Abstract
Purpose
The purpose of this study was to identify mutations that cause non-syndromic male infertility using whole exome sequencing of family cases.
Methods
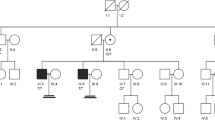
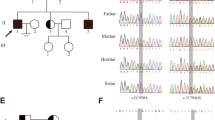
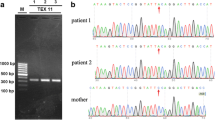
We recruited a consanguineous Turkish family comprising nine siblings with male triplets; two of the triplets were infertile as well as one younger infertile brother. Whole exome sequencing (WES) performed on two azoospermic brothers identified a mutation in the melanoma antigen family B4 (MAGEB4) gene which was confirmed via Sanger sequencing and then screened for on control groups and unrelated infertile subjects. The effect of the mutation on messenger RNA (mRNA) and protein levels was tested after in vitro cell transfection. Structural features of MAGEB4 were predicted throughout the conserved MAGE domain.
Results
The novel single-base substitution (c.1041A>T) in the X-linked MAGEB4 gene was identified as a no-stop mutation. The mutation is predicted to add 24 amino acids to the C-terminus of MAGEB4. Our functional studies were unable to detect any effect either on mRNA stability, intracellular localization of the protein, or the ability to homodimerize/heterodimerize with other MAGE proteins. We thus hypothesize that these additional amino acids may affect the proper protein interactions with MAGEB4 partners.
Conclusion
The whole exome analysis of a consanguineous Turkish family revealed MAGEB4 as a possible new X-linked cause of inherited male infertility. This study provides the first clue to the physiological function of a MAGE protein.





Similar content being viewed by others
References
Krausz C, Giachini C, Lo Giacco D, Daguin F, Chianese C, Ars E, Ruiz-Castane E, Forti G, Rossi E. High resolution X chromosome-specific array-CGH detects new CNVs in infertile males. PLoS One. 2012;7:e44887.
Jarow JP, Espeland MA, Lipshultz LI. Evaluation of the azoospermic patient. J Urol. 1989;142:62–5.
Baker HW. Clinical management of male infertility. In: Jameson JL, De Groot LI, editors. Endocronology. 6th ed. Philadelphia, PA: Elsevier Press; 2010. p. 2556–79.
Van Assche E, Bonduelle M, Tournaye H, Joris H, Verheyen G, Devroey P, Van Steirteghem A, Liebaers I. Cytogenetics of infertile men. Hum Reprod. 1996;11:1–24.
Rives N, Joly G, Machy A, Simeon N, Leclerc P, Mace B. Assessment of sex chromosome aneuploidy in sperm nuclei from 47,XXY and 46,XY/47,XXY males: comparison with fertile and infertile males with normal karyotype. Mol Hum Reprod. 2000;6:107–12.
Walsh TJ, Pera RR, Turek PJ. The genetics of male infertility. Semin Reprod Med. 2009;27:124–36.
Yatsenko SA, Rajkovic A. Chromosomal causes of infertility: the story continues. In: Sermon K, Viville S, editors. Textbook of Human Reproductive Genetics. Cambridge: Cambridge University Press; 2014. p. 98–100.
Krausz C, Hoefsloot L, Simoni M, Tüttelmann F. EAA/EMQN best practice guidelines for molecular diagnosis of Y-chromosomal microdeletions. State of the Art 2013. Andrology. 2014;2:5–19.
Hotaling J, Carrell DT. Clinical genetic testing for male factor infertility: current applications and future directions. Andrology. 2012;2:339–50.
Matzuk MM, Lamb DJ. The biology of infertility: research advances and clinical challenges. Nat Med. 2008;14:1197–213.
Mou L, Xie N, Yang L, Liu Y, Diao R, Cai Z, Li H, Gui Y. A novel mutation of DAX-1 associated with secretory azoospermia. PLoS One. 2015;10:e0133997.
Ramasamy R, Bakircioglu ME, Cengiz C, Karaca E, Scovell J, Jhangiani SN, Akdemir ZC, Bainbridge M, Yu Y, Huff C, Gibbs RA, Lupski JR, Lamb DJ. Whole-exome sequencing identifies novel homozygous mutation in NPAS2 in family with nonobstructive azoospermia. Fertil Steril. 2015;104:286–91.
Bashamboo A, Ferraz-de-Souza B, Lourenco D, Lin L, Sebire NJ, Montjean D, Bignon-Topalovic J, Mandelbaum J, Siffroi JP, Christin-Maitre S, Radhakrishna U, Rouba H, Ravel C, Seeler J, Achermann JC, McElreavey K. Human male infertility associated with mutations in NR5A1 encoding steroidogenic factor 1. Am J Hum Genet. 2010;87:505–12.
