Abstract
Aim
To assess patient perceptions regarding medical photography and the use of smart devices, namely mobile phones and tablets for medical photography.
Methods
A questionnaire-based survey was conducted among 280 consecutive adult patients who presented to the oculoplastics clinic at a tertiary eye care centre. The responses were tabulated and analysed.
Results
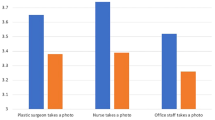
Of the 280 patients surveyed, 68% felt that medical photography had a positive impact on their understanding of their illnesses and 72% felt that the use of smartphones for medical photography was acceptable. Respondents below the age of 40 years were more likely to approve of the use of mobile phones for photography as compared to those over 40. Most patients (74%) preferred a doctor to be the person photographing them. While a majority approved of doctors and trainee physicians having access to their photographs, they felt non-physician healthcare personnel should not have access to clinical photographs. Also, 72% of the respondents felt that the patient’s consent should be taken before using their photographs. It was noted that patient identification and breach of confidentiality could be some of the potential issues with using smart devices as cameras in the clinic.
Conclusions
Clinical photography in general and, specifically, using smart devices for clinical photographs have gained acceptance among patients. The outcomes of this study may be utilized to create policy guidelines for the use of smart devices as photography tools in the clinics. The findings of this survey can also help to create standardized, uniform patient consent forms for clinical photography.
Similar content being viewed by others
References
Mukherjee B, Nair AG (2012) Principles and practice of external digital photography in ophthalmology. Indian J Ophthalmol 60:119–125
Nair AG, Santhanam A (2016) Clinical photography for periorbital and facial aesthetic practice. J Cutan Aesthet Dermat 9:115–121
Hsieh C, Yun D, Bhatia AC, Hsu JT, Ruiz de Luzuriaga AM (2015) Patient perception on the usage of smartphones for medical photography and for reference in dermatology. Dermatol Surg 41:149–154
Khanna V, Sambandam SN, Gul A, Mounasamy V (2015) “WhatsApp”ening in orthopedic care: a concise report from a 300-bedded tertiary care teaching center. Eur J Orthop Surg Traumatol 25:821–826
Adeyemo WL, Mofikoya BO, Akadiri OA, James O, Fashina AA (2013) Acceptance and perception of Nigerian patients to medical photography. Dev World Bioeth 13:105–110
Hacard F, Maruani A, Delaplace M, Caille A (2013) Patients’ acceptance of medical photography in a French adult and pediatric dermatology department: a questionnaire survey. Br J Dermatol 169:298–305
Lau CK, Schumacher HHA, Irwin MS (2010) Patients’ perception of medical photography. J Plast Reconstr Aesthet Surg 63:e507–e511
Leger MC, Wu T, Haimovic A, Kaplan R, Sanchez M, Cohen D, Leger EA, Stein JA (2014) Patient perspectives on medical photography in dermatology. Dermatol Surg 40:1028–1037
Natarajan S, Nair AG (2015) Outsmarted by the smartphone! Indian J Ophthalmol 63:757–758
Chan N, Charette J, Dumestre DO, Fraulin FO (2016) Should ‘smart phones’ be used for patient photography? Plast Surg (Oakv) 24:32–34
Stevenson P, Finnane AR, Soyer HP (2016) Teledermatology and clinical photography: safeguarding patient privacy and mitigating medico-legal risk. Med J Aust 204:198–200e1
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None of the authors have any conflicting interests pertaining to this manuscript to disclose.
Human and animals participants
No intervention was performed on human participants or animals in this project. This study was carried out in line with the Declaration of Helsinki and was approved by the Ethics Research Committee.
Informed consent
All subjects having participated in the study provided written informed consent prior to participation in the questionnaire.
Rights and permissions
About this article
Cite this article
Nair, A.G., Potdar, N.A., Dadia, S. et al. Patient perceptions regarding the use of smart devices for medical photography: results of a patient-based survey. Int Ophthalmol 39, 783–789 (2019). https://doi.org/10.1007/s10792-018-0878-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-018-0878-2