Abstract
Objective
To investigate the serum levels of tumor necrosis factor-alpha (TNF-α), interleukin-6 (IL-6), monocyte chemotactic protein-1 (MCP-1), and Paraoxonase-1 (PON-1) during fertility treatment of women with endometriosis (Endo), PCOS or unexplained infertility (Unexpl).
Methods
Thirty-six patients with Endo, PCOS or Unexpl undergoing controlled ovarian stimulation for IVF or IUI were consented and their serum, on day-3 (baseline) and at the end of FSH treatment (peak), was collected and investigated for levels of TNF-α, IL-6, MCP-1, and PON-1. Correlations, ANOVA and Student's t-test were used for statistical analysis.
Results
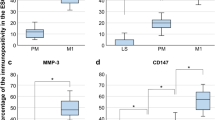
Peak serum levels of IL-6, MCP-1 and PON-1 were positively correlated to E2 peak levels. TNF-α levels were inversely correlated to estradiol levels and they were lower in patients who ultimately became pregnant when compared to non-pregnant (P < 0.05). Mean TNF-α levels were significantly higher in Unexpl group (P < 0.05). The mean levels of IL-6, and MCP-1 were significantly (p < 0.05) higher in women with PCOS compared with Endo and Unexpl. No differences were found between the three clinical groups in patient’s age, BMI, Day-3 FSH, PON-1 and pregnancy outcome.
Conclusion
Circulating cytokine levels were influenced by ovarian stimulation, as demonstrated by increased levels of IL-6, MCP-1 and PON-1, and decreased level of TNF-α at the end of controlled ovarian stimulation. While evidence of relationship between circulating cytokines with mild endometriosis was not found, PCOS was associated with elevated serum IL-6 and MCP-1 but lower TNF-α concentration. Unexplained infertility was associated with elevated TNF-α level. No relationship between serum PON-1 concentration and PCOS, mild endometriosis or unexplained infertility was noted.

Similar content being viewed by others
References
Adashi EY. The potential relevance of cytokines to ovarian physiology. J Steroid Biochem Mol Biol. 1992;43:439–44.
Brannstrom M. Potential role of cytokines in ovarian physiology: the case for Interleukin-1. In The Ovary (Elsevier). 2004: 261–271.
Zolti M, Ben-Rafael T, Meirom R, Shemesh M, Bider D, Mashiach S, et al. Cytokine involvement in oocytes and early embryos. Fertil Steril. 1991;56:265–72.
Premkumar B, Agarwal A. Female Infertility and Assisted Reproduction: Impact of Oxidative Stress –Current Women’s Health Reviews, 2012, 8: 183–207.
Demir B, Guven S, Guven ES, Atamer Y, Gul T. Serum IL-6 level may have role in the pathophysiology of unexplained infertility. Am J Repro Immunol. 2009;62:261–7.
Vassiliadis S., k Relakis, Papageorgiou, & i. Athanassakis. Endometriosis and infertility: A multi-cytokine imbalance versus ovulation, fertilization and early embryo development. Clinical & Develop Immunology, 2005; 12(2): 125–129.
Samy N, Hashim M, Sayed M, Said M. Clinical significance of inflammatory markers in polycystic ovary syndrome: their relationship to insulin resistance and body mass index. Dis Markers. 2009;26:163–70.
Dahm-Kähler P, Runesson E, Karin A, Brännström M. Monocyte chemotactic protein-1 in the follicle of the menstrual and IVF cycle. Mol Hum Reprod. 2006;12:1–6.
Marsillach J, Checa MA, Pedro-Botet J, Carreras R, Joven J, Camps J. Paraoxonase-1 in female infertility: a possible role against oxidative stress-induced inflammation. Fertil Steril. 2010;94:1132–4.
Younis A, Clower C, Nelsen D, Butler W, Carvalho A, Hok E, et al. The relationship between pregnancy and oxidative stress markers on patients undergoing ovarian stimulations. J Assist Reprod Genet. 2012;29(10):1083–9.
Adamson GD, Pasta DJ. Endometriosis fertility index: the new, validated endometriosis staging system. Fertil Steril. 2010;94:1609–15.
