Abstract
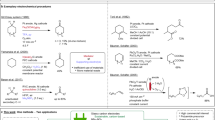
Oxidative cleavage of olefinic double bonds to carboxylic acids, aldehydes or ketones is one of the important reactions in organic synthesis. Ozonolysis is mostly applied for this purpose, however, high costs for safety precautions in technical scale conversions demand alternatives. For this purpose different electrochemical methods are investigated. In the direct oxidation of cyclohexene at a platinum or graphite anode no cleavage occurs, but substituted and rearranged products are obtained. At the boron doped diamond electrode (BDDE) aliphatic olefins with high oxidation potential are not converted. Electrochemical ozonolysis by oxidation of water to ozone at the lead dioxide electrode leads to carboxylic acids as cleavage products in high material yield but low current yield. Anodic bromo-formyloxylation followed by an anodic cleavage provides a two step conversion of cyclohexene to hexane-1,6-dial derivatives. Thereby anodic discharge of bromide in formic acid leads to (2-bromocyclohexyl)-formate, which is converted to cyclohexane-1,2-diol and the major part of potassium bromide is recovered for the next cycle. This electrochemical conversion appears to be an attractive alternative to chemical oxidations with oxygen and catalysts or with hydrogen peroxide. The diol is cleaved in high yield to hexane-1,6-dial or its acetal either directly or indirectly with periodate as mediator.
Similar content being viewed by others
References
Bailey P.S., (1978). Ozonization in Organic Chemistry. Academic Press, New York
Lee D.G. and Chen T., (1991). Comprehensive Organic Synthesis. Pergamon Press, Oxford, p. 574
Brown E., Deroye C. and Touet T., (1998). Tetrahedron 9:1605
Defacqz N., Touillaux R., Cordi A. and Marchand-Brynaert J., (2001). J. Chem Soc.Perkin Trans. 1:2632
Capon R.J., Jenkins A., Rooney F. and Ghisalberti E.L., (2001). J. Nat. Prod. 64:638
K. Weissermel and H.J. Arpe, ‘Industrielle Organische Chemie’, 5th ed., (VCH, 1998), p.172, 229.
Schober B.D., (1996). Chimica Oggi – Chemistry Today 13:21
C.F. Goebel, Emery Ind., US 2813113, Chem. Abstr. 52 (1958) 2431
Griffith W.P., Shoair A.G.and Suriaatmaja M., (2000). Synth. Commun. 30:3091
Kawatsura M., Uozumi Y., Ogasawara M. and Hayashi T., (2000). Tetrahedron 56:2247
Antonelli E., D’Aloisio R., Gambaro M., Fiorani T. and Venturello C., (1998). J. Org. Chem. 63:7190
Henry J.R. and Weinreb S.M., (1993). J.Org. Chem. 58:4745
Arney B.E., Jr., Wilcox E., Campbell M.O. and Gutierrez M.O., (1993). J. Org. Chem. 58:6126
Rossiter B.T., Katsuki T. and Sharpless K.B., (1981). J.Am.Chem. Soc. 103:464
Huang B., Khrapov M., Hansen K.C., Idoux J.P. and Gupton J.T., (1995). Synth. Commun. 25:2709
Carlsen P.H.J., Katsuki T., Martin V.S. and Sharpless K.B., (1981). J. Org. Chem. 46:3936
Sato K., Aoki M., Ogawa M., Hashimoto T., Panyella D. and Noyori R., (1997). Bull. Chem. Soc. Jpn. 70:905
Muryashi S.I., Suito T., Naota T., Kumobayashi H. and Akutagawa S., (1991). Tetrahedron Lett. 32:5991
K. Kaneda, S. Haruna, T. Imanaka and K. Kawamoto, J. Chem. Soc., Chem. Commun. (1990) 1467
Bäumer U.S. and Schäfer H.J., (2003). Electrochim. Acta 48:489
Wabner D.W., Fritz H.P., Missol D., Huß R. and Hindelang F., (1976). Z. Naturforsch. 31b:39
Gupton J.T. and Dale D.E., (1981). Synth. Commun. 11:571
Nikishin G.I., Elinson M.N. and Makhova I.V., (1988). Tetrahedron Lett. 29:1603
Neverov A.A. and Brown R.S., (1998). J. Org. Chem. 63:5977
Autorenkollektiv, ‘Organikum’, 18. Aufl., VEB Deutscher Verlag der Wissenschaften, Berlin (1990) p.258
Malanga C., Mannucci S. and Lardicci L., (1998). Tetrahedron 54:1021
Sharghi H., Niknam K. and Pooyan M., (2001). Tetrahedron 57:6057
Niizato H., Ueno Y. and Takemura S., (1972). Chem. Pharm. Bull. 20:2707
Iranpoor N., Firouzabadi H., Maryam C. and Jafari A.A., (2002). Tetrahedron 28:7037
Shono T., Matsumura Y., Hashimoto T., Hibino K., Hamaguchi H. and Aoki T. (1975). J.Am. Chem. Soc. 97:2546
Shono T., Hamaguchi H., Matsumura Y. and Yoshida K., (1977). Tetrahedron Lett. 41:3625
Griesbaum K., Jung I.C. and Mertens H., (1990). J. Org. Chem. 55:6024
Zhong Y.-L. and Shing T.K.M., (1997). J. Org. Chem. 62:2622
Griesbaum K. and Neumeister J., (1982). Chem. Ber.115:2697
Y.N. Ogibim, A.I. Ilovaisky and G.I. Nikishin, J. Org. Chem. 61 (1996) 3256; Electrochim. Acta. 42 (1997) 1933.
