Abstract
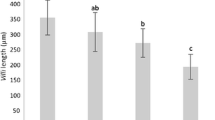
This study assessed the effects of the green seaweed Cladophora spp. as a protein source to substitute fishmeal protein in practical diets for postlarval tiger shrimp (Penaeus monodon). A control diet without containing seaweed meal was compared with five experimental diets in which fishmeal protein was replaced by increasing the levels of green seaweed protein (10, 20, 30, 40 and 50%) in the diets. All diets were formulated to be equivalent in crude protein (40%) and lipid (7%). Each dietary treatment was replicated in triplicate. After 45 days of the feeding trial, survival of the shrimp was not significantly influenced by the feeding treatments (p > 0.05). Growth rates of the shrimp fed with 10, 20, or 30% replacement levels of green seaweed protein in the diets were superior or similar to those fed with the control diet whereas shrimp fed with the diet containing 40 and 50% substitution levels showed significant reduction in growth (p < 0.05). The feed conversion ratio and protein efficiency ratio exhibited the similar trend as observed for growth performance. Moreover, shrimp fed with the control diet presented the lowest stress tolerance to formalin shock while the best performance was detected in the animals that received the 10 and 20% replacement diet. These results suggested that green seaweed Cladophora could replace up to 30% of fishmeal protein in the tiger shrimp postlarvae diet, and the optimal performance, including growth rate, feed efficiency, and stress resistance, appeared in shrimp fed with the diet of 10 to 20% substitution.


Similar content being viewed by others
References
Anh NTN, Toan NT, Hai TN (2013) Potential use of dried gut weed (Enterormorpha sp.) and blanket weed (Cladophoraceae) as a feed for the giant gourami (Osphronemus goramy) (abstract in English). Can Tho Uni J Sci 35:1040–1110
Anh NTN, Nhung DTK, Hai TN (2014) Replacement of soybean meal protein with gut weed (Enteromorpha sp.) and blanket weed (Cladophoracae) protein in practical diets for the white leg shrimp (Litopenaeus vannamei) postlarvae (abstract in English). Can Tho Uni J Sci 2:1580–1165
Anh NTN, Hong DT, Hai TN (2016) Investigating abundance and impacts of green seaweed (Cladophoraceae) in the improved extensive shrimp farms in Mekong delta. International Fisheries Symposium – IFS 2016 Promoting Healthier Aquaculture and Fisheries for Food Safety and Security. Phu Quoc Island of Vietnam, 31st October to 2nd November 2016, p 248
AOAC (2000) Official methods of analysis. Association of Official Analytical Chemists, Arlington 159 pp
Arnold SJ, Sellars MJ, Crocos PJ, Coman GJ (2006) Intensive production of juvenile tiger shrimp Penaeus monodon: an evaluation of stocking density and artificial substrates. Aquaculture 261:890–896
Azaza MS, Mensi F, Ksouri J, Dhraief MN, Brini B, Abdelmouleh A, Kraïem MM (2008) Growth of Nile tilapia, Oreochromis niloticus L., fed with diets containing graded levels of green algae Ulva meal, Ulva rigida, reared in geothermal waters of southern Tunisia. J Appl Ichthyol 24:202–207
Beiping T, Kangsen M, Shixuan Z, Qicun Z, Lihe L, Yu Y (2005) Replacement of fish meal by meat and bone meal in practical diets for the white shrimp Litopenaeus vannamei (Boone). Aquac Res 36:439–444
Briggs MRP, Funge-Smith SL (1996) The potential of Gracilaria spp. meal for supplementation of diets for juvenile Penaeus monodon Fabricius. Aquac Res 27:345–354
