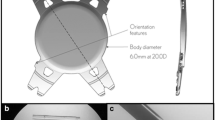
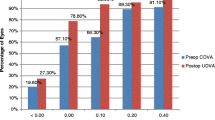
Abstract
Purpose
To evaluate clinical outcomes following sub-2-mm microincision cataract surgery (MICS) and intraocular lens (IOL) implantation.
Setting
Five EU clinical sites.
Design
Prospective, multicenter, open-label, single-arm, non-randomized.
Methods
Preoperative assessment involved visual acuity (VA), intraocular pressure and biometry measurements. 1.4-mm wound-assisted or 1.8-mm MICS was performed. Follow-up visits were made 1 day, 1–2 weeks, 1–2 and 4–6 months after surgery. The incision size, corrected distance VA (CDVA), uncorrected distance VA, manifest refraction spherical equivalent (MRSE), refraction predictability/stability and IOL decentration were assessed. At 12-, 18-, and 24-month, long-term centration, posterior capsular opacification (PCO) and Nd:YAG capsulotomy rates were investigated.
Results
A total of 103 eyes were implanted with the study IOL (INCISE, Bausch & Lomb), 96 of which were included in visual outcome analysis. A mean 6-month CDVA of − 0.02 logMAR (20/20 + 1) was observed and 75 eyes (79.8%) and 93 eyes (98.3%) achieved a visual acuity of at least 20/20 or 20/40. Mean MRSE was − 0.20 ± 0.60 D. Mean absolute predictive error was 0.44 ± 0.36 D, with 90.4% within 1.00 D of target. Mean total decentration was 0.35 ± 0.36 mm at 6 months and 0.32 ± 0.14 mm at 24 months (p > 0.05). 24-month evaluation of posterior capsular opacification score was 0.03 for the central area. A Nd:YAG rate of 3.4% was observed at 24 months.
Conclusions
The new MICS IOL provided excellent visual outcomes and was safe and effective for the sub-2-mm procedure. The MICS IOL demonstrated long-term centration, stability and a low rate of PCO development.




Similar content being viewed by others
References
Klonowski P, Rejdak R, Alió JL (2013) Microincision cataract surgery 1.8 mm incisional surgery. Expert Rev Ophthalmol 8(4):375–391
Yu JG, Zhao YE, Shi JL, Ye T, Jin N, Wang QM, Feng YF (2012) Biaxial microincision cataract surgery versus conventional coaxial cataract surgery: meta-analysis of randomized controlled trials. J Cataract Refract Surg 38:894–901
Agarwal A, Agarwal S, Agarwal A, Bagmar A, Patel N, Pandey SK, Shah SP (2001) Phakonit lens removal through a 0.9 mm incision [letter]. J Cataract Refract Surg 27:1531–1532 (reply by H Tsunoka, 1532–1533)
Tsuneoka H, Shiba T, Takahashi Y (2002) Ultrasonic phacoemulsification using a 1.4 mm incision: clinical results. J Cataract Refract Surg 28:81–86
Alio JL, Rodriguez Prats JL, Galal A (eds) (2004) MICS: micro-incision cataract surgery. Highlights of Ophthalmology International, El Dorado, Panama
Dick HB (2012) Controlled clinical trial comparing biaxial microincision with coaxial small incision for cataract surgery. Eur J Ophthalmol 22:739–750
Alio JL, Rodrıguez-Prats JL, Galal A, Ramzy M (2005) Outcomes of microincision cataract surgery versus coaxial phacoemulsification. Ophthalmology 112:1997–2003
Elkady B, Alio JL, Ortiz D, Montalban R (2008) Corneal aberrations after microincision cataract surgery. J Cataract Refract Surg 34:40–45
Hayashi K, Hayashi H, Nakao F, Hayashi F (1995) The correlation between incision size and corneal shape changes in sutureless cataract surgery. Ophthalmology 102:550–556
American National Standards Institute (2010) ANSI Z80.30—2010: ophthalmics—toric intraocular lenses. American National Standards Institute, New York, p 2010
International Organization for Standardization (2006) ISO 11979-7:2006(E) ophthalmic implants—intraocular lenses—Part 7: clinical investigations, 2006. International Organization for Standardization, Geneva
Lundström M, Barry P, Henry Y, Rosen P, Stenevi U (2012) Evidence-based guidelines for cataract surgery: guidelines based on data in the European Registry of Quality Outcomes for Cataract and Refractive Surgery database. J Cataract Refract Surg 38:1086–1093
ULIB User group for laser interference biometry. http://ocusoft.de/ulib/c1.htm. Accessed 22 March 2015
Klonowski P, Rejdak R, Alio JL (2013) Microincision cataract surgery: 1.8 mm incisional surgery. Expert Rev Ophthalmol 8:375–391
Wilczynski M, Supady E, Loba P, Synder A, Palenga-Pydyn D, Omulecki W (2009) Comparison of early corneal endothelial cell loss after coaxial phacoemulsification through 1.8 mm microincision and bimanual phacoemulsification through 1.7 mm microincision. J Cataract Refract Surg 35:1570–1574
Gale RP, Saldana M, Johnston RL, Zuberbuhler B, McKibbin M (2009) Benchmark standards for refractive outcomes after NHS cataract surgery. Eye (Lond) 23:149–152
Baumeister M, Buehren J, Kohnen T (2009) Tilt and decentration of spherical and aspheric intraocular lenses: effect on higher order aberrations. J Cataract Refract Surg 35:1006–1012
Buckhurst PJ, Woffsohn JS, Naroo SA, Davies LN (2010) Rotational and centration stability of an aspheric intraocular lens with a simulated toric design. J Cataract Refract Surg 36:1523–1528
Wolffsohn JS, Buckhurst PJ (2010) Objective analysis of toric intraocular lens rotation and centration. J Cataract Refract Surg 36:778–782
