Abstract
Purpose
In the present study, we reported two cases of TS with mosaic ring X chromosome showing common clinical characteristics of TS like growth retardation and ovarian dysfunction. The purpose of the present study was to cytogenetically characterize both cases.
Methods
Whole blood culture and G-banding were performed for karyotyping the cases following standard protocol. Origin of the ring chromosome and degree of mosaicism were further determined by fluorescence in situ hybridization (FISH). Breakpoints and loss of genetic material in formation of different ring X chromosomes r (X) in cases were determined with the help of cytogenetic microarray.
Results
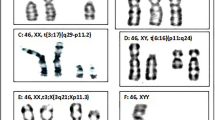
Cases 1 and 2 with ring chromosome were cytogenetically characterized as 45, X [114]/46Xr (X) (p22.11q21.32) [116] and 45, X [170]/46, Xr (X) (p22.2q21.33) [92], respectively. Sizes of these ring X chromosomes were found to be ~75 and ~95 Mb in cases 1 and 2, respectively, using visual estimation as part of cytogenetic observation. In both cases, we observed breakpoints on Xq chromosome were within relatively narrow region between Xq21.33 and Xq22.1 compared to regions in previously reported cases associated with ovarian dysgenesis.
Conclusions
Our observation agrees with the fact that despite of large heterogeneity, severity of the cases with intact X-inactive specific transcript (XIST) is dependent on degree of mosaicism and extent of Xq deletion having crucial genes involved directly or indirectly in various physiological involving ovarian cyclicity.




Similar content being viewed by others
References
Andreeva P. Thyroid gland and fertility. Akush Ginekol (Sofiia). 2014;53:18–23.
Atkins L, Sceery RT, Keenan ME. An unstable ring chromosome in a female infant with hypotonia, seizures, and retarded development. J Med Genet. 1966;3:134.
Baldwin EL, May LF, Justice AN, Martin CL, Led-better DH. Mechanisms and consequences of small supernumerary marker chromosomes: from Barbara McClintock to modern genetic counseling issues. Am J Hum Genet. 2008;82:398–410.
Cho EH, Kim SY, Kim JK. A case of 9.7 Mb terminal Xp deletion including OA1 locus associated with contiguous gene syndrome. J Korean Med Sci. 2012;27:1273–7.
Cho MK. Thyroid dysfunction and subfertility. Clin Exp Reprod Med. 2015;42:131–5.
de Moraes-Ruehsen M, Jones GS. Premature ovarian failure. Fertil Steril. 1967;18:440–61.
Dennis N, Coppin B, Turner C, Skuse D, Jacobs P. A clinical, cytogenetic and molecular study of 47 females with r (X) chromosomes. Ann Hum Genet. 2000;64:277–93.
Eggermann T, Meschede D, Schuler H, Palm S, Glaser D, Horsthemke B, et al. Premature ovarian failure associated with a small terminal Xq deletion: narrowing the POF1 region down to Xq27.2/Xq27.3-qter. Clin Genet. 2005;67(5):434–7.
Gazdag E, Santenard A, Ziegler-Birling C, Altobelli G, Poch O, Tora L, et al. TBP2 is essential for germ cell development by regulating transcription and chromatin condensation in the oocyte. Genes Dev. 2009;23:2210–23.
Guilherme RS, Klein E, Hamid AB, Bhatt S, Volleth M, Polityko A, et al. Human ring chromosomes—new insights for their clinical significance. BJMG. 2013;16:13–20.
Jacobs P, Dalton P, James R, Mosse K, Power M, Robinson D, et al. Turner syndrome: a cytogenetic and molecular study. Ann Hum Genet. 1997;61:471–83.
Jaiswal SK, Kumar A, Ali A, Rai AK. Co-occurrence of mosaic supernumerary isochromosome 18p and intermittent 2q13 deletions in a child with multiple congenital anomalies. Gene. 2015;559:94–8.
James RS, Coppin B, Dalton P, Dennis NR, Mitchell C, Sharp AJ, et al. A study of females with deletions of the short arm of the X chromosome. Hum Genet. 1998;102:507–16.
Kim MK, Seok HH , Kim YS, Chin MU, Sung SR, Lee WS, et al. Molecular genetic and cytogenetic characterization of a partial Xp duplication and Xq deletion in a patient with premature ovarian failure. Gene. 2014; 534:54–59.
Knijnenburg J, van Haeringen A, Hansson KB, Lankester A, Smit MJ, Belfroid RD, et al. Ring chromosome formation as a novel escape mechanism in patients with inverted duplication and terminal deletion. Eur J Hum Genet. 2007;15:548–55.
