Abstract
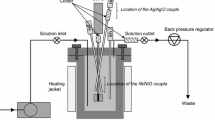
Electrochemical removal of sulfide ions was achieved in salt water using graphite anodes in an autoclave under high temperatures and pressures, simulating geothermal fluids. The reaction products were characterized using microscopy and X-ray photoelectron spectroscopy (XPS). At low temperatures the reaction rate is quite small. It decreases rapidly with time down to a negligibly small value, which increases only slightly with temperature. The reaction produces elemental sulfur, which was seen under the microscope and identified using XPS. It passivates the electrode and hence diminishes its activity. Above about 115 °C, much higher removal rates can be sustained for much longer times, while the increase of temperature has a much stronger effect on the reaction rate. Under this condition, elemental sulfur was no longer detected among the reaction products, while the electrode retained its activity for continuous operation. The XPS spectra at high temperatures reveal the presence of oxygen bearing sulfur species, such as sulfates. The melting of sulfur (at 115 °C) has a much stronger effect on the efficiency of the process than the transition of orthorhombic to monoclinic sulfur (at 95 °C). A Clausius-Clapeyron’s analysis reveals that the melting point of sulfur inside the autoclave is nearly equal to its normal melting point.






Similar content being viewed by others
References
U.S. Environmental Protection Agency (1988) Extremely hazardous substance: superfund chemical profiles, CAS registry number 7783-06-4, vol 1. Noys Data Corporation, New Jersey
Tuttle RN, Kane RD (eds) (1981) H2S corrosion in oil and gas production. National Association of Corrosion Engineers, Houston, Texas
Garverick L (ed) (1994) Corrosion in the petrochemical industry. ASM International, Metals Park, Ohio, p 259
Kagel A (2008) The state of geothermal technology, part II: surface technology. The Department of Energy, Washington, DC, p 5
Clauser C (2006) Geothermal energy, In Heinloth K (ed), Landolt-Börnstein, group VIII: Advanced materials and technologies, vol 3: energy technologies, subvol. C: renewable energies, Springer Verlag, Heidelberg-Berlin, 493–604, p 98
Xu Y, Shonnen MAA, Nordstram DK, Cunningham KM, Ball JW (2000) J Volcanol Geotherm Res 97:407
Cutter GA, Walsh RS, de Echols CS (1999) Deep-Sea Res II 46:991
Rajalo G, Petrovskaya T (1996) Environ Technol 17:605
Rao NN, Somasekhar KM, Kaul SN, Szpyrkowicz L (2001) J Chem Technol Biotechnol 76:1124
Szpyrkowicz L, Kaul SN, Neti RN (2005) J Appl Electrochem 35:381
Behm M, Simonsson D (1997) J Appl Electrochem 27:507
Garrett RL, Clark RK, Carney LL, Granthm CK (1997) Chemical scavengers for sulfides in water-based drilling fluids, SPE Reprint Series, 44:170
Singh AK, Kohil BS, Wendt RP (1989) World Oil, 209:99, 77
Al-Humaidan AY, Nasr-El-Din HA (1999) Optimization of hydrogen sulfide scavengers used during well stimulation, In: Proceedings of the SPE international symposium on oilfield chemistry
Schorling P Chr, Brauchle M (2001) Application of hydrogen sulfide scavengers in the oil and gas field, Erdoel Ergas Kohle/EKEP, 117:78
Scott P (1994) Oil Gas J 92:72
Weil ED, Sandler SR, (1991) Sulfur Compounds, In: Mary Howe-Grant (ed) Kirk-Othmer Encyclopedia of Chemical Technology, vol 23, 4th edn. Wiley, New York
Tatapundi P, Fenton JM (1995) Electrolytic processes for pollution treatment and pollution prevention. In: Gerischer H, Tobias CW (eds) Advances in electrochemical science and engineering, VCH, Weinheim, V 4 364
Lloyd CL, Gilbert II (1994) J. Electrochem. Soc. 141:2642
Pujare NU, Tsai KJ, Sammells AF (1989) J Electrochem Soc 136:3662
Schmidt DS, Winnick J (1998) AIchEJ 44:323
Anani A, Mao Z, White RE, Srinivasan S, Appleby AJ (1990) J Electrochem Soc 137:2703
Mao Z, Anani A, White RE, Srinivasan S, Appleby AJ (1991) J Electrochem Soc 138:1299
Ateya BG, AlKharafi FM, Alazab AS, Abdullah AM (2007) J Appl Electrochem 37:395
Briggs D, Seah MP (1990) Practical surface analysis. Wiley, New York, p 605
Gerson AR, Bredow T (2000) Surf Interface Anal 29:145
Ateya BG, AlKharafi FM, Abdullah RM, Alazab AS (2005) J Appl Electrochem 35:297
Zhdanow SI (1982) In Bard AJ (ed) Encyclopedia of the electrochemistry of the elements 6, Marcel Dekker Inc, New York
Bard AJ, Parson R, Jordan J (ed) (1985) Standard potentials in aqueous solutions, Marcel Dekker Inc., New York, p 94
Valensi G, van Muylder J, Pourbaix M (1974) In: Pourbaix M (ed) Atlas of electrochemical equilibria in aqueous media. NACE, Texas, p 545
Stefánsson A, Gunnarsson I (2005) Sulphur geochemistry of hydrothermal waters I. Report of preliminary study. Institute of Earth Sciences Report, RH-17
Kaasalainen H, Stefansson A (2008) The chemistry of sulfur in volcanic geothermal fluids Geochemica Et Cosmochimica Acta 72–12: A443–A443
Acknowledgments
The authors gratefully acknowledge the support of this work by the Research Administration of Kuwait University, under Grant numbers SC04/04 and GS01/01. They also acknowledge the help of the unit of Electron Microscopy for the SEM measurements.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ateya, B.G., Al Kharafi, F.M., El-Shamy, A.M. et al. Electrochemical desulfurization of geothermal fluids under high temperature and pressure. J Appl Electrochem 39, 383–389 (2009). https://doi.org/10.1007/s10800-008-9683-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10800-008-9683-3