Abstract
Purpose
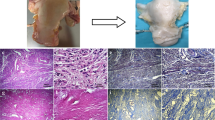
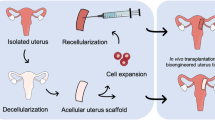
The main purpose of this investigation was to determine an efficient whole-organ decellularization protocol of a human-sized uterus and evaluate the in vivo properties of the bioscaffold.
Methods
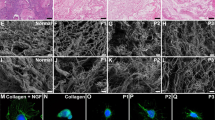
Twenty-four ovine uteri were included in this investigation and were decellularized by three different protocols (n 6). We performed histopathological and immunohistochemical evaluations, 4,6-diamidino-2-phenylindole (DAPI) staining, DNA quantification, MTT assay, scanning electron microscopy, biomechanical studies, and CT angiography to characterize the scaffolds. The optimized protocol was determined, and patches were grafted into the uterine horns of eight female Wistar rats. The grafts were extracted after 10 days; the opposite horns were harvested to be evaluated as controls.
Results
Protocol III (perfusion with 0.25% and 0.5% SDS solution and preservation in 10% formalin) was determined as the optimized method with efficient removal of the cellular components while preserving the extracellular matrix. Also, the bioscaffolds demonstrated native-like biomechanical, structural, and vascular properties. Histological and immunohistochemical evaluations of the harvested grafts confirmed the biocompatibility and recellularization potential of bioscaffolds. Also, the grafts demonstrated higher positive reaction for CD31 and Ki67 markers compared with the control samples which indicated eminent angiogenesis properties and proliferative capacity of the implanted tissues.
Conclusions
This investigation introduces an optimized protocol for whole-organ decellularization of the human-sized uterus with native-like characteristics and a prominent potential for regeneration and angiogenesis which could be employed in in vitro and in vivo studies. To the best of our knowledge, this is the first study to report biomechanical properties and angiographic evaluations of a large animal uterine scaffold.






Similar content being viewed by others
Change history
04 June 2019
The original version of this article unfortunately contained a mistake.
14 September 2019
The original version of this article unfortunately contained a mistake. The affiliation of Fariba Ghorbani should be Tracheal Diseases Research Center (TDRC), National Research Institute of Tuberculosis and Lung Diseases (NRITLD), Shahid Beheshti University of Medical Sciences, Tehran, Iran.
References
Milliez J. Uterine transplantation FIGO Committee for the ethical aspects of human reproduction and women’s health. Int J Gynaecol Obstet. 2009;106:270.
Kisu I, Mihara M, Banno K, Umene K, Araki J, Hara H, et al. Risks for donors in uterus transplantation. Reprod Sci. 2013 Dec;20(12):1406–15. https://doi.org/10.1177/1933719113493517.
Kisu I, Banno K, Mihara M, Suganuma N, Aoki D. Current status of uterus transplantation in primates and issues for clinical application. Fertil Steril. 2013;100(1):280–94.
Brännström M, Johannesson L, Bokström H, Kvarnström N, Mölne J, Dahm-Kähler P, et al. Live birth after uterus transplantation. Lancet. 2015;385:607–16.
Mats Brännström. Uterus transplantation and beyond. J Mater Sci Mater Med (2017) 28:70DOI https://doi.org/10.1007/s10856-017-5872-0, 70.
Hellström M, Bandstein S, Brännström M. Uterine tissue engineering and the future of uterus transplantation. Ann Biomed Eng. 2017 Jul;45(7):1718–30 Epub 2016 Dec 19.
Park DW, Choi DS, Ryu HS, Kwon HC, Joo H, Min CK. A well-defined in vitro three-dimensional culture of human endometrium and its applicability to endometrial cancer invasion. Cancer Lett. 2003;195:185–92.
Heidari Kani M, Chan EC, Young RC, Butler T, Smith R, Paul JW. 3D cell culturing and possibilities for myometrial tissue engineering. Ann Biomed Eng. 2017;45(7):1746–57.
Santoso EG, Yoshida K, Hirota Y, Aizawa M, Yoshino O, Kishida A, et al. Application of detergents or high hydrostatic pressure as decellularization processes in uterine tissues and their subsequent effects on in vivo uterine regeneration in murine models. PLoS One. 2014;9(7):e103201.
Hiraoka T, Hirota Y, Saito-Fujita T, Matsuo M, Egashira M, Matsumoto L, et al. STAT3 accelerates uterine epithelial regeneration in a mouse model of decellularized uterine matrix transplantation. JCI insight. 2016;1(8).
Hellström M, El-Akouri RR, Sihlbom C, Olsson BM, Lengqvist J, Backdahl H, et al. Towards the development of a bioengineered uterus: comparison of different protocols for rat uterus decellularization. Acta Biomater. 2014;10(12):5034–42.
