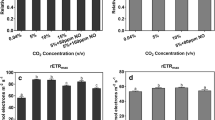
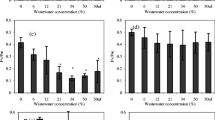
Abstract
Leachate sewage ponds at Phuket Integrated Waste Management (Phuket, Thailand) are typical hypereutrophic red-water ponds found at sewage treatment plants and piggery, feedlot and poultry waste ponds with mixed communities of anoxygenic purple photosynthetic bacteria (PPB) (Bacteriochlorophyll a) and Chlorella-type green algae (Chl a + b). In vivo integrating sphere spectrometer scans (Model A&E-S90-2D, A&E Lab (UK)) showed absorbance maxima at 678–680 nm (in vivo Chl a) and a double peak at 802 and 844 nm (in vivo BChl a). High Na2S (8.3 mol m−3) added to PM media selected for the PPB whereas Chlorella overwhelmed PPB in PM medium without high H2S. Photosynthetic electron transport rate (ETR) was measured using a blue-diode-based Junior PAM (Pulse Amplitude Modulation Fluorometer) on sewage pond leachate filtered onto glass fibre disks. Diuron herbicide resistance experiments allowed measurement of both oxygenic and anoxygenic photosynthesis of a mixed population of oxygenic and anoxygenic organisms to be estimated only in special circumstances. In separate culture, the ETR vs. E curves were Chlorella-type algae, Eopt ≈ 191 ± 10 μmol quanta m−2 s−1, ETRmax = 184 ± 6.7 μmol e− g−1 Chl a s−1; PPB, Eopt = 386 ± 15 μmol quanta m−2 s−1, ETRmax = 316 ± 7.3 μmol e− g−1 BChl a s−1 but in a mixture of Chlorella and PPB only the oxygenic photosynthesis could be detected. In sewage pond samples, PAM rapid light curves in the presence and absence of DCMU allowed separate estimates of oxygen and anoxygenic photosynthesis to be made only if the Chl a content was very low (Chl a ≈ 0.26 μg mL−1; BChl a ≈ 1.4 μg mL−1). If substantial amounts of Chl a were present, fluorescence from PSII overwhelmed the signal from RC-2 of PPB, preventing the detection of anoxygenic photosynthesis. New PAM technology to measure Chl a and BChl a fluorescence separately is needed.








Similar content being viewed by others
References
Apichatmeta K, Sudsiri CJ, Ritchie RJ (2017) Photosynthesis of oil palm (Elaeis guineensis). Sci Hortic 214:34–40
Belila B, Abbas B, Fazaa I, Saidi N, Snoussi M, Hassen A, Muyzer G (2013) Sulphur bacteria in wastewater stabilization ponds periodically affected by the “red-water” phenomenon. Appl Microbiol Biotechnol 97:379–394
Brestic M, Zivcak M (2013) PSII fluorescence techniques for measurement of drought and high temperature stress signal in plants: protocols and applications. In: Rout GR, Das AB (eds) Molecular stress physiology in plants. Springer, Dordrecht, pp 87–131
Blankenship RE, Madigan MT, Bauer CE (1995) Anoxygenic photosynthetic bacteria. Kluwer Academic Publishers, Dordrecht
Cochran WG, Snedecor GW (1989) Statistical methods, 8th edn. Iowa State University Press, Ames
Dewez B, Didur O, Vincent-Heroux J, Popovic R (2008) Validation of photosynthetic-fluorescence parameters as biomarkers for isoproturon toxic effect on alga Scenedesmus obliquus. Environ Pollut 151:93–100
Falkowski PG, Raven JA (2007) Aquatic photosynthesis, 2nd edn. Princeton University Press, Princeton
Genty B, Briantais JM, Baker NR (1989) The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. Biochim Biophys Acta 990:87–92
Gitelson A, Stark R, Oron G, Dor I (1997) Monitoring of polluted water bodies by remote sensing. In: Remote sensing and geographic information systems for design and operation of water resources systems (Proceedings of Rabat Symposium S3, April 1997). IAHS Publ 242:181–188