Jaichander P, Selvarajan K, Garelnabi M, Parthasarathy S. Induction of paraoxonase 1 & apolipoprotein A-I gene expression by aspirin. J Lipid Res. 2008;49(10):2142–8.
Buscher U, Chen FC, Kentenich H, Schmiady H. Cytokines in the follicular fluid of stimulated and nonstimulated human ovaries; is ovulation a suppressed inflammatory reaction? Hum Reprod. 1999;14(1):162–6.
Brännström M, Friden B, Jasper M, Norman R. Variations in peripheral blood levels of immunoreactive tumor necrosis factor alpha (TNF-α) throughout the menstrual cycle and secretion of TNF-α from the human corpus luteum. Eur J Obs Gyn Repro Biol. 1999;83:213–7.
Amato G, Conte M, Gherardo M, Lalli E, Vitolo G, Tucker A, et al. Serum and follicular fluid cytokines in polycystic ovary syndrome during stimulated cycles. Obstet Gynecol. 2003;101:1177–82.
Chiu KM, Arnaud AD, Ju J, Mayes D, Bacchetti P, Weitz S, et al. Correlation of estradiol, parathyroid hormone, interleukin-6, and soluble interleukin-6 receptor during the normal menstrual cycle. Bone. 2000;26:79–85.
Angstwurm MW, Gartner R, Ziegler-Heitbrock HW. Cyclic plasma IL-6 levels during normal menstrual cycle. Cytokine. 1997;9:370–4.
Souter I, Huang A, Martinez-Maza O, Breen EC, De Cherney AH, Chaudhuri G, et al. Serum levels of soluble vascular cell adhesion molecule-1, tumor necrosis factor-α, and interleukin-6 in in vitro fertilization cycles. Fertil Steril. 2009;91(5):2012–9.
Sarapik A., Velthut A., Haller-Kikkatalo K, Faure G, Marie-Christine, Carvalho M, Bittencourt, Fred´ericMassin, Raivo Uibo, and Salumets. Follicular Proinflammatory Cytokines and Chemokines as Markers of IVF Success. Clinical and Developmental Immunology. 2012; 606459, 10 pages
Browne RW, Shelly WB, Bloom MS, Ocquel AJ, Sandler JR, Huddleston HG. Distributions of high-density lipoprotein particle components in human follicular fluid and sera and their associations with embryo morphology parameters during IVF. Hum Reprod. 2008;23:1884–94.
Babayof R, Margalioth E, Huleihel M, Amash A, Zyllber-Haran E, Gal M, et al. Serum inhibin A, VEGF and TNF-a levels after triggering oocyte maturation with GnRH agonist compared with HCG in women with polycystic ovaries undergoing IVF treatment: a prospective randomized trial. Hum Reprod. 2006;21:1260–5.
Gonzalez F, Rote NS, Minium J, Kirwan JP. Evidence of proatherogenic inflammation in polycystic ovary syndrome. Metabolism. 2009;58:954–62.
Glintborg D, Andersen M, Richelsen B, Bruun JM. Plasma monocyte chemoattractant protein-1 (MCP-1) and macrophage inflammatory protein-1alpha are increased in patients with polycystic ovary syndrome (PCOS) and associated with adiposity, but unaffected by pioglitazone treatment. Clin Endocrinol. 2009;71:652–8.
Duleba AJ, Dokras A. Is PCOS an inflammatory process? Fertil Steril. 2012;97(1):7–12. doi:10.1016/j.fertnstert.2011.11.023.
Escobar-Morreale H, Luque-Ramírez M, González F. Circulating inflammatory markers in polycystic ovary syndrome: a systematic review and metaanalysis. Fertil Steril. 2011;95:1048–58.
Mohlig M, Spranger J, Osterhoff M, et al. The polycystic ovary syndrome per se is not associated with increased chronic inflammation. Eur J Endocrinol. 2004;150:525–32.
Pellicer A, Albert C, Mercader A, et al. The follicular and endocrine environment in women with endometriosis: local and systemic cytokine production. Fertil Steril. 1998;70:425–31.