Choi Y, Jeon S, Choi M, Lee MH, Park M, Lee DR, Jun KY, Kwon Y, Lee OH, Song SH, Kim JY, Lee KA, Yoon TK, Rajkovic A, Shim SH. Mutations in SOHLH1 gene associate with nonobstructive azoospermia. Hum Mutat. 2010;31:788–93.
Maor-Sagie E, Cinnamon Y, Yaacov B, Shaag A, Goldsmidt H, Zenvirt S, Laufer N, Richler C, Frumkin A. Deleterious mutation in SYCE1 is associated with non-obstructive azoospermia. J Assist Reprod Genet. 2015;32:887–91.
Ayhan O, Balkan M, Guven A, Hazan R, Atar M, Tok A, Tolun A. Truncating mutations in TAF4B and ZMYND15 causing recessive azoospermia. J Med Genet. 2014;51:239–44.
Yang F, Silber S, Leu NA, Oates RD, Marszalek JD, Skaletsky H, Brown LG, Rozen S, Page DC, Wang PJ. TEX11 is mutated in infertile men with azoospermia and regulates genome-wide recombination rates in mouse. EMBO Mol Med. 2015;7:1198–210.
Yatsenko AN, Georgiadis AP, Ropke A, Berman AJ, Jaffe T, Olszewska M, Westernstroer B, Sanfilippo J, Kurpisz M, Rajkovic A, Yatsenko SA, Kliesch S, Schlatt S, Tuttelmann F. X-linked TEX11 mutations, meiotic arrest, and azoospermia in infertile men. N Engl J Med. 2015;372:2097–107.
Okutman O, Muller J, Baert Y, Serdarogullari M, Gultomruk M, Piton A, Rombaut C, Benkhalifa M, Teletin M, Skory V, Bakircioglu E, Goossens E, Bahceci M, Viville S. Exome sequencing reveals a nonsense mutation in TEX15 causing spermatogenic failure in a Turkish family. Hum Mol Genet. 2015;24:5581–8.
Wang XN, Li ZS, Ren Y, Jiang T, Wang YQ, Chen M, Zhang J, Hao JX, Wang YB, Sha RN, Huang Y, Liu X, Hu JC, Sun GQ, Li HG, Xiong CL, Xie J, Jiang ZM, Cai ZM, Wang J, Huff V, Gui YT, Gao F. The Wilms tumor gene, Wt1, is critical for mouse spermatogenesis via regulation of sertoli cell polarity and is associated with non-obstructive azoospermia in humans. PLoS Genet. 2013;9:e1003645.
Wang PJ, McCarrey JR, Yang F, Page DC. An abundance of X-linked genes expressed in spermatogonia. Nat Genet. 2001;27:422–6.
Greenbaum MP, Yan W, Wu MH, Lin YN, Agno JE, Sharma M, Braun RE, Rajkovic A, Matzuk MM. TEX14 is essential for intercellular bridges and fertility in male mice. Proc Natl Acad Sci U S A. 2006;103:4982–7.
Tarabay Y, Kieffer E, Teletin M, Celebi C, Van Montfoort A, Zamudio N, Achour M, El Ramy R, Gazdag E, Tropel P, Mark M, Bourc’his D, Viville S. The mammalian-specific Tex19.1 gene plays an essential role in spermatogenesis and placenta-supported development. Hum Reprod. 2013;28:2201–14.
Yang F, Eckardt S, Leu NA, McLaughlin KJ, Wang PJ. Mouse TEX15 is essential for DNA double-strand break repair and chromosomal synapsis during male meiosis. J Cell Biol. 2008a;180:673–9.
Akinloye O, Gromoll J, Callies C, Nieschlag E, Simoni M. Mutation analysis of the X-chromosome linked, testis-specific TAF7L gene in spermatogenic failure. Andrologia. 2007;39:190–5.
Lee J, Park HS, Kim HH, Yun YJ, Lee DR, Lee S. Functional polymorphism in H2BFWT-5’UTR is associated with susceptibility to male infertility. J Cell Mol Med. 2009;13:1942–51.
Olesen C, Silber J, Eiberg H, Ernst E, Petersen K, Lindenberg S, Tommerup N. Mutational analysis of the human FATE gene in 144 infertile men. Hum Genet. 2003;113:195–201.
Ravel C, El Houate B, Chantot S, Lourenco D, Dumaine A, Rouba H, Bandyopadahyay A, Radhakrishna U, Das B, Sengupta S, Mandelbaum J, Siffroi JP, McElreavey K. Haplotypes, mutations and male fertility: the story of the testis-specific ubiquitin protease USP26. Mol Hum Reprod. 2006;12:643–6.
