Abstract
Purpose
To determine if diminished ovarian reserve (measured by maternal antimullerian hormone (AMH) levels), is associated with fetal aneuploidy (determined by prenatal karyotype).
Methods
This case-control study included 213 women with singleton pregnancies who underwent both serum aneuploidy screening and invasive prenatal diagnosis. 18 patients carrying an aneuploid fetus served as cases and the remaining 195 women with a euploid fetus were controls. Serum AMH was measured using two assays: AMHbc (Beckman-Coulter) and AMHdsl (Diagnostic Systems Laboratories). Karyotypes were determined by chorionic villus sampling or amniocentesis.
Results
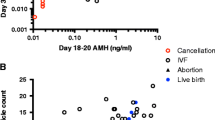
AMHbc levels did not differ between women with an aneuploid fetus and women with a euploid fetus (p = 0.46) and did not predict aneuploidy (ROC Area = 0.57). Additionally, AMHbc values declined significantly with advancing gestational age.
Conclusions
Maternal AMH does not appear to be a marker of fetal aneuploidy in ongoing pregnancies. Contrary to previous reports, we found a significant decline in maternal AMH levels with advancing gestational age.

Similar content being viewed by others
References
Maheshwari A, Fowler P, Bhattacharya S. Assessment of ovarian reserve—should we perform tests of ovarian reserve routinely? Hum Reprod. 2006;21(11):2729–35.
Hazout A, Bouchard P, Seifer DB, Aussage P, Junca AM, Cohen-Bacrie P. Serum antimullerian hormone/mullerian-inhibiting substance appears to be a more discriminatory marker of assisted reproductive technology outcome than follicle-stimulating hormone, inhibin B, or estradiol. Fertil Steril. 2004;82(5):1323–9.
Seifer DB, Maclaughlin DT. Mullerian Inhibiting Substance is an ovarian growth factor of emerging clinical significance. Fertil Steril. 2007;88(3):539–46.
Josso N, Picard JY, Rey R, di Clemente N. Testicular anti-Mullerian hormone: history, genetics, regulation and clinical applications. Pediatr Endocrinol Rev. 2006;3(4):347–58.
Jayaprakasan K, Campbell B, Hopkisson J, Johnson I, Raine-Fenning N. A prospective, comparative analysis of anti-Mullerian hormone, inhibin-B, and three-dimensional ultrasound determinants of ovarian reserve in the prediction of poor response to controlled ovarian stimulation. Fertil Steril. 2008 Nov 29.
Hehenkamp WJ, Looman CW, Themmen AP, de Jong FH, Te Velde ER, Broekmans FJ. Anti-Mullerian hormone levels in the spontaneous menstrual cycle do not show substantial fluctuation. J Clin Endocrinol Metab. 2006;91(10):4057–63.
Fanchin R, Schonauer LM, Righini C, Frydman N, Frydman R, Taieb J. Serum anti-Mullerian hormone dynamics during controlled ovarian hyperstimulation. Hum Reprod. 2003;18(2):328–32.
La Marca A, Giulini S, Orvieto R, De Leo V, Volpe A. Anti-Mullerian hormone concentrations in maternal serum during pregnancy. Hum Reprod. 2005;20(6):1569–72.
Somunkiran A, Yavuz T, Yucel O, Ozdemir I. Anti-Mullerian hormone levels during hormonal contraception in women with polycystic ovary syndrome. Eur J Obstet Gynecol Reprod Biol. 2007;134(2):196–201.
de Vet A, Laven JS, de Jong FH, Themmen AP, Fauser BC. Antimullerian hormone serum levels: a putative marker for ovarian aging. Fertil Steril. 2002;77(2):357–62.
van Rooij IA, Broekmans FJ, Scheffer GJ, Looman CW, Habbema JD, de Jong FH, et al. Serum antimullerian hormone levels best reflect the reproductive decline with age in normal women with proven fertility: a longitudinal study. Fertil Steril. 2005;83(4):979–87.
Mattei JF, Mattei MG, Ayme S, Giraud F. Origin of the extra chromosome in trisomy 21. Hum Genet. 1979;46(1):107–10.
Boue J, Bou A, Lazar P. Retrospective and prospective epidemiological studies of 1500 karyotyped spontaneous human abortions. Teratology. 1975;12(1):11–26.
Thum MY, Abdalla HI, Taylor D. Relationship between women’s age and basal follicle-stimulating hormone levels with aneuploidy risk in in vitro fertilization treatment. Fertil Steril. 2007 Oct 20.
Angell R. First-meiotic-division nondisjunction in human oocytes. Am J Hum Genet. 1997;61(1):23–32.
Angell R. Mechanism of chromosome nondisjunction in human oocytes. Prog Clin Biol Res. 1995;393:13–26.
Pellestor F, Andreo B, Arnal F, Humeau C, Demaille J. Maternal aging and chromosomal abnormalities: new data drawn from in vitro unfertilized human oocytes. Hum Genet. 2003;112(2):195–203.
Kuliev A, Cieslak J, Ilkevitch Y, Verlinsky Y. Chromosomal abnormalities in a series of 6, 733 human oocytes in preimplantation diagnosis for age-related aneuploidies. Reprod Biomed Online. 2003;6(1):54–9.
Tsuji K, Nakano R. Chromosome studies of embryos from induced abortions in pregnant women age 35 and over. Obstet Gynecol. 1978;52(5):542–4.
Speroff L, Fritz MA. Clinical gynecologic endocrinology and infertility. 7th ed. Philadelphia: Lippincott Williams & Wilkins; 2005.
