Abstract
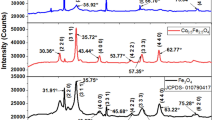
We have studied the magneto-optic Faraday effect (FE) and electron paramagnetic resonance (EPR) in an aluminum potassium borate glass containing Fe oxides as an impurity in a concentration of 1.5 mass % and Mn impurity in variable concentration from 0 to 1.2 mass %. When manganese oxide is added to the glass composition, the paramagnetic contribution to the Faraday effect increases more slowly than the change in the total concentration of paramagnetic ions, which allows us to hypothesize the appearance of clusters in which the paramagnetic ions are coupled by antiferromagnetic interactions. Formation of clusters upon addition of manganese oxide is confirmed by the change in the nature of the EPR spectra, where we observe a manganese concentration dependence of the distribution of iron atoms with respect to the different positions in the glass matrix. Heat treatment leads to a strong increase in the Faraday effect and a change in the spectral dependences of the Faraday effect and the EPR, which is explained by enlargement of the clusters and appearance of nanoparticles.
Similar content being viewed by others
References
S. A. Stepanov, G. T. Petrovskii, T. V. Zarubina, E. E. Kornilova, and I. S. Edel’man, Opt. Zh., 70, No. 12, 46–53 (2003).
I. S. Edel’man, S. A. Stepanov, G. T. Petrovskii, V. I. Zaikovskii, R. D. Ivantsov, O. S. Ivanova, D. E. Prokof’ev, T. V. Zarubina, and E. E. Kornilova, Fiz. i Khim. Stekla, 31, No. 2, 177–186 (2005).
R. Berger, J.-C. Bissey, and J. Kliava, J. Phys.: Condens. Matter., 12, 9347–9360 (2000).
I. Ardelean, P. Pascuta, and V. Ioncu, Int. J. Modern Phys. B, 17, 2633–2641 (2003).
J. Kliava and R. Berger, Recent Res. Devel. Non-Cryst. Solids, 3, 41–84 (2003).
J. Kliava and R. Berger, J. Magn. & Magn. Mater., 205, 328–342 (1999).
I. Ardelean, M. Peteanu, S. Filip, V. Simon, and G. Gyorgy, Solid State Commun., 102, 341–346 (1997).
D. Loveridge and S. Parke, Phys. Chem. Glasses, 12, 19–27 (1971).
A. Paul, Phys. Chem. Glasses, 14, 96–100 (1973).
T. Castner, G. S. Newell, W. C. Holton, and C. P. Slichter, J. Chem. Phys., 32, 668–673 (1960).
D. Loveridge and S. Parke, Phys. Chem. Glasses, 9, 73–81 (1968).
D. L. Griscom, J. Non-Cryst. Solids, 40, 211–272 (1980).
V. Cerny, B. Petrova, and M. Frumar, J. Non-Cryst. Solids, 125, 17–24 (1990).
I. Ardelean, M. Peteanu, V. Simon, S. Filip, F. Ciorcas, and I. Todor, J. Magn. & Magn. Mater., 258, 196–197 (1999).
D. W. Moon, M. J. M. Aitken, R. K. MacCrone, and G. S. Cieloszky, Phys. Chem. Glasses, 16, 91–102 (1975).
E. Burzo and I. Ardelean, Phys. Status Solidi (b), 87, K137–K140 (1978).
V. G. Miagkov, K. P. Polyakova, G. N. Bondarenko, and V. V. Polyakov, J. Magn. & Magn. Mater., 258–259, 358–360 (2003).
S. Simon, I. Ardelean, and M. Peteanu, Mod. Phys. Lett., 14, 1–6 (2000).
I. Ardelean and M. Flora, J. Mat. Sci.: Mat. Electr., 13, 357–362 (2002).
I. Ardelean, M. Peteanu, and S. Filip, Solid State Commun., 105, 339–344 (1998).
I. Ardelean, M. Peteanu, and V. Simon, J. Mat. Sci., 34, 6063–6068 (1999).
É. A. Petrakovskaya, V. G. Isakova, O. A. Bayukov, and D. A. Velikanov, Zh. Tekh. Fiz., 75, No. 5, 117–120 (2005).
Author information
Authors and Affiliations
Additional information
__________
Translated from Zhurnal Prikladnoi Spektroskopii, Vol. 73, No. 3, pp. 354–358, May–June, 2006.
Rights and permissions
About this article
Cite this article
Ivanova, O.S., Petrakovskaya, É.A., Ivantsov, R.D. et al. Effect of heat treatment and concentration of Mn and Fe on the structure of borate glass. J Appl Spectrosc 73, 400–405 (2006). https://doi.org/10.1007/s10812-006-0090-4
Received:
Issue Date:
DOI: https://doi.org/10.1007/s10812-006-0090-4