Brinkera A, Reiter R (2011) Fish meal replacement by plant protein substitution and guar gum addition in trout feed, part I: effects on feed utilization and fish quality. Aquaculture 10:350–360
Burtin P (2003) Nutritional value of seaweeds. Elect J Env Agricult Food Chem 2:1–6
Cárdenas JV, Gálvez AO, Brito LO, Galarza EV, Pitta DC, Rubin VV (2015) Assessment of different levels of green and brown seaweed meal in experimental diets for whiteleg shrimp (Litopenaeus vannamei, Boone) in recirculating aquaculture system. Aquac Int 23:1491–1504
Chien YH, Pan CH, Hunter B (2003) The resistance to physical stresses by Penaeus monodon juveniles fed diets supplemented with astaxanthin. Aquaculture 216:177–191
Cruz-Suárez LE, Tapia-Salazar M, Nieto-Lopez MG, Ricque-Marie D (2008) A review of the effect of macroalgae in shrimp feed and in co-culture. IX Simposio Internacional de Nutricion Acuicola, 24-27 Noviembre, Universidad Autonoma de Nuevo Leon, Monterrey, Nuevo Leon, Mexico:304-333
Cruz-Suárez LE, Tapia-Salazar M, Nieto-Lopez MG, Guajardo-Barbosa C, Ricque-Marie D (2009) Comparison of Ulva clathrata and the kelps Macrocystis pyrifera and Ascophyllum nodosum as ingredients in shrimp feeds. Aquac Nutr 15:421–430
da Silva RL, Barbosa JM (2009) Seaweed meal as a protein source for the white shrimp Litopenaeus vannamei. J Appl Phycol 21:193–197
Davis DA, Sookying D (2009) Strategies for reducing and/or replacing fish meal in production diets for the Pacific white shrimp, Litopenaeus vannamei. In: Browdy CL, Jory DE (eds) The rising tide, proceedings of the special session on shrimp farming. World Aquaculture Society, Baton Rouge, pp 91–96
Dhargalkar VK, Neelam P (2005) Seaweed: promising plant of the millennium. Sci Cult 71:60–66
Elizondo-Reyna E, Medina-González R, Nieto-López MG, Ortiz-López R, Elizondo-González R, Powell MS, Ricque-Marie D, Cruz-Suárez LE (2016) Consumption of Ulva clathrata as a dietary supplement stimulates immune and lipid metabolism genes in Pacific white shrimp Litopenaeus vannamei. J Appl Phycol 28:3667–3677
Felix N, Brindo RA (2014) Substituting fishmeal with fermented seaweed, Kappaphycus alvarezii in diets of juvenile freshwater prawn Macrobrachium rosenbergii. Int J Fish Aquat Stud 1:261–265
Ghosh R, Mitra A (2015) Suitability of green macroalgae Enteromorpha intestinalis as a feed form Macrobrachium rosenbergii. J Fish Livest Prod 3:138
Glencross BD, Booth M, Allan GL (2007) A feed is only as good as its ingredients—a review of ingredient evaluation for aquaculture feeds. Aquac Nutr 13:17–34
Hafezieh M, Ajdari D, Ajdehakosh PA, Hosseini SH (2014) Using Oman Sea Sargassum illicifolium meal for feeding white leg shrimp Litopenaeus vannamei. Iran J Fish Sci 13:73–80
Hai TN, Minh TH, Phu TQ, Phuong NT (2016) Shrimp industry in Vietnam. In: Liao IC, Chao NH, Leaño EM (ed) Progress of shrimp and prawn aquaculture in the world. National Taiwan University, the Fisheries Society of Taiwan, Asian Fisheries Society, and World Aquaculture Society, pp 181–204
ITB-Vietnam (2011) Study on distribution and culture of seaweeds and aquatic plants in the Mekong delta, Vietnam. International co-operation plan. Algen Sustainable & Center Novem, Netherlands
Khuantrairong T, Traichaiyaporn S (2011) The nutritional value of edible freshwater alga Cladophora sp. (Chlorophyta) grown under different phosphorus concentrations. Int J Agric Biol 13:1560–8530