Torii S., Inokuchi T. and Oi R., (1982). J. Org. Chem. 47:47
Favier R., Freppel C., Richer J.-C. and Zador M., (1971). Can. J. Chem. 49:2590
A.R. Blake, J.G. Sunderland and A.T. Kuhn, J. Chem. Soc. (A). (1969) 3015.
M. Fleischmann, D. Pletcher and G.M.Race, J. Chem. Soc. (B). (1970)174.
E. Steckhan and C.Kandzia, Synlett (1992) 139.
Torii S., Uneyama K., Tanaka H., Yamanaka T., Yasuda T., Ono M. and Kohmoto Y., (1981). J. Org. Chem. 46:3312
Shen Y., Atobe M., Li W., and Nonaka T., (2003). Electrochim. Acta 48:1041
Torii S., Uneyama K., Ono M., Tazawa H. and Matsunami S., (1979). Tetrahedron Lett. 48:4661
Möller K.C. and Schäfer H.J., (1996). Electrochim. Acta 42:1971
Gandini D., Michaud P.A., Duo I., Maké E., Haenni W., Perret A. and Comminellis Ch., (1999). New Diamond Front. Technol. 9:303
H. Pütter, A. Weiper-Idelmann and C. Merck, DE 19911746 A1, Chem Abstr. 133 (2000) 244253.
H. Pütter and C. Merck, DE 10045664 A1, Chem Abstr. 136 (2002) 253952.
Ch. Reufer and Th.Lehmann, GDCh-Jahrestagung, Fachgruppe Angewandte Elektrochemie, München 2003, Poster
Imamura A. and Hirao K., (1979). Bull. Chem. Soc. Jpn. 52:287
Beer H.B., (1980). J. Electrochem. Soc. 127:303C
Trasatti S., (1990). Croat. Chem. Acta 63:313
Schäfer H.J., (1987). Top. Curr. Chem. 142:102
M. Gnann, D.W. Wabner and W. Tillmetz, DE 3432684, Chem. Abstr. 104 (1986) 195543
Pavlov D. and Monahov B., (1996). J. Electrochem. Soc. 143:3616
Monahov B. and Pavlov D., (1993). J. Appl. Electrochem. 23:1244
Wabner D.W. and Grambow C., (1985). J. Electroanal. Chem. 195:95
Stucki S., Theis G., Kötz R., Devantay H. and Christ H.J., (1985). J. Electrochem. Soc. 132:367
Stucki S., Baumann H., Christen H.J. and Kötz R., (1987). J. Appl. Electrochem. 17:773
Poullien P., Minko R., Verniette M. and Martinett P., (1980). Electrochim. Acta. 25:711
Shono T. and Ikeda A., (1972). J.Am.Chem.Soc. 94:7892
Dubois J.E., Garnier F. and Villard H., (1965). Tetrahedron Lett. 17:1277
Haines A.H. (1991). Comprehensive Organic Synthesis. Pergamon, Oxford, p. 437
D.E.J.E. deVos, S.M.A. de Wildeman and P.A. Jacobs, EP 0970951 A1, Chem. Abstr. 132 (2000) 64159.
M. Beller, C. Döbler and G. Mehltretter, DE 19920038 A1, Chem Abstr. 133 (2000) 323285.
Herrmann W.A., Fischer R.W. and Marz D.W., (1991). Angew. Chem. 30:1638
Org. Synth. Coll. Vol.III, 217 (1953)
(a) V.F. Pfeifer, V.E. Sohns, H.F. Conway, E.B. Lancaster, S. Dabic and E.L. Griffin, Ind. Eng. Chem. 52 (1960) 201.
Yoshiyama A., Nonaka T., Baizer M.M. and Chou T.C., (1985). Bull. Chem. Soc. Jpn. 58:201
Acknowledgements
This work was supported through the Arbeitsgemeinschaft Industrieller Forschungsvereinigungen (AIF-project No 13227N/1) by the Bundeswirtschaftsminister. We thank Dr. W. Haenni, CSEM for a BDD/Si-electrode, Metakem for a BDD/Ti-electrode and the BASF AG for samples of supporting electrolytes.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bäumer, US., Schäfer, H. Cleavage of alkenes by anodic oxidation. J Appl Electrochem 35, 1283–1292 (2005). https://doi.org/10.1007/s10800-005-9060-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10800-005-9060-4