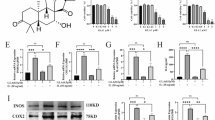
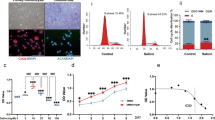
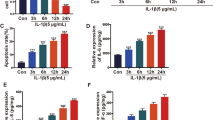
Abstract
Triptolide has been widely reported to exhibit potential therapeutic value in multiple inflammatory diseases, such as rheumatoid arthritis, systemic lupus erythematosus, and psoriasis. Although its safety and efficacy as an anti-inflammatory agent have been verified by many studies, the effect of triptolide on osteoarthritis (OA) was not clearly understood. In this study, we found that triptolide prevented OA development in a surgical destabilization of the medial meniscus (DMM) mouse model. In addition, triptolide inhibited both DMM-induced and LPS-induced expression of pro-inflammatory cytokines in the human monocytic cell line THP-1. Further mechanistic studies showed that the reduction of pro-inflammatory cytokines by triptolide was mediated by the upregulation of nucleotide-binding oligomerization domain-like receptor family pyrin domain-containing protein 3 (NLRP3) and downregulation of caspase-1. Finally, we identified that hsa-miR-20b, a microRNA targeting the NLRP3 gene, was downregulated by triptolide. This study provides a novel insight into the effect on triptolide in preventing OA pathogenesis.






Similar content being viewed by others
References
Abderrazak A, Syrovets T, Couchie D, El Hadri K, Friguet B, Simmet T, Rouis M (2015) NLRP3 inflammasome: from a danger signal sensor to a regulatory node of oxidative stress and inflammatory diseases. Redox Biol 4:296–307. https://doi.org/10.1016/j.redox.2015.01.008
Al-Khazraji BK, Appleton CT, Beier F, Birmingham TB, Shoemaker JK (2018) Osteoarthritis, cerebrovascular dysfunction and the common denominator of inflammation: a narrative review. Osteoarthr Cartil 26(4):462–470. https://doi.org/10.1016/j.joca.2018.01.011
Chang DM, Chang WY, Kuo SY, Chang ML (1997) The effects of traditional antirheumatic herbal medicines on immune response cells. J Rheumatol 24:436–441
Chen X, Murakami T, Oppenheim JJ, Howard OM (2005) Triptolide, a constituent of immunosuppressive Chinese herbal medicine, is a potent suppressor of dendritic-cell maturation and trafficking. Blood 106:2409–2416. https://doi.org/10.1182/blood-2005-03-0854
Denoble AE et al (2011) Uric acid is a danger signal of increasing risk for osteoarthritis through inflammasome activation. Proc Natl Acad Sci USA 108:2088–2093. https://doi.org/10.1073/pnas.1012743108
Fan D et al (2016) Triptolide modulates TREM-1 signal pathway to inhibit the inflammatory response in rheumatoid arthritis. Int J Mol Sci 17:498. https://doi.org/10.3390/ijms17040498
Farber JM (2018) The knee, osteoarthritis and biomarkers. Osteoarthr Cartil. https://doi.org/10.1016/j.joca.2018.01.023
Glasson SS, Blanchet TJ, Morris EA (2007) The surgical destabilization of the medial meniscus (DMM) model of osteoarthritis in the 129/SvEv mouse. Osteoarthr Cartil 15:1061–1069. https://doi.org/10.1016/j.joca.2007.03.006
Groslambert M, Py BF (2018) Regulation of the NLRP3 inflammasome. Med Sci M/S 34:47–53. https://doi.org/10.1051/medsci/20183401013
Han R, Rostami-Yazdi M, Gerdes S, Mrowietz U (2012) Triptolide in the treatment of psoriasis and other immune-mediated inflammatory diseases. Br J Clin Pharmacol 74:424–436. https://doi.org/10.1111/j.1365-2125.2012.04221.x
Hang LH, Li SN, Shao DH, Chen Z, Chen YF, Shu WW (2014) Evidence for involvement of spinal RANTES in the antinociceptive effects of triptolide, a diterpene triepoxide, in a rat model of bone cancer pain. Basic Clin Pharmacol Toxicol 115:477–480. https://doi.org/10.1111/bcpt.12265
Hu H, Luo L, Liu F, Zou D, Zhu S, Tan B, Chen T (2016) Anti-cancer and sensibilisation effect of triptolide on human epithelial ovarian cancer. J Cancer 7:2093–2099. https://doi.org/10.7150/jca.16178
Jiang D, Chen S, Sun R, Zhang X, Wang D (2018) The NLRP3 inflammasome: role in metabolic disorders and regulation by metabolic pathways. Cancer Lett 419:8–19. https://doi.org/10.1016/j.canlet.2018.01.034
Khella HWZ et al (2017) miR-10b is a prognostic marker in clear cell renal cell carcinoma. J Clin Pathol 70:854–859. https://doi.org/10.1136/jclinpath-2017-204341
