Abstract
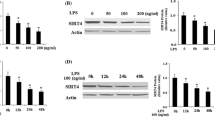
Endothelial cells play an important role in health and a variety of diseases. Recent evidences show that endothelial cells rely on glycolysis rather than on oxidative phosphorylation to generate energy to support cellular functions such as angiogenesis. However, the effect of endothelial glycolysis on vascular inflammation remains little known. Here, we investigate the role of key glycolytic enzyme PFKFB3 in tumor necrosis factor-α (TNF-α)-induced endothelial proinflammatory responses. siRNAs were used to knockdown the expression of PFKFB3. In some experiments, PFKFB3 inhibitors were also used. TNF-α at 20 ng/ml was added to confluent endothelial cells for different time period of stimulation. PFKFB3 expression was examined by RT-PCR and western blotting. Cytokine antibody panel membranes were employed to detect different cytokines/chemokines in culture supernatant of endothelial cells. The determination of monocyte adhesion to endothelial cells after TNF-α treatment was conducted using THP-1 cells. The monocyte attraction was performed using Transwell filters. For further mechanisms, NF-κB-p65 localization was examined by immunofluorescence. Expression of total IκB, phospho-IκB, phospho-NF-κB-p65, and Ikkβ was detected by western blotting. DNA-binding activity of NF-κB was assessed using electrophoretic mobility shift assay. We found that TNF-α increased endothelial PFKFB3 expression. Knockdown of PFKFB3 almost blocked all TNF-α-induced release of the proinflammatory cytokines/chemokines (MCP-1, IL-8, CXCL1, GMCSF, RANTES, TNF-α) and ICAM-1. PFKFB3 knockdown also significantly inhibited TNF-α-induced monocyte adhesion and transmigration. Furthermore, inhibition of PFKFB3 inhibited TNF-α-induced Ikkβ phosphorylation, IκBα phosphorylation and degradation, NF-κB-p65 phosphorylation, nuclear translocation, and DNA-binding activity. Thus, our results demonstrate that glycolytic enzyme PFKFB3 plays a critical role in TNF-α-induced endothelial inflammation.





Similar content being viewed by others
References
Aird, W.C. 2003. The role of the endothelium in severe sepsis and multiple organ dysfunction syndrome. Blood 101: 3765–3777. https://doi.org/10.1182/blood-2002-06-1887.
Aird, W.C. 2004. Endothelium as an organ system. Critical Care Medicine 32: S271–S279.
Aird, W.C. 2008. Endothelium in health and disease. Pharmacological Reports 60: 139–143.
Alkalay, I., A. Yaron, A. Hatzubai, A. Orian, A. Ciechanover, and Y. Ben-Neriah. 1995. Stimulation-dependent I kappa B alpha phosphorylation marks the NF-kappa B inhibitor for degradation via the ubiquitin-proteasome pathway. Proceedings of the National Academy of Sciences of the United States of America 92: 10599–10603.
Bando, H., T. Atsumi, T. Nishio, H. Niwa, S. Mishima, C. Shimizu, N. Yoshioka, R. Bucala, and T. Koike. 2005. Phosphorylation of the 6-phosphofructo-2-kinase/fructose 2,6-bisphosphatase/PFKFB3 family of glycolytic regulators in human cancer. Clinical Cancer Research 11: 5784–5792. https://doi.org/10.1158/1078-0432.CCR-05-0149.
Cantelmo, A.R., L.C. Conradi, A. Brajic, J. Goveia, J. Kalucka, A. Pircher, P. Chaturvedi, J. Hol, B. Thienpont, L.A. Teuwen, S. Schoors, B. Boeckx, J. Vriens, A. Kuchnio, K. Veys, B. Cruys, L. Finotto, L. Treps, T.E. Stav-Noraas, F. Bifari, P. Stapor, I. Decimo, K. Kampen, K. de Bock, G. Haraldsen, L. Schoonjans, T. Rabelink, G. Eelen, B. Ghesquière, J. Rehman, D. Lambrechts, A.B. Malik, M. Dewerchin, and P. Carmeliet. 2016. Inhibition of the glycolytic activator PFKFB3 in endothelium induces tumor vessel normalization, impairs metastasis, and improves chemotherapy. Cancer Cell 30: 968–985. https://doi.org/10.1016/j.ccell.2016.10.006.
