Abstract
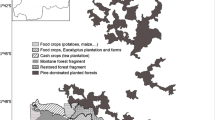
As forest loss and degradation continues, the human-dominated landscape outside protected areas should become increasingly relevant to primate conservation. Here we consider the Tanzanian endemic kipunji, Rungwecebus kipunji, whose small extent of occurrence (42 km2) and population (1117 individuals) qualify it for Critically Endangered status on the IUCN Red List. Habitat models suggest there is limited potential for expansion within the kipunji’s current protected forest habitat. In 2010, we examined the potential conservation role of land surrounding the forests using ecological surveys and structured interviews. Land outside protected forest is dominated by subsistence agriculture interspersed with tiny forest patches (almost all <0.4 km2) that cover only 2.4 % of the surveyed area located within 10 km of the forest boundary. Habitat bordering the forest forms a “hard edge” for kipunji, although some sites with single kipunji food trees, e.g., Ficus, offer some potential for use. However, tolerance of kipunji in the agricultural landscape may be limited in areas where kipunji was recorded crop raiding maize along the forest edge, and protection/retaliatory measures are employed. The Bujingijila corridor (2.1 km2) is a priority site for reforestation, particularly in the context of ongoing “Reducing Emissions from Deforestation and Forest Degradation (REDD+)” activities. We recorded the presence of kipunji food trees and little agriculture. Bujingijila could provide habitat for an additional 88 kipunji (8 % population increase), using density estimates from a 2006 census. Bujingijila has the additional benefit of reconnecting the Mt. Rungwe and Livingstone kipunji subpopulations.





Similar content being viewed by others
References
Akaike, H. (1973). Information theory as an extension of the maximum likelihood principle. In B. N. Petrov & F. Csaki (Eds.), Second international symposium on information theory (pp. 267–281). Budapest: Akademiai Kiado.
Anderson, J., Rowcliffe, J. M., & Cowlishaw, G. (2007). Does the matrix matter? A forest primate in a complex agricultural landscape. Biological Conservation, 135, 212–222.
Barbosa, C. E. A., Benato, T., Cavalheiro, A. L., & Torezan, J. M. D. (2009). Diversity of regenerating plants in reforestations with Araucaria angustifolia (Bertol.) O. Kuntze of 12, 22, 35, and 43 years of age in Paraná State, Brazil. Restoration Ecology, 17, 60–67.
Bonilla-Sánchez, Y. M., Serio-Silva, J. C., Pozo-Montuy, G., & Chapman, C. A. (2012). Howlers are able to survive in Eucalyptus plantations where remnant and regenerating vegetation is available. International Journal of Primatology, 33, 233–245.
Bracebridge, C. E., Davenport, T. R. B., & Marsden, S. J. (2011). Can we extend the area of occupancy of the kipunji, a critically endangered African primate? Animal Conservation, 14, 687–696.
Bracebridge, C. E., Davenport, T. R. B., & Marsden, S. J. (2012). The impact of forest disturbance on the seasonal foraging ecology of a Critically Endangered African primate. Biotropica, 44, 560–565.
Brown, N., Jennings, S., Wheeler, P., & Nabe-Nielsen, J. (2000). An improved method for the rapid assessment of forest understorey light environments. Journal of Applied Ecology, 37, 1044–1053.
Caro, T., Jones, T., & Davenport, T. R. B. (2009). Realities of documenting wildlife corridors in tropical countries. Biological Conservation, 142, 2807–2811.
Chaves, O. M., Stoner, K. E., & Arroyo-Rodríguez, V. (2012). Differences in diet between spider monkey groups living in forest fragments and continuous forest in Mexico. Biotropica, 44, 105–113.
Chazdon, R. L., Harvet, C. A., Komar, O., Griffith, D. M., Ferguson, B. G., Martínez-Ramos, M., Morales, H., Nigh, R., Soto-Pinto, L., van Breugal M., & Philpott, S. M. (2009a). Beyond reserves: A research agenda for conserving biodiversity in human-modified tropical landscapes. Biotropica, 41, 142–153.
Chazdon, R. L., Peres, C. A., Dent, D., Sheil, D., Lugo, A. E., Lamb, D., Stork, N. E., & Miller, S. E. (2009b). The potential for species conservation in tropical secondary forests. Conservation Biology, 23, 1406–1417.
