Abstract
Purpose
To investigate the effect of serum supplementing on short-term culture, fate determination and gene expression of goat spermatogonial stem cells (SSCs).
Methods
Crude testicular cells were plated over Datura-Stramonium Agglutinin (DSA) for 1 h, and non-adhering cells were cultured in the presence of different serum concentrations (1, 5, 10, and 15%) for 7 days in a highly enriched medium initially developed in mice. Colonies developed in each group were used for the assessment of morphology, immunocytochemistry, and gene expression.
Results
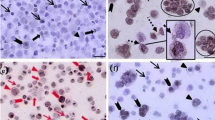
Brief incubation of testicular cells with DSA resulted in a significant increase in the number of cells that expressed the germ cell marker (VASA). The expression of THY1, a specific marker of undifferentiated spermatogonia, was significantly higher in colonies developed in the presence of 1% rather than 5, 10 and 15% serum.
Conclusion
Goat SSCs could proliferate and maintain in SSC culture media for 1 week at serum concentrations as low as 1%, while higher concentrations had detrimental effects on SSC culture/expansion.





Similar content being viewed by others
References
Tegelenbosch RA, de Rooij DG. A quantitative study of spermatogonial multiplication and stem cell renewal in the C3H/101 F1 hybrid mouse. Mutat Res. 1993;290:193–200.
Meistrich ML, van Beek MEAB. Spermatogonial stem cells. In: Desjardins CC, Ewing LL, editors. Cell and molecular biology of the testis. New York: Oxford University Press; 1993. p. 266–95.
Nagano M, Avarbock MR, Leonida EB, Brinster CJ, Brinster RL. Culture of mouse spermatogonial stem cells. Tissue Cell. 1998;30:389–97.
Kanatsu-Shinohara M, Inoue K, Ogonuki N, Morimoto H, Ogura A, Shinohara T. Serum- and feeder-free culture of mouse germline stem cells. Biol Reprod. 2011;84:97–105.
Phillips BT, Gassei K, Orwig KE. Spermatogonial stem cell regulation and spermatogenesis. Philos Trans R Soc Lond B Biol Sci. 2010;365:1663–78.
Kanatsu-Shinohara M, Ogonuki N, Inoue K, Miki H, Ogura A, Toyokuni S, et al. Long-term proliferation in culture and germline transmission of mouse male germline stem cells. Biol Reprod. 2003;69:612–6.
Lee J, Kanatsu-Shinohara M, Ogonuki N, Miki H, Inoue K, Morimoto T, et al. Heritable imprinting defect caused by epigenetic abnormalities in mouse spermatogonial stem cells. Biol Reprod. 2009;80:518–27.
Kanatsu-Shinohara M, Muneto T, Lee J, Takenaka M, Chuma S, Nakatsuji N, et al. Long-term culture of male germline stem cells from hamster testes. Biol Reprod. 2008;78:611–7.
Kubota H, Avarbock MR, Brinster RL. Growth factors essential for self-renewal and expansion of mouse spermatogonial stem cells. Proc Natl Acad Sci U S A. 2004;23(101):16489–94.
Meade HM, Echelard Y, Ziomek CA, Young MW, Harvey M, Cole ES, et al. Expression of recombinant proteins in the milk of transgenic animals. In: Fernandez JM, Hoeffler JP, editors. Gene expression systems: using nature for the art of expression. San Diego: Academic Press; 1998. p. 399–427.
Honaramooz A, Behboodi E, Hausler CL, Blash S, Mengee SO, Dobrinski I. Germ cell transplantation in goats. Mol Reprod Dev. 2003;64:422–8.
Honaramooz A, Behboodi E, Megee SO, Overton SA, Galantino-Homer H, Echelard Y, et al. Fertility and germline transmission of donor haplotype following germ cell transplantation in immunocompetent goats. Biol Reprod. 2003;69:1260–4.
Honaramooz A, Behboodi E, Hausler CL, Blash S, Ayres S, Azuma C, et al. Depletion of endogenous germ cells in male pigs and goats in preparation for germ cell transplantation. J Androl. 2005;26:698–705.
Wu Z, Falciatori I, Molyneux LA, Richardson TE, Chapman KM, Hamra FK. Spermatogonial culture medium: an effective and efficient nutrient mixture for culturing rat spermatogonial stem cells. Biol Reprod. 2009;81:77–86.
Freshney R. Culture of animal cells: a manual of basic technique. New York: Alan R. Liss, Inc; 1987. p. 117.
Scarpino S, Morena AR, Petersen C, Fröysa B, Söder O, Boitani C. A rapid method of Sertoli cell isolation by DSA lectin, allowing mitotic analyses. Mol Cell Endocrinol. 1998;25(146):121–7.
Kanatsu M, Nishikawa SI. In vitro analysis of epiblast tissue potency for hematopoietic cell differentiation. Development. 1996;122:823–30.
Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)). Methods. 2001;25:402–8.
Herrid M, Davey RJ, Hutton K, Colditz IG, Hill JR. A comparison of methods for preparing enriched populations of bovine spermatogonia. Reprod Fertil Dev. 2009;21:393–9.
Aponte PM, Soda T, Teerds KJ, Mizrak SC, van de Kant HJ, de Rooij DG. Propagation of bovine spermatogonial stem cells in vitro. Reproduction. 2008;136:543–57.
Kanatsu-Shinohara M, Miki H, Inoue K, Ogonuki N, Toyokuni S, Ogura A, et al. Long-term culture of mouse male germline stem cells under serum-or feeder-free conditions. Biol Reprod. 2005;72:985–91.
Acknowledgement
This study was funded by a grant from the Royan Institute (P83P88213). Authors declare that there is no conflict of interest in this study.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Summary Serum concentration adversely affects in-vitro culture of goat spermatogonial stem cells.
Capsule The result of this study showed maintenance and proliferation of goat SSC are adversely affected by high serum concentration.
Rights and permissions
About this article
Cite this article
Bahadorani, M., Hosseini, S.M., Abedi, P. et al. Short-term in-vitro culture of goat enriched spermatogonial stem cells using different serum concentrations. J Assist Reprod Genet 29, 39–46 (2012). https://doi.org/10.1007/s10815-011-9687-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-011-9687-5