Abstract
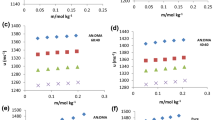
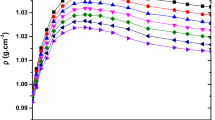
Ultrasonic-velocity and density measurements of aqueous solutions of maleic acid and tartaric acid have been made as a function of molality, at T = (298.15 and 308.15) K, and at atmospheric pressure. A molality range has been studied from (0.2603 to 2.6309) mol·kg−1 and (0.4451 to 2.6621) mol·kg−1 for maleic and tartaric acids, respectively. The experimental data have been correlated with molality using a polynomial equation. Furthermore, apparent molar volume, partial molar volume, apparent-specific molar volume, isentropic compressibility, apparent molar isentropic compressibility, limiting apparent molar isentropic compressibility, and isentropic apparent-specific compressibility values have been calculated from experimental values of densities and ultrasonic velocities. The calculated parameters have been interpreted in terms of solute–solvent interactions, solute–solute interactions, structure making/breaking behavior of acids, and their taste quality in water.
Similar content being viewed by others
References
J.O. Maryadele, The Merck Index: An Encyclopedia of Chemicals, Drugs and Biologicals, 4th edn. (Merck and Co., Inc., Whitehouse Station, NJ, 2006)
Hayvarinen A.P., Lihaveinen H., Gaman A., Vairila L., Ojala H., Kulmala M., Viisanen Y.: J. Chem. Eng. Data 51, 255 (2006)
Manzurola E., Apelblat A.: J. Chem. Thermodyn. 17, 579 (1985)
Hoiland H., Vikingstad E.: J. Chem. Soc. Faraday Trans. 1 71, 2007 (1975)
Benedetto G., Gavioso R.M., Giuliano Albo P.A., Lago S., Mandonna Ripa D., Spagnolo R.: Int. J. Thermophys. 26, 1667 (2005)
Resa J.M., Gonzalez C., Goenaga J.M., Iglesias M.: J. Therm. Anal. Calorim. 87, 237 (2007)
Wanger W., Pruss A.: J. Phys. Chem. Ref. Data 31, 387 (2002)
Riddick J.A., Bunger W.B., Sakana T.K.: Organic Solvents. Wiley-Interscience, New York (1986)
Polt A., Platzer B., Maurer G.: Chem. Tech. (Leipzig) 44, 216 (1992)
Pandey J.D., Jain P., Vyas V.: Can J. Chem. 72, 2486 (1994)
Pandey J.D., Mishra R.K., Dey R.: J. Mol. Liquids 123, 4 (2006)
Kupke D.W.: Physical Principles and Techniques of Physical Chemistry, Part C. Academic Press, New York (1973)
I.M. Plotz, R.M. Rosenberg, Chemical Thermodynamic Theory and Methods, 3rd edn. (W.A. Benjamin, Menlo Park, CA, 1972)
Redlich D., Mayer D.M.: Chem. Rev. 64, 222 (1964)
Ali A., Shahjahan : J. Iran. Chem. Soc. 3, 340 (2006)
Robinson R.A., Stokes R.H.: Electrolyte Solutions. Butterworths, London (1959)
Harned H.S., Owen B.B.: Physical Chemistry of Electrolyte Solutions. Chapman and Hall, London (1957)
Sadeghi R., Ziamajidi F.: J. Chem. Thermodyn. 39, 1118 (2007)
Soto A., Arce A., Khoshkbarchi M.K.: Biophys. Chem. 74, 165 (1998)
Songjun L., Jie H., Bailing L.: Polym. Int. 54, 96 (2004)
Parke S.A., Birch G.G., Roelina D.: Chem. Senses 24, 271 (1999)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kharat, S.J. Ultrasonic Velocity and Density Studies of Solutions of Maleic Acid and Tartaric Acid in Water at T = (298.15 and 308.15) K. Int J Thermophys 31, 585–594 (2010). https://doi.org/10.1007/s10765-010-0736-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10765-010-0736-6