Davenport, T. R. B., De Luca, D. W., Bracebridge, C. E., Machaga, S. J., Mpunga, N. E., Kibure, O., & Abeid, Y. S. (2010). Diet and feeding patterns in the kipunji Rungwecebus kipunji in the forests of Tanzania’s Southern Highlands. Primates, 51, 213–220.
Davenport, T. R. B., De Luca, D. W., Jones, T., Mpunga, N. E., Machaga, S. J., Kitegile, A., & Picton Phillips, G. (2008). The Critically Endangered kipunji Rungwecebus kipunji of southern Tanzania: First census and assessments of distribution and conservation status. Oryx, 42, 352–359.
Davenport, T. R. B., Nowak, K., & Perkin, P. (2013). Priority primate areas in Tanzania. Oryx. doi:10.1017/S0030605312001676.
Davenport, T. R. B., Stanley, W. T., Sargis, E. J., De Luca, D. W., Mpunga, N. E., Machaga, S. J., & Olson, L. E. (2006). A new genus of African monkey, Rungwecebus: Morphology, ecology, and molecular phylogenetics. Science, 312, 1378–1381.
DeClerck, F. A. J., Chazdon, R., Holl, K. D., Milder, J. C., Finegan, B., Martinez-Salinas, A., Imbach, P., Canet, L., & Ramos, Z. (2010). Biodiversity conservation in human-modified landscapes of Mesoamerica: Past, present and future. Biological Conservation, 143, 2301–2313.
Estrada, A., Raboy, B. E., & Oliveira, L. C. (2012). Agroecosystems and primate conservation in the tropics: A review. American Journal of Primatology, 74, 696–711.
Fa, J. E., Ryan, S. F., & Bell, D. J. (2005). Hunting vulnerability, ecological characteristics and harvest rates of bushmeat species in afrotropical forests. Biological Conservation, 121, 167–176.
Fashing, P. J., Nguyen, N., Luteshi, P., Opondo, W., Cash, J. F., & Cords, M. (2012). Evaluating the suitability of planted forests for African forest monkeys: A case study from Kakamega Forest, Kenya. American Journal of Primatology, 74, 77–90.
Gibson, L., Lee, T. M., Brook, B. W., Gardner, T. A., Barlow, J., Peres, C. A., Bradshaw, C. J. A., Laurance, W. F., Lovejoy, T. E., & Sodhi, N. S. (2011). Primary forests are irreplaceable for sustaining tropical biodiversity. Nature, 478, 378–381.
Harkin, D. A. (1960). The Rungwe Volcanics at the northern end of Lake Nyasa. Geological survey of Tanganyika Memoir II. Dar es Salaam: Government Printer.
Hill, C., Osborn, F., & Plumptre, A. J. (2002). Human-wildlife conflict: Identifying the problem and possible solutions. Albertine Rift Technical Report Series, Vol. 1. New York: Wildlife Conservation Society.
Hill, C. M., & Wallace, G. E. (2012). Crop protection and conflict mitigation: Reducing the costs of living alongside non-human primates. Biodiversity and Conservation. doi:10.1007/s10531-012-0318-y.
Jones, T., Ehardt, C. L., Butynski, T. M., Davenport, T. R. B., Mpunga, N. E., Machaga, S. J., & De Luca, D. W. (2005). The highland mangabey Lophocebus kipunji: A news species of African monkey. Science, 308, 1161–1164.
King, L. E., Lawrence, A., Douglas-Hamilton, I., & Vollrath, F. (2009). Beehive fence deters crop-raiding elephants. African Journal of Ecology, 47, 131–137.
Lamb, D. (1998). Large-scale ecological restoration of degraded tropical forest lands: the potential role of timber plantations. Restoration Ecology, 6, 271–279.
McCullagh, P., & Nelder, J. A. (1989). Generalised linear models (2nd ed.). London: Chapman and Hall.
McLennan, M. R., & Plumptre, A. J. (2012). Protected apes, unprotected forest: Composition, structure and diversity of riverine forest fragments and their conservation value in Uganda. Tropical Conservation Science, 5, 79–103.
Moore, R. S., Nekaris, K. A. I., & Eschmann, C. (2010). Habitat use by western purple-faced langurs Trachypithecus vetulus nestor (Colobinae) in a fragmented suburban landscape. Endangered Species Research, 12, 227–234.
Naughton-Treves, L., Treves, A., Chapman, C., & Wrangham, R. (1998). Temporal patterns of crop raiding by primates: Linking food availability in croplands and adjacent forest. Journal of Applied Ecology, 35, 596–606.
