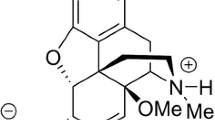
Abstract
Purpose
There are several lines of evidence on the protective roles of opioids in gastrointestinal inflammatory conditions. This study aims to distinguish the central and peripheral roles of methadone, a non-selective opioid receptor agonist, in an acute model of ulcerative colitis in male rats.
Methods
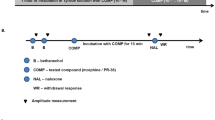
Ulcerative colitis was induced by intrarectal administration of acetic acid 4%. Methadone was injected subcutaneously (s.c.), 5 and 10 mg/kg, and intracerebroventricular (i.c.v.), 50 and 300 ng/rat. Opioid antagonists were employed. Methylnaltrexone (MNTX; 5 mg/kg, i.p.), a peripherally acting opioid receptor antagonist, and naltrexone (NTX; 5 mg/kg, i.p. and 10 ng/rat, i.c.v.), a peripherally and centrally acting opioid receptor antagonist were injected before methadone (10 mg/kg, s.c. and or 300 ng/rat, i.c.v.) administration. NTX (5 mg/kg, i.p. and 10 ng/rat, i.c.v.) were administered 30 min prior to administration of methadone (10 mg/kg, s.c. and 300 ng/rat, i.c.v.), respectively. MNTX (5 mg/kg, i.p.) was injected 30 min prior to methadone (10 mg/kg, s.c.). Seventy-two hours following colitis induction, macroscopic and microscopic mucosal lesions, and the colonic levels of tumor necrosis factor-alpha (TNF-α) and interleukin-1β (IL-1β) were determined.
Results
Methadone (300 ng/rat, i.c.v.) and Methadone (5 and 10 mg/kg, s.c.) improved the macroscopic and microscopic scores through opioid receptors. Also, a significant reduction in TNF-α and IL-1β was observed. Peripherally and centrally injected NTX significantly reversed methadone 10 mg/kg s.c. anti-inflammatory effects while MNTX could not completely reverse this effect. Moreover, centrally administered methadone (300 ng/rat) showed the anti-inflammatory effect which was reversed by central administration of NTX (10 ng/rat).
Conclusions
The opioid receptors mainly the central opioid receptors may mediate the protective actions of methadone on the experimental model of inflammatory bowel disease in rat.








Similar content being viewed by others
References
Al-Hashimi M, Scott SW, Thompson JP, Lambert DG (2013) Opioids and immune modulation: more questions than answers. Br J Anaesth 111:80–88
Altschuler SM, Escardo J, Lynn RB, Miselis RR (1993) The central organization of the vagus nerve innervating the colon of the rat. Gastroenterology 104:502–509
Amirshahrokhi K, Dehpour AR, Hadjati J, Sotoudeh M, Ghazi-Khansari M (2008) Methadone ameliorates multiple-low-dose streptozotocin-induced type 1 diabetes in mice. Toxicol Appl Pharmacol 232:119–124
Antonioli L, Fornai M, Colucci R, Ghisu N, Da Settimo F, Natale G, Kastsiuchenka O et al (2007) Inhibition of adenosine deaminase attenuates inflammation in experimental colitis. J Pharmacol Exp Ther 322(2):435–442
Baumgart DC, Sandborn WJ (2007) Inflammatory bowel disease: clinical aspects and established and evolving therapies. The Lancet 369:1641–1657
Bercik P, Verdu EF, Foster JA, Macri J, Potter M, Huang X, Malinowski P et al (2010) Chronic gastrointestinal inflammation induces anxiety-like behavior and alters central nervous system biochemistry in mice. Gastroenterology 139:2102–2112
Bidlack JM (2000) Detection and function of opioid receptors on cells from the immune system. Clin Diagn Lab Immunol 7(5):719–723
Bonaz B (2007) The cholinergic anti-inflammatory pathway and the gastrointestinal tract. Gastroenterology 133:1370–1373
Bonaz BL, Bernstein CN (2013) Brain–gut interactions in inflammatory bowel disease. Gastroenterology 144:36–49
Bonaz B, Sinniger V, Pellissier S (2017) Vagus nerve stimulation: a new promising therapeutic tool in inflammatory bowel disease. J interna med 282(1):46–63
Borron SW, Monier C, Risede P, Baud FJ (2002) Flunitrazepam variably alters morphine, buprenorphine, and methadone lethality in the rat. Hum Exp Toxico 21(11):599–605