Hassold T, Chiu D. Maternal age-specific rates of numerical chromosome abnormalities with special reference to trisomy. Hum Genet. 1985;70(1):11–7.
Menken J, Trussell J, Larsen U. Age and infertility. Science. 1986;233(4771):1389–94.
Nasseri A, Mukherjee T, Grifo JA, Noyes N, Krey L, Copperman AB. Elevated day 3 serum follicle stimulating hormone and/or estradiol may predict fetal aneuploidy. Fertil Steril. 1999;71(4):715–8.
van Montfrans JM, Dorland M, Oosterhuis GJ, van Vugt JM, Rekers-Mombarg LT, Lambalk CB. Increased concentrations of follicle-stimulating hormone in mothers of children with Down’s syndrome. Lancet. 1999;353(9167):1853–4.
Freeman SB, Yang Q, Allran K, Taft LF, Sherman SL. Women with a reduced ovarian complement may have an increased risk for a child with Down syndrome. Am J Hum Genet. 2000;66(5):1680–3.
van Montfrans JM, van Hooff MH, Martens F, Lambalk CB. Basal FSH, estradiol and inhibin B concentrations in women with a previous Down’s syndrome affected pregnancy. Hum Reprod. 2002;17(1):44–7.
Kline J, Kinney A, Reuss ML, Kelly A, Levin B, Ferin M, et al. Trisomic pregnancy and the oocyte pool. Hum Reprod. 2004;19(7):1633–43.
Massie JA, Burney RO, Milki AA, Westphal LM, Lathi RB. Basal follicle-stimulating hormone as a predictor of fetal aneuploidy. Fertil Steril. 2008;90(6):2351–5.
Weghofer A, Barad D, Li J, Gleicher N. Aneuploidy rates in embryos from women with prematurely declining ovarian function: a pilot study. Fertil Steril. 2007;88(1):90–4.
van Montfrans JM, Lambalk CB, van Hooff MH, van Vugt JM. Are elevated FSH concentrations in the pre-conceptional period a risk factor for Down’s syndrome pregnancies? Hum Reprod. 2001;16(6):1270–3.
Seifer DB, MacLaughlin DT, Cuckle HS. Serum mullerian-inhibiting substance in Down’s syndrome pregnancies. Hum Reprod. 2007;22(4):1017–20.
Penarrubia J, Fabregues F, Manau D, Creus M, Casals G, Casamitjana R, et al. Basal and stimulation day 5 anti-Mullerian hormone serum concentrations as predictors of ovarian response and pregnancy in assisted reproductive technology cycles stimulated with gonadotropin-releasing hormone agonist—gonadotropin treatment. Hum Reprod. 2005;20(4):915–22.
Eldar-Geva T, Ben-Chetrit A, Spitz IM, Rabinowitz R, Markowitz E, Mimoni T, et al. Dynamic assays of inhibin B, anti-Mullerian hormone and estradiol following FSH stimulation and ovarian ultrasonography as predictors of IVF outcome. Hum Reprod. 2005;20(11):3178–83.
Ebner T, Sommergruber M, Moser M, Shebl O, Schreier-Lechner E, Tews G. Basal level of anti-Mullerian hormone is associated with oocyte quality in stimulated cycles. Hum Reprod. 2006;21(8):2022–6.
Lie Fong S, Baart EB, Martini E, Schipper I, Visser JA, Themmen AP, et al. Anti-Mullerian hormone: a marker for oocyte quantity, oocyte quality and embryo quality? Reprod Biomed Online. 2008;16(5):664–70.
Lutterodt M, Byskov AG, Skouby SO, Tabor A, Yding Andersen C. Anti-Mullerian hormone in pregnant women in relation to other hormones, fetal sex and in circulation of second trimester fetuses. Reprod Biomed Online. 2009;18(5):694–9.
Nelson SM, Stewart F, Fleming R, Freeman DJ. Longitudinal assessment of antimullerian hormone during pregnancy-relationship with maternal adiposity, insulin, and adiponectin. Fertil Steril. 2009 Sep 30.
Freeman EW, Gracia CR, Sammel MD, Lin H, Lim LC, Strauss 3rd JF. Association of anti-mullerian hormone levels with obesity in late reproductive-age women. Fertil Steril. 2007;87(1):101–6.
Freour T, Mirallie S, Bach-Ngohou K, Denis M, Barriere P, Masson D. Measurement of serum anti-Mullerian hormone by Beckman Coulter ELISA and DSL ELISA: comparison and relevance in assisted reproduction technology (ART). Clin Chim Acta. 2007;375(1–2):162–4.
Acknowledgements
We thank Beckman Coulter for donating the Immunotech AMH assay kits used for this project.
Funding
1. University of North Carolina Medical Alumni Endowment Fund Grant
2. University of North Carolina Women’s Reproductive HealthResearch (WRHR) Program, 5K12HD050113 (NICHD)
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding author
Additional information
IRB approval: Protocol approved by the Institutional Review Board at the University of North Carolina at Chapel Hill
Capsule
Maternal antimullerian hormone is not a marker of fetal aneuploidy. Contrary to previous reports, antimullerian hormone levels in pregnancy decline significantly with advancing gestational age.
Rights and permissions
About this article
Cite this article
Plante, B.J., Beamon, C., Schmitt, C.L. et al. Maternal antimullerian hormone levels do not predict fetal aneuploidy. J Assist Reprod Genet 27, 409–414 (2010). https://doi.org/10.1007/s10815-010-9433-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-010-9433-4