Stouffs K, Lissens W, Tournaye H, Van Steirteghem A, Liebaers I. Possible role of USP26 in patients with severely impaired spermatogenesis. Eur J Hum Genet. 2005;13:336–40.
Stouffs K, Lissens W, Tournaye H, Van Steirteghem A, Liebaers I. Alterations of the USP26 gene in Caucasian men. Int J Androl. 2006;29:614–7.
Stouffs K, Tournaye H, Van der Elst J, Liebaers I, Lissens W. Is there a role for the nuclear export factor 2 gene in male infertility? Fertil Steril. 2008;90:1787–91.
Stouffs K, Tournaye H, Liebaers I, Lissens W. Male infertility and the involvement of the X chromosome. Hum Reprod Update. 2009;15:623–37.
Visser L, Westerveld GH, Xie F, van Daalen SK, van der Veen F, Lombardi MP, Repping SA. Comprehensive gene mutation screen in men with asthenozoospermia. Fertil Steril. 2011;95:1020–4. e1021-1029
Borgmann J, Tüttelmann F, Dworniczak B, Röpke A, Song HW, Kliesch S, Wilkinson MF, Laurentino S, Gromoll J. The human RHOX gene cluster: target genes and functional analysis of gene variants in infertile men. Hum Mol Genet 2016; Sep 15. pii: ddw313.
Yang F, Gell K, van der Heijden GW, Eckardt S, Leu NA, Page DC, Benavente R, Her C, Höög C, McLaughlin KJ, Wang PJ. Meiotic failure in male mice lacking an X-linked factor. Genes Dev. 2008b;22(5):682–91.
World Health Organization. WHO laboratory manual for the examination and processing of human semen. 5th ed. WHO Press, 2010.
Geoffroy V, Pizot C, Redin C, Piton A, Vasli N, Stoetzel C, Blavier A, Laporte J, Muller J. VaRank: a simple and powerful tool for ranking genetic variants. Peer J. 2015;3:e796.
Quan J, Tian J. Circular polymerase extension cloning of complex gene libraries and pathways. PLoS One. 2009;4:e6441.
Rost B, Sander C. Prediction of protein secondary structure at better than 70% accuracy. J Mol Biol. 1993;232:584–99.
Rost B, Sander C. Combining evolutionary information and neural networks to predict protein secondary structure. Proteins. 1994;19:55–72.
Osterlund C, Tohonen V, Forslund KO, Nordqvist K. Mage-b4, a novel melanoma antigen (MAGE) gene specifically expressed during germ cell differentiation. Cancer Res. 2000;60:1054–61.
Newman JA, Cooper CDO, Roos AK, Aitkenhead H, Oppermann UCT, Cho HJ, Osman R, Gileadi O. Structures of two melanoma-associated antigens suggest allosteric regulation of effector binding. PLoS One. 2016;11:e0148762.
Avenarius MR, Hildebrand MS, Zhang Y, Meyer NC, Smith LL, Kahrizi K, Najmabadi H, Smith RJ. Human male infertility caused by mutations in the CATSPER1 channel protein. Am J Hum Genet. 2009;84:505–10.
Ben Khelifa M, Coutton C, Zouari R, Karaouzene T, Rendu J, Bidart M, Yassine S, Pierre V, Delaroche J, Hennebicq S, Grunwald D, Escalier D, Pernet-Gallay K, Jouk PS, Thierry-Mieg N, Toure A, Arnoult C, Ray PF. Mutations in DNAH1, which encodes an inner arm heavy chain dynein, lead to male infertility from multiple morphological abnormalities of the sperm flagella. Am J Hum Genet. 2014;94:95–104.
Caburet S, Arboleda VA, Llano E, Overbeek PA, Barbero JL, Oka K, Harrison W, Vaiman D, Ben-Neriah Z, Garcia-Tunon I, Fellous M, Pendas AM, Veitia RA, Vilain E. Mutant cohesin in premature ovarian failure. N Engl J Med. 2014;370:943–9.
Dam AH, Koscinski I, Kremer JA, Moutou C, Jaeger AS, Oudakker AR, Tournaye H, Charlet N, Lagier-Tourenne C, van Bokhoven H, Viville S. Homozygous mutation in SPATA16 is associated with male infertility in human globozoospermia. Am J Hum Genet. 2007;81:813–20.
Dieterich K, Soto Rifo R, Faure AK, Hennebicq S, Ben Amar B, Zahi M, Perrin J, Martinez D, Sele B, Jouk PS, Ohlmann T, Rousseaux S, Lunardi J, Ray PF. Homozygous mutation of AURKC yields large-headed polyploid spermatozoa and causes male infertility. Nat Genet. 2007;39:661–5.
