Abstract
Purpose
Heat shock protein A2 (HspA2) expression was quantitatively measured in human testis and its relationship with the spermatogenetic status and laboratory outcomes of intracytoplasmic sperm injection (ICSI) was investigated.
Methods
Testicular tissues of azoospermia men were divided into four groups according to histopahtology: normal spermatogenesiss, hypospermatogenesis, maturation arrest and Sertoli cell-only syndrome (SCOS). HspA2 immunostaining was measured by Image Pro-Plus (IPP) and laboratory outcomes were calculated. The regression analysis between HspA2 expression and Johnsen score of as well as fertilization, cleavage and high quality embryo rate was performed.
Results
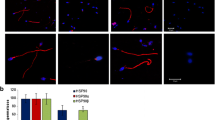
HspA2 was strongly present in the cytoplasm of spermatocytes and spermatides in normal testis. However, hypospermatogenesis and maturation arrest testicular tissues demonstrated light staining and no staining for SCOS. Quantitative image analysis showed that there were significant differences among groups (P = 0.000 & P = 0.001). HspA2 exspression was founded significantly correlated spermatogenetic status (R2 = 0.726, P = 0.000) as well as fertilization rate in ICSI (R2 = 0.569, P = 0.000).
Conclusions
The fertilization rate with ICSI is associated with HspA2 expression in the testis from which sperm retrieved and the alteration of HspA2 expression has been involved in spermatogenic impairment.




Similar content being viewed by others
References
Georgopoulos C, Welch WJ. Role of the major heat shock proteins as molecular chaperones. Annu Rev Cell Biol. 1993;9:601–34.
Huszar G, Stone K, Dix D, Vigue L. Putative creatine kinase M-isoform in human sperm is identifiedas the 70-kilodalton heat shock protein HspA2. Biol Reprod. 2000;63(3):925–32.
Feng HL, Sandlow JI, Sparks AE. Decreased expression of the heat shock protein hsp70-2 is associated with the pathogenesis of male infertility. Fertil Steril. 2001;76(6):1136–9.
Son WY, Han CT, Hwang SH, Lee JH, Kim S, Kim YC. Repression of hspA2 messenger RNA in human testes with abnormal spermatogenesis. Fertil Steril. 2000;73(6):1138–44.
Huszar G, Vigue L, Morshedi M. Sperm creatine phosphokinase M-isoform ratios and fertilizing potential of men: a blinded study of 84 couples treated with in vitro fertilization. Fertil Steril. 1992;57(4):882–8.
Huszar G, Vigue L, Corrales M. Sperm creatine phosphokinase activity as a measure of sperm quality in normospermic, variablespermic, and oligospermic men. Biol Reprod. 1988;38(5):1061–6.
Ergur AR, Dokras A, Giraldo JL, Habana A, Kovanci E, Huszar G. Sperm maturity and treatment choice of in vitro fertilization (IVF) or intracytoplasmic sperm injection: diminished sperm HspA2 chaperone levels predict IVF failure. Fertil Steril. 2002;77(5):910–8.
Johnsen SG. Testicular biopsy score count–a method for registration of spermatogenesis in human testes: normal values and results in 335 hypogonadal males. Hormones. 1970;1(1):2–25.
Gaskell TL, Esnal A, Robinson LL, Anderson RA, Saunders PT. Immunohistochemical profiling of germ cells within the human fetal testis: identification of three subpopulations. Biol Reprod. 2004;71(6):2012–21.
Veek LL. The morphological assessment of human oocytes and early conception. In: Keel BA, Webster BW, editors. Hand-book of the laboratory diagnosis and treatment of infertility. Voca Raton: CRC Press; 1990. p. 353–69.
Mayer MP, Bukau B. Hsp70 chaperones: cellular functions and molecular mechanism. Cell Mol Life Sci. 2005;62(6):670–84.
Dix DJ, Allen JW, Collins BW, Poorman-Allen P, Mori C, Blizard DR, et al. HSP70-2 is required for desynapsis of synaptonemal complexes during meiotic prophase in juvenile and adult mouse spermatocytes. Development. 1997;124(22):4595–603.
Dix DJ, Allen JW, Collins BW, Mori C, Nakamura N, Poorman-Allen P, et al. Targeted gene disruption of Hsp70-2 results in failed meiosis, germ cell apoptosis, and male infertility. Proc Natl Acad Sci U S A. 1996;93(8):3264–8.
Bonnycastle LL, Yu CE, Hunt CR, Trask BJ, Clancy KP, Weber JL, et al. Cloning, sequencing, and mapping of the human chromosome 14 heat shock protein gene (HSPA2). Genomics. 1994;23(1):85–93.
Cedenho AP, Lima SB, Cenedeze MA, Spaine DM, Ortiz V, Oehninger S. Oligozoospermia and heat-shock protein expression in ejaculated spermatozoa. Hum Reprod. 2006;21(7):1791–4.
Lima SB, Cenedeze MA, Bertolla RP, Filho PA, Oehninger S, Cedenho AP. Expression of the HSPA2 gene in ejaculated spermatozoa from adolescents with and without varicocele. Fertil Steril. 2006;86(6):1659–63.
Motiei M, Tavalaee M, Rabiei F, Hajihosseini R, Nasr-Esfahani MH. Evaluation of HSPA2 in fertile and infertile individuals. Andrologia. 2013;45(1):66–72.
Khosravanian N, Razi M, Farokhi F, Khosravanian H. Testosterone and vitamin E administration up-regulated varicocele-reduced Hsp70-2 protein expression and ameliorated biochemical alterations. J Assist Reprod Genet. 2014;31(3):341–54.