Khuantrairong T, Traichaiyaporn S (2012) Enhancement of carotenoid and chlorophyll content of an edible freshwater alga (Kai: Cladophora sp.) by supplementary inorganic phosphate and investigation of its biomass production. Maejo Int J Sci Technol 6:1–11
Kumar V, Kaladharan P (2007) Amino acids in the seaweeds as an alternate source of protein for animal feed. J Mar Biol Assoc India 49:35–40
Lan NTP (2013) Social and ecological challenges of market-oriented shrimp farming in Vietnam. Spring 2:675
Meyers SP (1994) Developments in world aquaculture, feed formulations and role of carotenoids. Pure Appl Chem 66:1069–1076
Millamena OM, Bautista-Teruel MN, Kanazawa A (1996) Methionine requirement of juvenile tiger shrimp Penaeus monodon Fabricius. Aquaculture 143:403–410
Millamena OM, Bautista-Teruel MN, Reyes OS, Kanazawa A (1998) Requirements of juvenile marine shrimp, Penaeus monodon (Fabricius) for lysine and arginine. Aquaculture 164:95–104
Molina–Poveda C, Morales ME (2004) Use of a mixture of barley–based fermented grains and wheat gluten as an alternative protein source in practical diets for Litopenaeus vannamei. Aquac Res 35:1158–1165
Olsen RL, Hasan MR (2012) A limited supply of fishmeal: impact on future increases in global aquaculture production. Trends Food Sci Technol 27:120–128
Pallaoro MF, do Nascimento Vieira F, Hayashi L (2016) Ulva lactuca (Chlorophyta Ulvales) as co-feed for Pacific white shrimp. J Appl Phycol 28:3659–3665
Peixoto MJ, Salas-Leitóna E, Pereiraa LF, Queiroza A, Magalhãesa F, Pereirad R, Abreu H, Reis P, Goncalves JFM, Ozórioa ROA (2016) Role of dietary seaweed supplementation on growth performance, digestive capacity and immune and stress responsiveness in European seabass (Dicentrarchus labrax). Aquac Rep 3:189–197
Portley N (2016) Report on the shrimp sector: Asian shrimp trade and sustainability. Sustainable Fisheries Partnership. https://www.sustainablefish.org/. Accessed 14 Jun 2017
Potty HV (1996) Physio-chemical aspects, physiological functions, nutritional importance and technological significance of dietary fibres a critical appraisal. J Food Sci Technol 33:1–18
Pushparajan N, Soundarapandian P (2010) Recent farming of marine black tiger shrimp, Penaeus monodon (Fabricius) in South India. Afr J Basic Appl Sci 2:33–36
Qiu X, Neori A, Kim JK, Yarish C, Shpigel M, Guttman L, Ben Ezra D, Odintsov V, Davis DA (2017) Green seaweed Ulva sp. as an alternative ingredient in plant-based practical diets for Pacific white shrimp, Litopenaeus vannamei. J Appl Phycol. https://doi.org/10.1007/s10811-017-1288-y
Rivera G, Yoong F, Riofrio G, Reinoso B, Hurtado F, Massuh P (2002). Inclusión de harina de kelp (Macrocystis pyrifera) en alimentos balanceados para el camarón. Congreso Iberiamericano Virtual de Acuícultura. CIVA. http://www.civa2002.org, searched on 26 June 2013
Rodríguez-González H, Orduña-Rojas, Villalobos-Medina JP, García-Ulloa M, Polanco-Torres A, López-Álvarez ES, Montoya-Mejía M, Hernández-Llamas A (2014) Partial inclusion of Ulva lactuca and Gracilaria parvispora meal in balanced diets for white leg shrimp (Litopenaeus vannamei). J Appl Phycol 26:2453–2459
Samocha TM, Guajardob H, Lawrence AL, Castillec FL, Speedd M, McKee DA, Pagee KI (1998) A simple stress test for Penaeus vannamei postlarvae. Aquaculture 165:233–242
Santizo R, Serrano AE Jr, Corre VL (2014) Proximate composition and dry matter digestibility of Ulva lactuca in the black tiger shrimp Penaeus monodon. ABAH Bioflux 6:75–83