Koscinski I, Elinati E, Fossard C, Redin C, Muller J, Velez de la Calle J, Schmitt F, Ben Khelifa M, Ray PF, Kilani Z, Barratt CL, Viville S. DPY19L2 deletion as a major cause of globozoospermia. Am J Hum Genet. 2011;88:344–50.
Doyle JM, Gao J, Wang J, Yang M, Potts PR. MAGE-RING protein complexes comprise a family of E3 ubiquitin ligases. Mol Cell. 2010;39:963–74.
Lo Giacco D, Chianese C, Ars E, Ruiz-Castané E, Forti G, Krausz C. Recurrent X chromosome-linked deletions: discovery of new genetic factors in male infertility. J Med Genet. 2014;51:340–4.
Stenson PD, Mort M, Ball EV, Howells K, Phillips AD, Thomas NS, Cooper DN. The Human Gene Mutation Database: 2008 update. Genome Med. 2009;1:13.
Hamby SE, Thomas NS, Cooper DN, Chuzhanova N. A meta-analysis of single base-pair substitutions in translational termination codons (‘nonstop’ mutations) that cause human inherited disease. Hum Genomics. 2011;5:241–64.
Cacciottolo M, Numitone G, Aurino S, Caserta IR, Fanin M, Politano L, Minetti C, Ricci E, Piluso G, Angelini C, Nigro V. Muscular dystrophy with marked dysferlin deficiency is consistently caused by primary dysferlin gene mutations. Eur J Hum Genet. 2011;19:974–80.
Gu LL, Li XH, Han Y, Zhang DH, Gong QM, Zhang XX. A novel homozygous no-stop mutation in G6PC gene from a Chinese patient with glycogen storage disease type Ia. Gene. 2014;536:362–5.
Oegema R, Hulst JM, Theuns-Valks SD, van Unen LM, Schot R, Mancini GM, Schipper ME, de Wit MC, Sibbles BJ, de Coo IF, Nanninga V, Hofstra RM, Halley DJ, Brooks AS. Novel no-stop FLNA mutation causes multi-organ involvement in males. Am J Med Genet A. 2013;161A:2376–84.
Wong SW, Liu HC, Han D, Chang HG, Zhao HS, Wang YX, Feng HL. A novel non-stop mutation in MSX1 causing autosomal dominant non-syndromic oligodontia. Mutagenesis. 2014;29:319–23.
Jenkins MM, LeBoeuf RD, Ruth GR, Bloomer JR. A novel stop codon mutation (X417L) of the ferrochelatase gene in bovine protoporphyria, a natural animal model of the human disease. Biochim Biophys Acta. 1998;1408:18–24.
Goldstein O, Jordan JA, Aguirre GD, Acland GM. A non-stop S-antigen gene mutation is associated with late onset hereditary retinal degeneration in dogs. Mol Vis. 2013;19:1871–84.
Chomez P, De Backer O, Bertrand M, De Plaen E, Boon T, Lucas S. An overview of the MAGE gene family with the identification of all human members of the family. Cancer Res. 2001;61:5544–51.
Acknowledgements
We thank all patients for their participation and donation of samples. We would also like to thank Robert Drillien for his critical reading of the manuscript. We are grateful to the IGBMC platforms. We thank Anne-Lena Bröcher from the Institute of Human Genetics, University of Münster for her excellent technical support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The study was funded by Agence Nationale de la Recherché (ANR-11-BSV2-002 “TranspoFertil”), Fondation Maladies Rares (“High throughput sequencing and rare diseases”), and l’Agence de BioMédecine (“AMP, diagnostic prénatal et diagnostic génétique”). This work was supported by the French Centre National de la Recherche Scientifique (CNRS), Institut National de la Santé et de la Recherche Médicale (INSERM), the Ministère de l’Education Nationale et de l’Enseignement Supérieur et de la Recherche, the University of Strasbourg, and Strasbourg University Hospital. The study was also supported by the German Research Foundation (DFG) (TU298/1-2 to FT and AR).
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This project has been approved by the Comité de Protection de la Personne (CPP) of Strasbourg University Hospital, France (CPP 09/40—W AC-2008-438 1W DC-2009-I 002), the “Istanbul University, Faculty of Medicine, Ethics Committee for Clinical Research Faculty of Medicine” (2012/1671-1265), and the Ethics Committee of the Medical Faculty in Münster (2010-578-f-S).
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Frank Tüttelmann and STéphane Viville are co-last authors.
Electronic supplementary material
ESM 1
(DOCX 32 kb).
Rights and permissions
About this article
Cite this article
Okutman, O., Muller, J., Skory, V. et al. A no-stop mutation in MAGEB4 is a possible cause of rare X-linked azoospermia and oligozoospermia in a consanguineous Turkish family. J Assist Reprod Genet 34, 683–694 (2017). https://doi.org/10.1007/s10815-017-0900-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-017-0900-z