Lachlan KL, Youings S, Costa T, Jacobs PA, Thomas NS. A clinical and molecular study of 26 females with Xp deletions with special emphasis on inherited deletions. Hum Genet. 2006;118:640–51.
Leppig KA, Disteche CM. Ring X and other structural X chromosome abnormalities: X inactivation and phenotype. Semin Reprod Med. 2001;19:147–57.
Leppig KA, Sybert VP, Ross JL, Cunniff C, Trejo T, Raskind WH, et al. Phenotype and X inactivation in 45, X/46, X, r (X) cases. Am J Med Genet A. 2004;128:276–84.
Lindgren V, Chen CP, Bryke CR, Lichter P, Page DC, Yang-Feng TL. Cytogenetic and molecular characterization of marker chromosomes in patients with mosaic 45, X karyotypes. Hum Genet. 1992;88:393–8.
Maraschio P, Tupler R, Barbierato L, Dainotti E, Larizza D, Bernardi F, et al. An analysis of Xq deletions. Hum Genet. 1996;97:375–81.
Marozzi A, Manfredini A, Tibileth MG, Furlan D, Villa N, Vegetti W, et al. Molecular definition of Xq common-deleted region in patients affected by premature ovarian failure. Hum Genet. 2000;1074:304–11.
Matsuo M, Muroya K, Adachi M, Tachibana K, Asakura Y, Nakagomi Y, et al. Clinical and molecular studies in 15 females with ring X chromosomes: implications for r (X) formation and mental development. Hum Genet. 2000;107:433–9.
Mercer CL, Lachlan K, Karcanias A, Affara N, Huang S, Jacobs PA, et al. Detailed clinical and molecular study of 20 females with Xq deletions with special reference to menstruation and fertility. Eur J Med Genet. 2013;56:1–6.
Merhi ZO, Roberts JL, Awonuga AO. A case of 46, X, der (X) t (X;X) (q22.1;p11) Xq22.1> Xqter in a 12 year old girl with premature ovarian failure. Gynecol Obstet Invest. 2007;63:1379.
Miller SA, Dykes DD, Polesky HF. A simple salting out procedure for extracting DNA from human nucleated cells. Nucl Acids Res. 1988;16:1215.
Ogata T, Matsuo N. Turner syndrome and female sex chromosome aberrations: deduction of the principal factors involved in the development of clinical features. Hum Genet. 1995;95:607–29.
Ogata T, Matsuo N, Fukushima Y, Saito M, Nose O, Miharu N, et al. FISH analysis for apparently simple terminal deletions of the X chromosome: identification of hidden structural abnormalities. Am J Med Genet. 2001;104:307–11.
Pfeiffer RA. Observations in a case of an X/Y translocation, t (X; Y) (p22; q11), in a mother and son. Cytogenet Cell Genet. 1980;26:150–7.
Powell CM, Taggart RT, Drumheller TC, Wangsa D, Qian C, Nelson LM, et al. Molecular and cytogenetic studies of an X autosome translocation in a patient with premature ovarian failure and review of the literature. Am J Med Genet. 1994;52:19–26.
Prandstraller D, Mazzanti L, Picchio FM, Magnani C, Bergamaschi R, Perri A, et al. Turner’s syndrome: cardiologic profile according to the different chromosomal patterns and long-term clinical follow-up of 136 nonpreselected patients. Pediatr Cardiol. 1999;20:108–12.
Rice S, Christoforidis N, Gadd C, Nikolaou D, Seyani L, Donaldson A, et al. Impaired insulin-dependent glucose metabolism in granulosa-lutein cells from anovulatory women with polycystic ovaries. Hum Reprod. 2005;20:373–81.
Rizzolio F, Bione S, Sala C, Goegan M, Gentile M, Gregato G, et al. Chromosomal rearrangements in Xq and premature ovarian failure: mapping of 25 new cases and review of the literature. Hum Reprod. 2006;21:1477–83.
Rossetti F, Rizzolio F, Pramparo T, Sala C, Bione S, Bernadi F, et al. A susceptibility gene for premature ovarian failure (POF) maps to proximal Xq28. Eur J Hum Genet. 2004;12:829–34.
Sala C, Arrigo G, Torri G, Martinazzi F, Riva P, Larizza L, et al. Eleven X chromosome breakpoints associated with premature ovarian failure (POF) map to a 15-Mb Yac contig spanning Xq21. Genomics. 1997;40:123–31.
Schlessinger D, Herrera L, Crisponi L, Mumm S, Percesepe A, Pellegrini M, et al. Genes and translocations involved in POF. Am J Med Genet. 2002;111:328–33.