Serrano AE Jr, Santizo R (2014) Dietary substitution of protein concentrate of Ulva lactuca for soybean meal in the black tiger shrimp Penaeus monodon fry. ABAH Bioflux 6:140–147
Serrano AE Jr, Tumbokomn BLM (2015) Optimum dietary inclusion of Ulva intestinalis to the diet of the black tiger shrimp Penaeus monodon postlarvae. AACL Bioflux 7:169–176
Serrano AE Jr, Santizo RB, Tumbokon BLM (2015) Potential use of the sea lettuce Ulva lactuca replacing soybean meal in the diet of the black tiger shrimp Penaeus monodon juvenile. AACL Bioflux 8:245–252
Smith DM, Allan GL, Williams KC and Barlow C (2000) Fishmeal replacement research for shrimp feed in Australia. In: Cruz-Suárez LE, Ricque-Marie D, Tapia-Salazar M, Olvera-Novoa MA, Civera-Cerecedo R, (eds) Avances en Nutrición Acuícola V. Memorias del V Simposium Internacional de Nutrición Acuícola. 19-22 Noviembre, 2000. Mérida, Yucatán, México
Soltani S, Saadatmand S, Khavarinejad R, Nejadsattari T (2011) Antioxidant and antibacterial activities of Cladophora glomerata (L.) Kütz. in Caspian Sea coast, Iran. Afr J Biotechnol 10:7684–7689
Stabili L, Acquaviva MI, Biandolino F, Cavallo RA, De Pascali SA, Fanizzi FP, Narracci M, Cecere E, Petrocelli A (2014) Biotechnological potential of the seaweed Cladophora rupestris (Chlorophyta, Cladophorales) lipidic extract. Nat Biotechnol 31:436–444
Sudaryono A, Haditomo AH, Isnansetyo A (2015) Evaluation of dietary supplementation of aqueous extract of brown algae Sargassum cristaefolium on growth performance and feed utilization of juvenile white shrimp Litopenaeus vannamei. AACL Bioflux 8:142–149
Teshima S, Ishikawa M, Koshio S (2000) Nutritional assessment and feed intake of microparticulate diets in crustaceans and fish. Aquac Res 31:691–702
Thanh T, Nghia TT, Phuong NT (2002) Advanced technologies for shrimp larviculture of Penaeus monodon in the bio-filter system (abstract in English). Can Tho Uni J Sci 2:185–190
Wells ML, Potin P, Craigie JS, Raven JA, Merchant SS, Helliwell KE, Smith AG, Camire ME, Brawley SH (2017) Algae as nutritional and functional food sources: revisiting our understanding. J Appl Phycol 29:949–982
Yahyavi M, Takami GHA, Vosoughi GH (2007) The study of salinity and formalin stress tests in postlarval feeding of Indian white shrimp by enriched rotifers with unsaturated fatty acids (DHA, EPA) and vitamin C. JWSS 10:519–531
Zbakh H, Chiheb I, Motilva V, Riadi H (2014) Antibacterial, cytotoxic and antioxidant potentials of Cladophora prolifera (Roth) Kutzing collected from the Mediterranean coast of Morocco. Am J Phytomed Clin Therapeut 2:1187–1199
Zorriehzahra MJ, Banaederakhshan R (2015) Early mortality syndrome (EMS) as new emerging threat in shrimp industry. Adv Anim Vet Sci 3:64–72
Acknowledgments
The authors sincerely thank Dr. Nguyen Xuan Hoa, Nha Trang Institute of Oceanography and Ms. Huynh Ly Huong for their help in identifying the genus of green seaweed and Le Thanh Y for his help during the feeding trial.
Funding
This study was funded by the Vietnamese Ministry of Education and Training for the scientific research programs.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Anh, N.T.N., Hai, T.N. & Hien, T.T.T. Effects of partial replacement of fishmeal protein with green seaweed (Cladophora spp.) protein in practical diets for the black tiger shrimp (Penaeus monodon) postlarvae. J Appl Phycol 30, 2649–2658 (2018). https://doi.org/10.1007/s10811-018-1457-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-018-1457-7