Bragatto FB, Barbosa CP, Christofolini DM, Peluso C, dos Santos AA, Mafra FA, et al. There is no relationship between Paraoxonase serum level activity in women with endometriosis and the stage of the disease: an observational study. Reprod Health. 2013;10:32–7.
Verit FF, Erel O, Celik N. Serum paraoxonase-1 activity in women with endometriosis and its relationship with the stage of the disease. Hum Reprod. 2008;23:100–4.
Matorras R, Rodriguez F, Pijoan JI, Etaxanojaurequi A, Neyso JZ, Elorriaga MA, et al. Women who are not exposed to spermatozoa and infertile women have similar rates of stage I endometriosis. Fertil Steril. 2001;76:923–8.
Bedaiwy MA, Falcone T, Sharma RK, Goldberg JM, Attaran M, Nelson DR, et al. Prediction of endometriosis with serum and peritoneal fluid markers: a prospective controlled trial. Hum Reprod. 2002;17:426–31.
Melo A, Rosa-e-Silva C, Rosa-e-Silva A, Poli-Neto O, Ferriani R, Vieira C. Unfavorable lipid profile in women with endometriosis. Fertil Steril. 2010;10:2433–6.
Mihalyi A, Gevaert O, Kyama CM, Simsa P, Pochet N, De Smet F, et al. Non-invasive diagnosis of endometriosis based on a combined analysis of six plasma biomarkers. Hum Reprod. 2010;25:654–64.
Fassbender A, Vodolazkaia A, Saunders P, Lebovic D, Waelkens E, Bart De M, et al. Biomarkers of endometriosis. Fertil Steril. 2013;99:1135–45.
Tortorella C1, Piazzolla G1, Matteo M2, Pinto V2, Tinelli R2, Sabbà C1, Fanelli M1, Cicinelli E3. Interleukin-6, interleukin-1b, & tumor necrosis factor a in menstrual effluents as biomarkers of chronic endometritis. Fertil Steril. 2014; 101:242–247.
Gur M, Aslan M, Yildiz A, Demirbag R. Paraoxonase and arylesterase activities in coronary artery disease. Eur J Clin Invest. 2006;10:779–87.
Dursun P, Demirtas E, Bayrak A, Yarali H. Decreased serum paraoxonase 1 (PON1) activity: an additional risk factor for atherosclerotic heart disease in patients with PCOS? Hum Reprod. 2006;21:104–8.
Baskol G, Aygen E, Erdem F, Caniklioğlu A, Narin F, Şahin Y, et al. Assessment of paraoxonase 1, xanthine oxidase and glutathione peroxidase activities, nitric oxide and thiol levels in women with polycystic ovary syndrome. Acta Obstet Gynecol Scand. 2012;91:326–30.
Ray A, Shah A, Gudi A. Unexplained infertility: an update and review of practice. Reprod Bio Med Online. 2012;24(6):591–602.
Markert UR, Morales-Prieto DM, Fitzgerald JS. Understanding the link between the IL-6 cytokine family and pregnancy: implications for future therapeutics. Expert Rev Clin Immunol. 2011;7(5):603–9.
Demir B, Guven S, Guven ES, Atamer Y, Gul T. Serum IL-6 level may have role in the pathophysiology of unexplained infertility. Am J Repr Immunol. 2009;62:261–7.
Acknowledgments
The authors wish to thank all the staff at Central Georgia Fertility Institute, specially Cynthia Clower, RNC and Deanna Nelsen, RN, BSN, for their enthusiastic support. This work was partially supported by a faculty startup grant from the University of Massachusetts Lowell to M.G.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Capsule summary
The same group had higher MCP-1 levels when compared with endometriosis and unexplained groups at peak E2. Unexplained infertility was associated with elevated TNF-α level. PON-1 serum level increased after gonadotropin stimulation in all patient groups and the increased level was positively correlated with serum E2 concentration.
Rights and permissions
About this article
Cite this article
Younis, A., Hawkins, K., Mahini, H. et al. Serum tumor necrosis factor-α, interleukin-6, monocyte chemotactic protein-1 and paraoxonase-1 profiles in women with endometriosis, pcos, or unexplained infertility. J Assist Reprod Genet 31, 1445–1451 (2014). https://doi.org/10.1007/s10815-014-0237-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-014-0237-9