Brown DR, Goldberg LI (1985) The use of quaternary narcotic antagonists in opiate research. Neuropharmacology 24(3):181–191
Bryant HU, Bernton EW, Holaday JW (1987) Immunosuppressive effects of chronic morphine treatment in mice. Life Sci 41:1731–1738
Burton MB, Gebhart GF (1998) Effects of kappa-opioid receptor agonists on responses to colorectal distension in rats with and without acute colonic inflammation. J Pharmacol Exp Ther 285:707–715
Chen W, Taché Y, Marvizón JC (2018) Corticotropin-releasing factor in the brain and blocking spinal descending signals induce hyperalgesia in the latent sensitization model of chronic pain. Neuroscience 381:149–158
Chugh SS, Socoteanu C, Reinier K, Waltz J, Jui J, Gunson K (2008) A community-based evaluation of sudden death associated with therapeutic levels of methadone. Am J Med 121:66–71
Collins S, Verma-Gandhu M (2006) The putative role of endogenous and exogenous opiates in inflammatory bowel disease. Gut 55:756–757
Daneshmand A, Rahimian R, Mohammadi H, Ejtemaee-Mehr S, Tavangar SM, Kelishomi RB, Dehpour AR (2009) Protective effects of lithium on acetic acid-induced colitis in rats. Dig Dis Sci 54:1901–1907
Dieleman LA, Palmen MJ, Akol H, Bloemena E, Peña AS, Meuwissen SG, Van Rees EP (1998) Chronic experimental colitis induced by dextran sulphate sodium (DSS) is characterized by Th1 and Th2 cytokines. Clin Exp Immunol 114:385–391
Earl JR, Claxson AW, Blake DR, Morris CJ (1994) Proinflammatory effects of morphine in the rat adjuvant arthritis model. Int J Tissue React 16:163–170
Eisenstein TK, Hilburger ME (1998) Opioid modulation of immune responses: effects on phagocyte and lymphoid cell populations. J Neuroimmunol 83:36–44
Elson CO, Sartor RB, Tennyson GS, Riddell H (1995) Experimental models of inflammatory bowel disease. Gastroenterology 109:1344–1367
Fakhraei N, Abdolghaffari AH, Delfan B, Abbasi A, Rahimi N, Khansari A, Rahimian R, Dehpour AR (2014) Protective effect of hydroalcoholic olive leaf extract on experimental model of colitis in rat: involvement of nitrergic and opioidergic systems. Phytother Res 28:1367–1373
Fecho K, Maslonek KA, Dykstra LA, Lysle DT (1996) Assessment of the involvement of central nervous system and peripheral opioid receptors in the immunomodulatory effects of acute morphine treatment in rats. J Pharmacol Exp Ther 276:626–636
Greenwood-Van Meerveld B, Johnson AC, Schulkin J, Myers DA (2006) Long-term expression of corticotropin-releasing factor (CRF) in the paraventricular nucleus of the hypothalamus in response to an acute colonic inflammation. Brain Res 1071:91–96
Grimm MC, Ben-Baruch A, Taub DD, Howard OM, Wang JM, Oppenheim JJ (1998) Opiate inhibition of chemokine-induced chemotaxis. Ann N Y Acad Sci 840:9–20
Gyires K, Budavári I, Fürst S, Molnár I (1985) Morphine inhibits the carrageenan-induced oedema and the chemoluminescence of leucocytes stimulated by zymosan. J Pharm Pharmacol 37:100–104
Hanauer SB (2007) The benefits of loperamide in the treatment of patients with IBS or IBD. Introduction. Rev gastroenterol disord 7:S1
Hashemizadeh S, Sardari M, Rezayof A (2014) Basolateral amygdala CB1 cannabinoid receptors mediate nicotine-induced place preference. Prog Neuropsychopharmacol Biol Psychiatry 51:65–71
Heydarpour P, Rahimian R, Fakhfouri G, Khoshkish S, Fakhraei N, Salehi-Sadaghiani M, Wang H, Abbasi A, Dehpour AR, Ghia JE (2016) Behavioral despair associated with a mouse model of Crohn’s disease: role of nitric oxide pathway. Prog Neuropsychopharmacol Biol Psychiatry 64:131–141
Hua S (2014) Development of an effective topical liposomal formulation for localized analgesia and anti-inflammatory actions in the complete Freund’s adjuvant rodent model of acute inflammatory pain. Pain Physician 17:E719–E735