Kim JA et al (2015) Inhibitory effects of triptolide on titanium particle-induced osteolysis and receptor activator of nuclear factor-kappaB ligand-mediated osteoclast differentiation. Int Orthop 39:173–182. https://doi.org/10.1007/s00264-014-2596-3
Kim JW, Roh YS, Jeong H, Yi HK, Lee MH, Lim CW, Kim B (2018) Spliceosome-associated protein 130 exacerbates alcohol-induced liver injury by inducing NLRP3 inflammasome-mediated IL-1β in mice. Am J Pathol 188(4):967–980. https://doi.org/10.1016/j.ajpath.2017.12.010
Kong X et al (2013) Anti-angiogenic effect of triptolide in rheumatoid arthritis by targeting angiogenic cascade. PLoS One 8:e77513. https://doi.org/10.1371/journal.pone.0077513
Li Y, Yu C, Zhu WM, Xie Y, Qi X, Li N, Li JS (2010) Triptolide ameliorates IL-10-deficient mice colitis by mechanisms involving suppression of IL-6/STAT3 signaling pathway and down-regulation of IL-17. Mol Immunol 47:2467–2474. https://doi.org/10.1016/j.molimm.2010.06.007
Li X et al (2016) Triptolide reduces proliferation and enhances apoptosis of human non-small cell lung cancer cells through PTEN by targeting miR-21. Mol Med Rep 13:2763–2768. https://doi.org/10.3892/mmr.2016.4844
Liacini A, Sylvester J, Zafarullah M (2005) Triptolide suppresses proinflammatory cytokine-induced matrix metalloproteinase and aggrecanase-1 gene expression in chondrocytes. Biochem Biophys Res Commun 327:320–327. https://doi.org/10.1016/j.bbrc.2004.12.020
Liu J, Wu QL, Feng YH, Wang YF, Li XY, Zuo JP (2005) Triptolide suppresses CD80 and CD86 expressions and IL-12 production in THP-1 cells. Acta Pharmacol Sin 26:223–227. https://doi.org/10.1111/j.1745-7254.2005.00035.x
Liu C et al (2013a) Triptolide prevents bone destruction in the collagen-induced arthritis model of rheumatoid arthritis by targeting RANKL/RANK/OPG signal pathway. Evid Based Complement Alternat Med 2013:626038. https://doi.org/10.1155/2013/626038
Liu W et al (2013b) A novel benzo[d]imidazole derivate prevents the development of dextran sulfate sodium-induced murine experimental colitis via inhibition of NLRP3 inflammasome. Biochem Pharmacol 85:1504–1512. https://doi.org/10.1016/j.bcp.2013.03.008
Liu YP, Li J, Xin SB (2018) Study the relevance between inflammatory factors and estradiol and their association with knee osteoarthritis in postmenopausal women. Eur Rev Med Pharmacol Sci 22:472–478. https://doi.org/10.26355/eurrev_201801_14197
Liu-Bryan R (2013) Synovium and the innate inflammatory network in osteoarthritis progression. Curr Rheumatol Rep 15:323. https://doi.org/10.1007/s11926-013-0323-5
Maddur MS, Vani J, Hegde P, Lacroix-Desmazes S, Kaveri SV, Bayry J (2011) Inhibition of differentiation, amplification, and function of human TH17 cells by intravenous immunoglobulin. J Allergy Clin Immunol 127(823–830):e821–e827. https://doi.org/10.1016/j.jaci.2010.12.1102
Massy-Westropp N, Krishnan J, Ahern M (2004) Comparing the AUSCAN osteoarthritis hand index michigan hand outcomes questionnaire, and sequential occupational dexterity assessment for patients with rheumatoid arthritis. J Rheumatol 31:1996–2001
Oo WM et al (2018) Clinimetrics of ultrasound pathologies in osteoarthritis: systematic literature review and meta-analysis. Osteoarthr Cartil. https://doi.org/10.1016/j.joca.2018.01.021
Orlowsky EW, Kraus VB (2015) The role of innate immunity in osteoarthritis: when our first line of defense goes on the offensive. J Rheumatol 42:363–371. https://doi.org/10.3899/jrheum.140382
Premkumar V, Dey M, Dorn R, Raskin I (2010) MyD88-dependent and independent pathways of toll-like receptors are engaged in biological activity of triptolide in ligand-stimulated macrophages. BMC Chem Biol 10:3. https://doi.org/10.1186/1472-6769-10-3
Radstake TR et al (2004) Expression of toll-like receptors 2 and 4 in rheumatoid synovial tissue and regulation by proinflammatory cytokines interleukin-12 and interleukin-18 via interferon-gamma. Arthritis Rheum 50:3856–3865. https://doi.org/10.1002/art.20678