Cayli S, Sakkas D, Vigue L, Demir R, Huszar G. Cellular maturity and apoptosis in human sperm: creatine kinase, caspase-3 and Bcl-XL levels in mature and diminished maturity sperm. Mol Hum Reprod. 2004;10(5):365–72.
Dix DJ, Rosario-Herrle M, Gotoh H, Mori C, Goulding EH, Barrett CV, et al. Developmentally regulated expression of Hsp70-2 and a Hsp70-2/lacZ transgene during spermatogenesis. Dev Biol. 1996;174(2):310–21.
Afiyani AA, Deemeh MR, Tavalaee M, Razi M, Bahadorani M, Shokrollahi B, et al. Evaluation of heat-shock protein A2 (HSPA2) in male rats before and after varicocele induction. Mol Reprod Dev. 2014;81(8):766–76.
Kovanci E, Kovacs T, Moretti E, Vigue L, Bray-Ward P, Ward DC, et al. FISH assessment of aneuploidy frequencies in mature and immature human spermatozoa classified by the absence or presence of cytoplasmic retention. Hum Reprod. 2001;16(6):1209–17.
Nasr-Esfahani MHAH, Mirhosseini Z, Ghasemi N, Razavi SH, Tavalaei M, Tanhaei S, et al. Can altered expression of can altered expression of HSPA2 in varicocele patients lead to abnormal spermatogenesis? Int J Fertil Steril. 2010;4(3):104–13.
Redgrove KA, Nixon B, Baker MA, Hetherington L, Baker G, Liu DY, et al. The molecular chaperone HSPA2 plays a key role in regulating the expression of sperm surface receptors that mediate sperm-egg recognition. PLoS One. 2012;7(11):e50851.
Huszar G, Ozenci CC, Cayli S, Zavaczki Z, Hansch E, Vigue L. Hyaluronic acid binding by human sperm indicates cellular maturity, viability, and unreacted acrosomal status. Fertil Steril. 2003;79 Suppl 3:1616–24.
Wang CJ, Zhou ZG, Holmqvist A, Zhang H, Li Y, Adell G, et al. Survivin expression quantified by Image Pro-Plus compared with visual assessment. Appl Immunohistochem Mol Morphol. 2009;17(6):530–5.
Redgrove KA, Anderson AL, McLaughlin EA, O’Bryan MK, Aitken RJ, Nixon B. Investigation of the mechanisms by which the molecular chaperone HSPA2 regulates the expression of sperm surface receptors involved in human sperm-oocyte recognition. Mol Hum Reprod. 2013;19(3):120–35.
Francisco JS, Moraes HP, Dias EP. Evaluation of the Image-Pro Plus 4.5 software for automatic counting of labeled nuclei by PCNA immunohistochemistry. Braz Oral Res. 2004;18(2):100–4.
Prasad K, Prabhu GK. Image analysis tools for evaluation of microscopic views of immunohistochemically stained specimen in medical research — a review. J Med Syst. 2012;36(4):2621–31.
Eddy EM. HSP70-2 heat-shock protein of mouse spermatogenic cells. J Exp Zool. 1998;282(1–2):261–71.
Schwartz D, Goldfinger N, Rotter V. Expression of p53 protein in spermatogenesis is confined to the tetraploid pachytene primary spermatocytes. Oncogene. 1993;8(6):1487–94.
Kwon YK, Hecht NB. Cytoplasmic protein binding to highly conserved sequences in the 3′ untranslated region of mouse protamine 2 mRNA, a translationally regulated transcript of male germ cells. Proc Natl Acad Sci U S A. 1991;88(9):3584–8.
Huszar G, Jakab A, Sakkas D, Ozenci CC, Cayli S, Delpiano E, et al. Fertility testing and ICSI sperm selection by hyaluronic acid binding: clinical and genetic aspects. Reprod BioMed Online. 2007;14(5):650–63.
Huszar G, Ozkavukcu S, Jakab A, Celik-Ozenci C, Sati GL, Cayli S. Hyaluronic acid binding ability of human sperm reflects cellular maturity and fertilizing potential: selection of sperm for intracytoplasmic sperm injection. Curr Opin Obstet Gynecol. 2006;18(3):260–7.
Acknowledgments
Grant sponsor: National Natural Science Foundation, grant number: 30901493; National Key Technology Support Program, grant number: 2012BAI32B04 and the Ph.D. Programs Foundation of Ministry of Education of China, grant number: 200803351041.
Conflict of interest
No potential conflicts of interest relevant to this article were reported.
Author information
Authors and Affiliations
Corresponding author
Additional information
Capsule Testicular HspA2 is related with spermatogenesis and fertilization.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplement fig 1
Western blotting analysis of hsp70-2 protein in normal spermatogenesis, hypospermatogenesis and Sertoli cell only sydrome human testicular tissues. NS:normal spermatogenesis; HYPO: hypospermatogenesis; SCOS: Sertoli cell only sydrome. (GIF 38 kb)
Supplement fig 2
There was no significant correlation between HSPA2 immunostaining intensity and oocyte cleavage rate. Solid line indicates the regression line for immunostaining intensity and fertilization rate, while the two dotted curves depict the 95% confidence limits for the population regression line. (GIF 57 kb)
Supplement fig 3
There was no significant correlation between HSPA2 immunostaining intensity and high quality embryo rate. Solid line indicates the regression line for immunostaining intensity and fertilization rate, while the two dotted curves depict the 95% confidence limits for the population regression line. (GIF 56 kb)
Rights and permissions
About this article
Cite this article
Tian, Y., Zhang, F., Zhang, X. et al. Depression of HspA2 in human testis is associated with spermatogenic impairment and fertilization rate in ICSI treatment for azoospermic individuals. J Assist Reprod Genet 31, 1687–1693 (2014). https://doi.org/10.1007/s10815-014-0360-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-014-0360-7