Miyazaki K, Maruyama T. Partial regeneration and reconstruction of the rat uterus through recellularization of a decellularized uterine matrix. Biomaterials. 2014;35(31):8791–800.
Hellström M, Moreno-Moya JM, Bandstein S, Bom E, Akouri RR, Miyazaki K, et al. Bioengineered uterine tissue supports pregnancy in a rat model. Fertil Steril. 2016;106(2):487–96 e1.
Campo H, Baptista PM, Lopez-Perez N, Faus A, Cervello I, Simon C. De- and recellularization of the pig uterus: a bioengineering pilot study. Biol Reprod. 2017;96(1):34–45.
Barakat O, Abbasi S, Rodriguez G, Rios J, Wood RP, Ozaki C, et al. Use of decellularized porcine liver for engineering humanized liver organ. J Surg Res. 2012;173(1):e11–25.
Hashemi J, Pasalar P, Soleimani M, Khorramirouz R, Fendereski K, Enderami SE, et al. Application of a novel bioreactor for in vivo engineering of pancreas tissue. J Cell Physiol. 2018;233(5):3805–16.
Kajbafzadeh AM, Khorramirouz R, Kameli SM, Fendereski K, Daryabari SS, Tavangar SM, et al. Three-year efficacy and patency follow-up of decellularized human internal mammary artery as a novel vascular graft in animal models. J Thorac Cardiovasc Surg. 2019;157(4):1494–502.
Khorramirouz R, Kameli SM, Fendereski K, Daryabari SS, Kajbafzadeh AM. Evaluating the efficacy of tissue-engineered human amniotic membrane in the treatment of myocardial infarction. Regen Med. 2019;14(2):113–26.
Kitahara H, Yagi H, Tajima K, Okamoto K, Yoshitake A, Aeba R, et al. Heterotopic transplantation of a decellularized and recellularized whole porcine heart. Interact Cardiovasc Thorac Surg. 2016;22(5):571–9.
Young RC, Goloman G. Allo- and xeno-reassembly of human and rat myometrium from cells and scaffolds. Tissue Eng A. 2013;19(19–20):2112–9.
Ott HC, Matthiesen TS, Goh SK, Black LD, Kren SM, Netoff TI, et al. Perfusion-decellularized matrix: using nature’s platform to engineer a bioartificial heart. Nat Med. 2008;14(2):213–21.
Myers KM, Elad D. Biomechanics of the human uterus. Wiley Interdiscip Rev Syst Biol Med. 2017;9(5).
Bhrany AD, Lien CJ, Beckstead BL, Futran ND, Muni NH, Giachelli CM, et al. Crosslinking of an oesophagus acellular matrix tissue scaffold. J Tissue Eng Regen Med. 2008;2(6):365–72.
Baptista PM, Siddiqui MM, Lozier G, Rodriguez SR, Atala A, Soker S. The use of whole organ decellularization for the generation of a vascularized liver organoid. Hepatology (Baltimore, Md). 2011;53(2):604–17.
Williams C, Liao J, Joyce EM, Wang B, Leach JB, Sacks MS, et al. Altered structural and mechanical properties in decellularized rabbit carotid arteries. Acta Biomater. 2009;5(4):993–1005.
Arenas-Herrera JE, Ko IK, Atala A, Yoo JJ. Decellularization for whole organ bioengineering. Biomed Mater (Bristol, England). 2013;8(1):014106.
Cebotari S, Tudorache I, Jaekel T, Hilfiker A, Dorfman S, Ternes W, et al. Detergent decellularization of heart valves for tissue engineering: toxicological effects of residual detergents on human endothelial cells. Artif Organs. 2010;34(3):206–10.
Acknowledgments
We would like to express our sincere gratitude to Dr. Torabi for providing the organs and Mr. Reza Esmaili and Mr. Nourbakhsh for their kind cooperation during this project.
Funding
This study was funded by Tehran University of Medical Sciences (grant number 96-03-30-36497).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All the animal procedures were approved by The Animal Ethics Committee of the Tehran University of Medical Sciences, School of Medicine and Education Section of Basic Sciences and were performed in accordance with the Animal Welfare Act and the Guide for the Care and Use of Laboratory Animals.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Daryabari, S.S., Kajbafzadeh, AM., Fendereski, K. et al. Development of an efficient perfusion-based protocol for whole-organ decellularization of the ovine uterus as a human-sized model and in vivo application of the bioscaffolds. J Assist Reprod Genet 36, 1211–1223 (2019). https://doi.org/10.1007/s10815-019-01463-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-019-01463-4