Newing, H., Eagle, C. M., Puri, R. K., & Watson, C. W. (2011). Conducting research in conservation: A social science perspective. London: Routledge.
Phalan, B., Onial, M., Balmford, A., & Green, R. E. (2011). Reconciling food production and biodiversity conservation: Land sharing and land sparing compared. Science, 333, 1289–1291.
Plumptre, A. J., Bizumuremyi, J.-B., Uwimana, F., & Ndaruhebeye, J.-D. (1997). The effects of the Rwandan civil war on poaching of ungulates in the Parc National des Volcans. Oryx, 31, 265–273.
Pozo-Montuy, G., Serio-Silva, J. C., & Bonilla-Sánchez, Y. M. (2011). Influence of the landscape matrix on the abundance of arboreal primates in fragmented landscapes. Primates, 52, 139–147.
R Development Core Team (2012). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. Available at: http://www.R-project.org (Accessed April 15, 2012).
Riley, E. P. (2007). Flexibility in diet and activity patterns of Macaca tonkeana in response to anthropogenic habitat alteration. International Journal of Primatology, 28, 107–133.
Sitati, N. W., & Walpole, M. J. (2006). Assessing farm-based measures for mitigating human-elephant conflict in Transmara District, Kenya. Oryx, 40, 279–286.
Strum, S. C. (2010). The development of primate raiding: Implications for management and conservation. International Journal of Primatology, 31, 133–156.
Suresh Kumar, R., Gama, N., Raghunath, R., Sinha, A., & Mishra, C. (2008). In search of the munzala: Distribution and conservation status of the newly-discovered Arunachal macaque Macaca munzala. Oryx, 42, 360–366.
Tanzanian Bureau of Statistics. (2002). Population and housing census 2002. Dar es Salaam: Bureau of Statistics.
Thapa, S. (2010). Effectiveness of crop protection methods against wildlife damage: A case study of two villages at Bardia National Park, Uganda. Crop Protection, 29, 1297–1304.
UN-REDD (2009). UN-REDD programme. Available at: http://www.UN-REDD.org (Accessed November 20, 2012).
Warren, Y., Buba, B., & Ross, C. (2007). Patterns of crop raiding by wild and domestic animals near Gashaka Gumti National Park, Nigeria. International Journal of Pest Management, 53, 207–216.
WCS (2003). Socio-economic surveys in communities around Mt. Rungwe and Livingstone forests, Southern Highlands, Tanzania. Unpublished data.
WCS. (2011a). Approach overview for tier 1 baseline calculation using international and freely available datasets. Report to Tanzanian REDD Taskforce and Norwegian. Mbeya: Embassy.
WCS (2011b). REDD readiness in Southwest Tanzania: The development of standardised approaches for establishing baseline information, monitoring forest degradation and supporting community incentives, education and resource supply in support of REDD in Tanzania, with a focus on the montane forests of the Southern Highlands. Annual Report to Norwegian Embassy.
Wieczkowski, J. (2010). Tana River mangabey use of nonforest areas: Functional connectivity in a fragmented landscape in Kenya. Biotropica, 42, 598–604.
Wiederholt, R., & Post, E. (2010). Tropical warming and the dynamics of endangered primates. Biology Letters, 6, 257–260.
Acknowledgments
Work in the Southern Highlands was funded by the Wildlife Conservation Society (WCS). We thank Karen, Dan, and Zak Pritzker; Katie Carpenter; and an anonymous donor. Research permission was granted by the Tanzania Wildlife Research Institute, Tanzania Commission for Science and Technology, Tanzania National Parks, Tanzania Forestry and Beekeeping Division, Mbeya Regional Administration, and Rungwe District. Many thanks to field assistants Geofrey Fokile and Paulo Maingo for the transect work. We thank Yahya Abeid, Roy Gereau, and Omari Kibure for plant specimen verification, and Buto Kilasa and Ramathan Mduruma for logistical support. We thank two anonymous reviewers for constructive comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 24 kb)
Rights and permissions
About this article
Cite this article
Bracebridge, C.E., Davenport, T.R.B., Mbofu, V.F. et al. Is There a Role for Human-Dominated Landscapes in the Long-Term Conservation Management of the Critically Endangered Kipunji (Rungwecebus kipunji)?. Int J Primatol 34, 1122–1136 (2013). https://doi.org/10.1007/s10764-013-9719-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10764-013-9719-3