—review and editing, approval of final manuscript. Kelton Tremellen: conceptualisation and writing—review and editing, approval of final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
NOM, LPI and KT declare that they are paid employees of Monash IVF Group Lmt.
Ethics approval
Institutional Scientific Advisory Board (SAC) (Project number 1M2020) approved this retrospective study. Patients had previously given written consent to allow their records to be accessed for these types of low-risk retrospective audit type studies.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 18 kb).
Rights and permissions
About this article
Cite this article
McPherson, N.O., Vincent, A.D., Pacella-Ince, L. et al. Comparison of in vitro fertilisation/intracytoplasmic sperm injection on live birth rates in couples with non-male factor infertility and advanced maternal age. J Assist Reprod Genet 38, 669–678 (2021). https://doi.org/10.1007/s10815-020-02026-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-020-02026-8