Gitelson A, Stark R, Dor I, Michielson O, Yacobi YZ (1999) Optical characteristics of the phototroph Thiocapsa roseopersicina and implication for real time monitoring of the bacteriochlorophyll concentration. Appl Environ Microbiol 65:3392–3397
Goericke R (2002) Bacteriochlorophyll a in the ocean: is anoxygenic bacterial photosynthesis important. Limnol Oceanogr 47:290–295
Haynes D, Ralph P, Pranges J, Dennison B (2000) The impact of the herbicide diuron on photosynthesis in three species of tropical seagrass. Mar Pollut Bull 41:288–293
Hubas C, Jesus B, Passarelli C, Jeanthon C (2011) Tools providing new insight into coastal anoxygenic purple bacterial mats. Res Microbiol 162:858–868
Irving DE, Dromgoole FI (1986) Algal populations and characteristics of oxygen exchange of effluent samples from a facultative oxidation pond. N Z J Mar Fresh 20:9–16
Koelsch RK, Chen TT, Sculte DD (1997) Purple sulphur bacteria in anaerobic treatment lagoons. Nebraska Swine Rep Pap 205:36–38 http://digitalcommons.unl.edu/coopext_swine/205
Kim MK, Harwood CS (1991) Regulation of benzoate-CoA ligase in Rhodopseudomonas palustris. FEMS Microbiol Lett 83:199–203
Kim JK, Lee BK (2000) Mass production of Rhodopseudomonas palustris as diet for aquaculture. Aquac Eng 23:281–293
Kim MK, Choi K-M, Yin C-R, Lee K-Y, Im WT, Lim JH, Lee ST (2004) Odorous swine wastewater treatment by purple non-sulfur bacteria, Rhodopseudomonas palustris, isolated from eutrophicated ponds. Biotechnol Lett 26:819–822
Kolber ZS, Van Dover CL, Niederman RA, Falkowski PG (2000) Bacterial photosynthesis in surface waters of the open ocean. Nature 407:177–179
Larimer FW, Chain P, Hauser L, Lamerdin J, Malfatti S, Do L, Land ML, Pelletier DA, Beatty JT, Lang AS, Tabita FR, Gibson JL, Hanson TE, Bobst C, Torres JLT, Peres C, Harrison FH, Gibson J, Harwood CS (2004) Complete genome sequence of the metabolically versatile photosynthetic bacterium Rhodopseudomonas palustris. Nat Biotechnol 22:55–61
Nicholls HW (1973) Culture media-freshwater. In: Stein JR (ed) Handbook of phycological methods: culture methods and growth measurements, vol 1. Cambridge University Press, Cambridge, pp 7–24
Papineau D, Walker JJ, Mojzsis SJ, Pace NR (2005) Composition and structure of microbial communities from stromatolites of Hamelin Pool in Shark Bay, Western Australia. Appl Environ Microbiol 2005:4822–4832
Porra RJ (2006) Spectrometric assays for plant, algal and bacterial chlorophylls. In: Grimm B, Porra RJ, Rüdiger W, Scheer H (eds) Chlorophylls and bacteriochlorophylls. Springer, Dordrecht, pp 95–107
Quinnell R, Howell D, Ritchie RJ (2017) Photosynthesis of an epiphytic resurrection fern Davallia angustata (Wall, ex Hook. & Grev.) Aust J Bot 65:348–356
Ralph PJ, Gademann R (2005) Rapid light curves: a powerful tool to assess photosynthetic activity. Aquat Bot 82:222–237
Rascher U, Liebig M, Lüttge U (2000) Evaluation of instant light-response curves of chlorophyll fluorescence parameters obtained with a portable chlorophyll fluorometer on site in the field. Plant Cell Environ 23:1397–1405
Ritchie RJ (2006) Consistent sets of spectrophotometric equations for acetone, methanol and ethanol solvents. Photosynth Res 89:27–41
Ritchie RJ (2008) Fitting light saturation curves measured using PAM fluorometry. Photosynth Res 96:201–215