Crnej A, Hirnschall N, Nishi Y et al (2011) Impact of intraocular lens haptic design and orientation on decentration and tilt. J Cataract Refract Surg 37:1768–1774
Xing XJ, Tang X, Song H (2012) Comparison of optical performance and stability of five different kinds of aspheric IOLs. Zhonghua Yan Ke Za Zhi 48:297–301
Nishi O, Nishi K (1999) Preventing posterior capsule opacification by creating a discontinuous sharp bend in the capsule. J Cataract Refract Surg 25:521–526
Nishi O, Nishi K, Sakanishi K (1998) Inhibition of migrating lens epithelial cells at the capsular bend created by the rectangular optic edge of a posterior chamber intraocular lens. Ophthalmic Surg Lasers 29:587–594
Nishi O, Nishi K, Osakabe Y (2004) Effect of intraocular lenses on preventing posterior capsule opacification: design versus material. J Cataract Refract Surg 30:2170–2276
Buehl W, Findl O (2008) Effect of Intraocular lens design on posterior capsule opacification. J Cataract Refract Surg 34:1976–1985
Findl O, Buehl W, Bauer P, Sycha T (2010) Interventions for preventing posterior capsule opacification. Cochrane Collab 2010:1–81
Werner L, Müeller M, Tetz M (2008) Evaluating and defining the sharpness of intraocular lenses: microedge structure of commercially available suare-edged hydrophobic lenses. J Cataract Refract Surg 34:310–317
Werner L, Tetz M, Feldmann I, Bücker M (2009) Evaluating and defining the sharpness of intraocular lenses: microedge structure of commercially available square-edged hydrophilic intraocular lenses. J Cataract Refract Surg 35:556–566
Nanavaty MA, Spalton DJ, Boyce J, Brain A, Marshall J (2008) Edge profile of commercially available square-edged intraocular lenses. J Cataract Refract Surg 34:677–686
Roshdy MM, Riad RF, Morkos FF, Hassouna AK, Wahba SS (2013) Effect of a single-piece aspheric hydrophobic acrylic intraocular lens design on centration and rotation. J Cataract Refract Surg 39:408–413
Miyata K, Kataoka Y, Matsunaga J, Honbo M, Minami K (2015) Prospective comparison of one-piece and three-piece Tecnis aspheric intraocular lenses: 1-year stability and its effect on visual function. Curr Eye Res 40:930–935
Johansson B (2010) Clinical consequences of acrylic intraocular lens material and design: Nd:YAG-laser capsulotomy rates in 3 × 300 eyes 5 years after phacoemulsification. Br J Ophthalmol 94:450–455
Zacharopoulos IP, Papadaki T, Segev F, Jager-Roshu S, Assia E (2010) Two-year results of cataract extraction with implantation of the hydrophilic acrylic B-lens. Semin Ophthalmol 25:1–7
Gauthier L, Lafuma A, Laurendeau C, Berdeaux G (2010) Nd:YAG laser rates after bilateral implantation of hydrophobic or hydrophilic multifocal intraocular lenses: twenty-four month retrospective comparative study. J Cataract Refract Surg 36:1195–1200
Iwase T, Nishi Y, Oveson BC, Jo YJ (2011) Hydrophobic versus double-square-edged hydrophilic foldable acrylic intraocular lens: effect on posterior capsule opacification. J Cataract Refract Surg 36:1060–1068
Spyridaki PM, Hoeh H (2010) Comparison of four MICS intraocular lenses regarding their rates of neodymium:YAG laser capsulotomy. Klin Monatsbl Augenheilkd 227:208–214
Selvam S, Khan IJ, Craig EA (2011) Neodymium:YAG laser capsulotomy rate of microincision hydrophilic acrylic intraocular lens. J Cataract Refract Surg 37:2080–2081
Alió JL, Elkady B, Ortiz D (2011) Corneal optical quality following sub 1.8 mm micro-incision cataract surgery vs. 2.2 mm mini-incision coaxial phacoemulsification. Middle East Afr J Ophthalmol 17:94–99
Tong N, He JC, Lu F, Wang Q, Qu J, Zhao YE (2008) Changes in corneal wavefront aberrations in microincision and small-incision cataract surgery. J Cataract Refract Surg 34:2085–2090
Can I, Bayhan HA, Çelik H, Ceran BB (2012) Comparison of corneal aberrations after biaxial microincision and microcoaxial cataract surgeries: a prospective study. Curr Eye Res 37:18–24
Denoyer A, Denoyer L, Marotte D, Georget M, Pisella PJ (2008) Intraindividual comparative study of corneal and ocular wavefront aberrations after biaxial microincision versus coaxial small-incision cataract surgery. Br J Ophthalmol 92:1679–1684
Funding
This clinical research was initiated and financially supported by Bausch & Lomb. None of the authors have a financial or proprietary interest in any material or method mentioned in this paper. Dr Johansson, Prof Dick, Dr Morselli, Dr Toso received travel support for scientific meetings.
Author information
Authors and Affiliations
Contributions
The authors confirm that they have made a significant contribution to this study and have read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts of interest to disclose.
Ethical approval
All procedures performed in this study were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki Declaration and its later amendments.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Dick, H.B., Schultz, T., Lesieur, G. et al. Evaluation of clinical outcomes following implantation of a sub-2-mm hydrophilic acrylic MICS intraocular lens. Int Ophthalmol 39, 1043–1054 (2019). https://doi.org/10.1007/s10792-018-0905-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-018-0905-3