Ravi B et al (2012) A systematic review and meta-analysis comparing complications following total joint arthroplasty for rheumatoid arthritis versus for osteoarthritis. Arthritis Rheum 64:3839–3849. https://doi.org/10.1002/art.37690
Riddle DL, Golladay GJ (2018) Pre-operative risk factors for postoperative falls in persons with hip or knee arthroplasty: a longitudinal study of data from the osteoarthritis initiative. Arch Phys Med Rehabil. https://doi.org/10.1016/j.apmr.2017.12.030
Scanzello CR, Plaas A, Crow MK (2008) Innate immune system activation in osteoarthritis: is osteoarthritis a chronic wound? Curr Opin Rheumatol 20:565–572. https://doi.org/10.1097/BOR.0b013e32830aba34
Stelzner K et al (2016) Free fatty acids sensitize dendritic cells to amplify TH1/TH17-immune responses. Eur J Immunol 46:2043–2053. https://doi.org/10.1002/eji.201546263
Takakubo Y, Barreto G, Konttinen YT, Oki H, Takagi M (2014) Role of innate immune sensors, TLRs, and NALP3 in rheumatoid arthritis and osteoarthritis. J Long Term Eff Med Implants 24:243–251
Wang Y, Mei Y, Feng D, Xu L (2008) Triptolide modulates T-cell inflammatory responses and ameliorates experimental autoimmune encephalomyelitis. J Neurosci Res 86:2441–2449. https://doi.org/10.1002/jnr.21683
Wang X, Zhang L, Duan W, Liu B, Gong P, Ding Y, Wu X (2014) Anti-inflammatory effects of triptolide by inhibiting the NF-kappaB signalling pathway in LPS-induced acute lung injury in a murine model. Mol Med Rep 10:447–452. https://doi.org/10.3892/mmr.2014.2191
Wei D, Huang Z (2014) Anti-inflammatory effects of triptolide in LPS-induced acute lung injury in mice. Inflammation 37:1307–1316. https://doi.org/10.1007/s10753-014-9858-5
Wu L et al (2013) Efficacy of triptolide for children with moderately severe Henoch-Schonlein purpura nephritis presenting with nephrotic range proteinuria: a prospective and controlled study in China. Biomed Res Int 2013:292865. https://doi.org/10.1155/2013/292865
Yamasaki K et al (2009) NLRP3/cryopyrin is necessary for interleukin-1beta (IL-1beta) release in response to hyaluronan, an endogenous trigger of inflammation in response to injury. J Biol Chem 284:12762–12771. https://doi.org/10.1074/jbc.M806084200
Yu C et al (2011) Triptolide ameliorates Crohn’s colitis is associated with inhibition of TLRs/NF-kappaB signaling pathway. Fitoterapia 82:709–715. https://doi.org/10.1016/j.fitote.2011.02.011
Zeng R, Zeng L, Chen Y, Zhao F, Li R, Wen L, Zhang C (2011) Triptolide-induced apoptosis by inactivating nuclear factor-kappa B apoptotic pathway in multiple myeloma in vitro. J Huazhong Univ Sci Technol Med Sci31:446–451 https://doi.org/10.1007/s11596-011-0471-7
Zheng CX, Chen ZH, Zeng CH, Qin WS, Li LS, Liu ZH (2008) Triptolide protects podocytes from puromycin aminonucleoside induced injury in vivo and in vitro. Kidney Int 74:596–612. https://doi.org/10.1038/ki.2008.203
Zheng L et al (2017) Triptolide-assisted phosphorylation of p53 suppresses inflammation-induced NF-kB survival pathways in cancer cells. Mol Cell Biol 37(15):e00149–17. https://doi.org/10.1128/MCB.00149-17
Zhou Y et al (2016a) p38alpha has an important role in antigen cross-presentation by dendritic cells. Cell Mol Immunol 15:246–259. https://doi.org/10.1038/cmi.2016.49
Zhou Y, Hong Y, Huang H (2016b) Triptolide attenuates inflammatory response in membranous glomerulo-nephritis rat via downregulation of NF-kappaB signaling pathway. Kidney Blood Press Res 41:901–910. https://doi.org/10.1159/000452591
Ziaei S, Halaby R (2016) Immunosuppressive, anti-inflammatory and anti-cancer properties of triptolide: a mini review. Avicenna J Phytomed 6:149–164
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Research involving human participants and/or animals
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Informed consent
Not applicable.
Funding
None.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Qian, K., Zhang, L. & Shi, K. Triptolide prevents osteoarthritis via inhibiting hsa-miR-20b. Inflammopharmacol 27, 109–119 (2019). https://doi.org/10.1007/s10787-018-0509-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-018-0509-6