Schuh-Huerta SM, Johnson NA, Rosen MP, Sternfeld B, Cedars MI, Reijo Pera RA. Genetic markers of ovarian follicle number and menopause in women of multiple ethnicities. Hum Genet. 2012;131:1709–24.
Seghezzi L, Maraschio P, Bozzola M, Maserati E, Tupler R, Marchi A, et al. Ring chromosome 9 with a 9p22.3-p24.3 duplication. Eur J Pediat. 1999;158:791–3.
Sigurdardottir S, Goodman BK, Rutberg J, Thomas GH, Jabs EW, Geraghty MT. Clinical, cytogenetic, and fluorescence in situ hybridization findings in two cases of “complete ring” syndrome. Am J Med Genet. 1999;87:384–90.
Stochholm K, Juul S, Juel K, Naeraa RW, Gravholt CH. Prevalence, incidence, diagnostic delay, and mortality in Turner syndrome. J Clin Endocrinol Metab. 2006;91:3897–902.
Sybert VP. The adult with Turners syndrome: May 18-21; Gothenburg, Sweden. Edited by: Albertsson-Wikland K, Ranke MB. Amsterdam, Lausanne, New York, Oxford, Shannon, Tokyo: Elsevier 1995.
Tharapel AT, Anderson KP, Simpson JL, Martens PR, Wilroy RS, Llerena JC, et al. Deletion (X) (q26.1 –N q28) in a proband and her mother: molecular characterization and phenotypic–karyotypic deductions. Am J Hum Genet. 1993;52:463–71.
Therman E, Laxova R, Susman B. The critical region on the humanXq. Hum Genet. 1990;85:455–61.
Tomkins DJ, McDonald HL, Farrell SA, Brown CJ. Lack of expression of XIST from a small ring X chromosome containing the XIST locus in a girl with short stature, facial dysmorphism and developmental delay. Eur J Hum Genet. 2002;10:44–51.
Toniolo D. X-linked premature ovarian failure: a complex disease. Curr Opin Genet Dev. 2006;16:293–300.
Van Dyke DL, Wiktor A, Palmer CG, Miller DA, Witt M, Babu VR, et al. Ullrich-Turner syndrome with a small ring X chromosome and presence of mental retardation. Am J Med Genet. 1992;43:996–1005.
Wei Y, Reveal B, Reich J, Laursen WJ, Senger S, Akbar T, et al. TORC1 regulators Iml1/GATOR1 and GATOR2 control meiotic entry and oocyte development in Drosophila. Proc Natl Acad Sci U S A. 2014;111:5670–7.
Wyss D, DeLozier CD, Daniell J, Engel E. Structural anomalies of the X chromosome: personal observation and review of non-mosaic cases. Clin Genet. 1982;21:145–59.
Zinn AR, Ross JL. Molecular analysis of genes on Xp controlling Turner syndrome and premature ovarian failure (POF). Semin Reprod Med. 2001;19:141–6.
Zinn AR, Tonk VS, Chen Z, Flejter WL, Gardner HA, Guerra R, et al. Evidence for a Turner syndrome locus or loci at Xp11.2–p22.1. Am J Hum Genet. 1998;63:1757–66.
Acknowledgments
We thank the parents for giving consent to publish the data. We record words of gratitude to our teacher Professor Rajiva Raman for critical analysis and comments. We are thankful to anonymous reviewers for their suggestions in improving the manuscript. We are thankful to Centre for Genetic Disorders, BHU, for chromosomal analysis and innovative programme of UGC for hormonal assay experiment and Interdisciplinary School of Life Sciences, BHU, Varanasi, for cytogenetic microarrays. Banaras Hindu University, Varanasi, and Indian Council of Medical Research, New Delhi, are highly acknowledged for providing fellowship. This work forms part of the contribution of the Disease biology thrust area under DBT, New Delhi, sponsored by Interdisciplinary School of Life Sciences, BHU.
Author information
Authors and Affiliations
Corresponding author
Additional information
Capsule
In the present study, molecular cytogenetic mapping of ring X chromosomes narrowed down the critical region associated with ovarian dysgenesis between Xq21.33 to Xq22.1. The study further suggested that severity of clinical manifestation in the cases is dependent on degree of mosaicism and extent of Xq deletion.
Pooja Chauhan and Sushil Kumar Jaiswal contributed equally to this work.
Rights and permissions
About this article
Cite this article
Chauhan, P., Jaiswal, S.K., Lakhotia, A.R. et al. Molecular cytogenetic characterization of two Turner syndrome patients with mosaic ring X chromosome. J Assist Reprod Genet 33, 1161–1168 (2016). https://doi.org/10.1007/s10815-016-0761-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-016-0761-x