Janecka A, Fichna J, Janecki T (2004) Opioid receptors and their ligands. Curr Top Med Chem 4:1–17
Kafami L, Etesami I, Felfeli M, Enayati N, Ghiaghi R, Aminian A, Dehpour A (2013) Methadone diminishes neuroinflammation and disease severity in EAE through modulating T cell function. J Neuroimmunol 255:39–44
Khalifeh S, Fakhfouri G, Mehr SE, Mousavizadeh K, Dehpour AR, Khodagholi F, Kazmi S, Rahimian R (2015) Beyond the 5-HT3 receptors: a role for α7nACh receptors in neuroprotective aspects of tropisetron. Hum Exp Toxicol 34:922–931
Levine JD, Moskowitz MA, Basbaum AI (1985) The contribution of neurogenic inflammation in experimental arthritis. J Immunol 135(2 Suppl):843s–847s
Liu SJ, Roerig DL, Wang RI (1983) Brain and plasma levels of methadone and their relationships to analgesic activity of methadone in rats. Drug Metab Dispos 11:335–338
Lysle DT, Coussons-Read ME (1995) Mechanisms of conditioned immunomodulation. Int J Immunopharmacol 17:641–647
Mackner LM, Clough-Paabo E, Pajer K, Lourie A, Crandall WV (2011) Psychoneuroimmunologic factors in inflammatory bowel disease. Inflamm Bowel Dis 17:849–857
Mahgoub A, El-Medany A, Mustafa A, Arafah M, Moursi M (2005) Azithromycin and erythromycin ameliorate the extent of colonic damage induced by acetic acid in rats. Toxicol Appl Pharmacol 205:43–52
McCarthy L, Wetzel M, Sliker JK, Eisenstein TK, Rogers TJ (2001) Opioids, opioid receptors, and the immune response. Drug Alcohol Depend 62:111–123
Medany A, Mahgoub A, Mustafa A, Arafa M, Morsi M (2005) The effects of selective cyclooxygenase-2 inhibitors, celcoxib and rofecoxib, on experimental colitis induced by acetic acid in rats. Eur J Pharmacol 507:291–295
Meregnani J, Clarençon D, Vivier M, Peinnequin A, Mouret C, Sinniger V, Picq C, Job A, Canini F, Jacquier-Sarlin M, Bonaz B (2011) Anti-inflammatory effect of vagus nerve stimulation in a rat model of inflammatory bowel disease. Auton Neurosci 160:82–89
Morris DD, Moore JN (1989) Antibody titres to core lipopolysaccharides in horses with gastrointestinal disorders which cause colic. Equine Vet J Suppl7:29–32
Mousavizadeh K, Rahimian R, Fakhfouri G, Aslani FS, Ghafourifar P (2009) Anti-inflammatory effects of 5-HT3 receptor antagonist, tropisetron on experimental colitis in rats. Eur J Clin Invest 39:375–383
Owczarek D, Cibor D, Mach T, Cieśla A, Pierzchała-Koziec K, Sałapa K, Kuśnierz-Cabała B (2011) Met-enkephalins in patients with inflammatory bowel diseases. Adv Med Sci 56:158–164
Pakkanen JS, Nousiainen H, Yli-Kauhaluoma J, Kylänlahti I, Möykkynen T, Korpi ER, Peng JH, Lukas RJ, Ahtee L, Tuominen RK (2005) Methadone increases intracellular calcium in SH-SY5Y and SH-EP1-hα7 cells by activating neuronal nicotinic acetylcholine receptors. J Neurochem 94:1329–1341
Pavlov VA, Parrish WR, Rosas-Ballina M, Ochani M, Puerta M, Ochani K, Chavan S, Al-Abed Y, Tracey KJ (2009) Brain acetylcholinesterase activity controls systemic cytokine levels through the cholinergic anti-inflammatory pathway. Brain Behav Immun 23:41–45
Paxinos G, Watson C (2007) The rat brain in stereotaxic coordinates. Academic Press, San Diego
Perez-Navarro R, Ballester I, Zarzuelo A, S¢anchez de Medina F (2005) Disturbances in epithelial ionic secretion in different experimental models of colitis. Life Sci 76:1489–1501
Peterson PK, Molitor TW, Chao CC (1998) The opioid–cytokine connection. J Neuroimmunol 83:63–69
Philippe D, Dubuquoy L, Groux H, Brun V, Chuoï-Mariot MT, Gaveriaux-Ruff C, Colombel JF, Kieffer BL, Desreumaux P (2003) Anti-inflammatory properties of the μ opioid receptor support its use in the treatment of colon inflammation. J Clin Invest 111:1329–1338
Pol O, Puig MM (2004) Expression of opioid receptors during peripheral inflammation. Curr Top Med Chem 4:51–61
Pol O, Ferrer I, Puig MM (1994) Diarrhea associated with intestinal inflammation increases the potency of mu and delta opioids on the inhibition of gastrointestinal transit in mice. J Pharmacol Exp Ther 270:386–391