Ritchie RJ (2013) The use of solar radiation by a photosynthetic bacterium, Rhodopseudomonas palustris: model simulation of conditions found in a shallow pond or flatbed reactor. Photochem Photobiol 89:1143–1162
Ritchie RJ (2015) Photosynthetic light curve fitting models. Available at www.researchgate.net/publication/283052624_Photosynthetic_Light_Curve_Fitting_Models. [Verified 26 May 2016]
Ritchie RJ (2018) Measurement of chlorophylls a and b and bacteriochlorophyll a in organisms from hypereutrophic auxinic waters. J Appl Phycol. https://doi.org/10.1007/s10811-018-1431-4
Ritchie RJ, Larkum AWD (2013) Modelling photosynthesis in shallow algal production ponds. Photosynthetica 50:481–500
Ritchie RJ, Larkum AWD, Ribas A (2017) Could photosynthesis function on Proxima Centauri b? Int J Astrobiol:1–30
Ritchie RJ, Mekjinda N (2015) Measurement of photosynthesis using PAM technology in a purple sulphur bacterium Thermochromatium tepidum (Chromatiaceae). Photochem Photobiol 91:350–358
Ritchie RJ, Runcie JW (2013) Measurement of the photosynthetic electron transport rate in an anoxygenic photosynthetic bacterium Afifella (Rhodopseudomonas) marina using PAM fluorometry. Photochem Photobiol 89:370–383
Ritchie RJ, Runcie JW (2014) A portable reflectance-absorptance-transmittance (RAT) meter for vascular plant leaves. Photosynthetica 52:614–626
Siefert E, Irgens RL, Pfennig N (1978) Phototrophic purple and green bacteria in a sewage treatment plant. Appl Environ Microbiol 35:38–41
Sinning I (1992) Herbicide binding in the bacterial photosynthetic reaction center. TIBS 17:150–154
Sinning I, Michel H, Mathis P, Rutherford AW (1989) Terbutryn resistance in a purple bacterium can induce sensitivity toward the plant herbicide DCMU. FEBS Lett 256:192–194
Sinning I, Koepke J, Michel H (1990) Recent advances in the structure analysis of Rhodopseudomonas viridis reaction center mutants. In: Michel-Bayerle M-E (ed) Reaction centers of photosynthetic bacteria. Springer-Verlag, Berlin, pp 199–208
Takahashi M, Ichimura S (1970) Photosynthetic properties and growth of photosynthetic sulfur bacteria in lakes. Limnol Oceanogr 15:929–944
van Niel CB (1944) The culture, general physiology, morphology and classification of the non-sulphur purple and brown bacteria. Bacteriol Rev 8:1–118
van Niel CB (1971) Techniques for the enrichment, isolation and maintenance of the photosynthetic bacteria. Methods Enzymol 23:3–28
Yurkov VV, Beatty JT (1998) Aerobic anoxygenic phototrophic bacteria. Microbiol Mol Biol Rev 62:695–724
Zhang D, Yang H, Huang Z, Zhang W, Liu S-J (2002) Rhodopseudomonas faecalis sp. nov., a phototrophic bacterium isolated from an anaerobic reactor that digests chicken faeces. Int J Syst Evol Microbiol 52:2055–2060
Acknowledgements
The author wishes to thank Prince Songkla University-Phuket for providing facilities for the project. The project was partially funded by the Faculty of Technology and Environmental Science, Prince Songkla University-Phuket. The co-operation of Phuket Integrated Waste Management (Wichit Sub-district, Mueang Phuket, District, Phuket 83000, Thailand) in encouraging this study and allowing us to collect sewage pond water samples is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
(PDF 151 kb)
Rights and permissions
About this article
Cite this article
Chandaravithoon, P., Nakphet, S. & Ritchie, R.J. Oxygenic and anoxygenic photosynthesis in a sewage pond. J Appl Phycol 30, 3089–3102 (2018). https://doi.org/10.1007/s10811-018-1432-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-018-1432-3