Porcher C, Sinniger V, Juhem A, Mouchet P, Bonaz B (2004) Neuronal activity and CRF receptor gene transcription in the brains of rats with colitis. Am J Physiol Gastrointest Liver Physiol 287:G803–G814
Rahimian R, Zirak MR, Keshavarz M, Fakhraei N, Mohammadi-Farani A, Hamdi H, Mousavizadeh K (2016) Involvement of PPARγ in the protective action of tropisetron in an experimental model of ulcerative colitis. Immunopharmacol Immunotoxicol 38:432–440
Ricardo Buenaventura M, Rajive Adlaka M, Nalini Sehgal M (2008) Opioid complications and side effects. Pain Physician 11:S105–S120
Rogers TJ, Peterson PK (2003) Opioid G protein-coupled receptors: signals at the crossroads of inflammation. Trends Immunol 24:116–121
Sengupta JN, Snider A, Su X, Gebhart GF (1999) Effects of kappa opioids in the inflamed rat colon. Pain 79:175–185
Seyedabadi M, Rahimian R, Ghia JE (2018) The role of alpha7 nicotinic acetylcholine receptors in inflammatory bowel disease: involvement of different cellular pathways. Expert Opin Ther Targets 22:161–176
Shahabi NA, Burtness MZ, Sharp BM (1991) N-acetyl-β-endorphin1–31 antagonizes the suppressive effect of β-endorphin1–31 on murine splenocyte proliferation via a naloxone-resistant receptor. Biochem Biophys Res Commun 175:936–942
Shannon HE, Lutz EA (2002) Comparison of the peripheral and central effects of the opioid agonists loperamide and morphine in the formalin test in rats. Neuropharmacology 42:253–261
Smith JP, Bingaman SI, Ruggiero F, Mauger DT, Mukherjee A, McGovern CO, Zagon IS (2011) Therapy with the opioid antagonist naltrexone promotes mucosal healing in active Crohn’s disease: a randomized placebo-controlled trial. Dig Dis Sci 56:2088–2097
Sternberg EM (2006) Neural regulation of innate immunity: a coordinated nonspecific host response to pathogens. Nat Rev Immunol 6:318
Straub RH, Wolff C, Fassold A, Hofbauer R, Chover-Gonzalez A, Richards LJ, Jessop DS (2008) Antiinflammatory role of endomorphins in osteoarthritis, rheumatoid arthritis, and adjuvant-induced polyarthritis. Arthritis Rheum 58:456–466
Taché Y, Bernstein CN (2009) Evidence for the role of the brain–gut axis in inflammatory bowel disease: depression as cause and effect? Gastroenterology 136:2058
Walker JS (2003) Anti-Inflammatory effects of opioids. In: Machelska H, Stein C (eds) Immune mechanisms of pain and analgesia. ©Eurekah.com and Kluwer Academic/Plenum Publishers, Dordrecht
Walker JS, Chandler AK, Wilson JL, Binder W, Day RO (1996) Effect of μ-opioids morphine and buprenorphine on the development of adjuvant arthritis in rats. Inflamm Res 45:557–563
Wei G, Moss J, Yuan CS (2003) Opioid-induced immunosuppression: is it centrally mediated or peripherally mediated? Biochem Pharmacol 65:1761–1766
Willette RE, Barnett G (1981) Narcotic antagonists: naltrexone pharmacochemistry and sustained-release preparations. Department of Health and Human Services, Public Health Service, Alcohol, Drug Abuse, and Mental Health Administration, National Institute on Drug Abuse, Division of Research
Yamada T, Zimmerman BJ, Specian RD, Grisham MB (1991) Role of neutrophils in acetic acid-induced colitis in rats. Inflammation 15:399–411
Zagon IS, McLaughlin PJ (2011) Targeting opioid signaling in Crohn’s disease: new therapeutic pathways. Expert Rev Gastroenterol Hepatol 5:555–558
Acknowledgements
The study was supported by a Grant from Tehran University of Medical Sciences, Tehran, Iran (No. 16555-15-04-90).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Fakhraei, N., Javadian, N., Rahimian, R. et al. Involvement of central opioid receptors in protective effects of methadone on experimental colitis in rats. Inflammopharmacol 26, 1399–1413 (2018). https://doi.org/10.1007/s10787-018-0538-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-018-0538-1