Chen, Z., J. Hagler, V.J. Palombella, F. Melandri, D. Scherer, D. Ballard, and T. Maniatis. 1995. Signal-induced site-specific phosphorylation targets I kappa B alpha to the ubiquitin-proteasome pathway. Genes & Development 9: 1586–1597.
Clem, B., S. Telang, A. Clem, A. Yalcin, J. Meier, A. Simmons, M.A. Rasku, S. Arumugam, W.L. Dean, J. Eaton, A. Lane, J.O. Trent, and J. Chesney. 2008. Small-molecule inhibition of 6-phosphofructo-2-kinase activity suppresses glycolytic flux and tumor growth. Molecular Cancer Therapeutics 7: 110–120. https://doi.org/10.1158/1535-7163.MCT-07-0482.
Culic, O., M.L. Gruwel, and J. Schrader. 1997. Energy turnover of vascular endothelial cells. The American Journal of Physiology 273: C205–C213.
De Bock, K., M. Georgiadou, S. Schoors, A. Kuchnio, B.W. Wong, A.R. Cantelmo, A. Quaegebeur, et al. 2013. Role of PFKFB3-driven glycolysis in vessel sprouting. Cell 154: 651–663. https://doi.org/10.1016/j.cell.2013.06.037.
Dobrina, A., and F. Rossi. 1983. Metabolic properties of freshly isolated bovine endothelial cells. Biochimica et Biophysica Acta 762: 295–301.
Gatenby, R.A., and R.J. Gillies. 2004. Why do cancers have high aerobic glycolysis? Nature Reviews. Cancer 4: 891–899. https://doi.org/10.1038/nrc1478.
Gimbrone, M.A., Jr., and G. Garcia-Cardena. 2016. Endothelial cell dysfunction and the pathobiology of atherosclerosis. Circulation Research 118: 620–636. https://doi.org/10.1161/CIRCRESAHA.115.306301.
Libby, P., P.M. Ridker, and A. Maseri. 2002. Inflammation and atherosclerosis. Circulation 105: 1135–1143.
Lunt, S.Y., and M.G. Vander Heiden. 2011. Aerobic glycolysis: meeting the metabolic requirements of cell proliferation. Annual Review of Cell and Developmental Biology 27: 441–464. https://doi.org/10.1146/annurev-cellbio-092910-154237.
Obach, M., A. Navarro-Sabate, J. Caro, X. Kong, J. Duran, M. Gomez, J.C. Perales, F. Ventura, J.L. Rosa, and R. Bartrons. 2004. 6-Phosphofructo-2-kinase (pfkfb3) gene promoter contains hypoxia-inducible factor-1 binding sites necessary for transactivation in response to hypoxia. The Journal of Biological Chemistry 279: 53562–53570. https://doi.org/10.1074/jbc.M406096200.
Parra-Bonilla, G., D.F. Alvarez, A.B. Al-Mehdi, M. Alexeyev, and T. Stevens. 2010. Critical role for lactate dehydrogenase A in aerobic glycolysis that sustains pulmonary microvascular endothelial cell proliferation. American Journal of Physiology. Lung Cellular and Molecular Physiology 299: L513–L522. https://doi.org/10.1152/ajplung.00274.2009.
Peters, K., G. Kamp, A. Berz, R.E. Unger, S. Barth, A. Salamon, J. Rychly, and C.J. Kirkpatrick. 2009. Changes in human endothelial cell energy metabolic capacities during in vitro cultivation. The role of “aerobic glycolysis” and proliferation. Cellular Physiology and Biochemistry 24: 483–492. https://doi.org/10.1159/000257490.
Pfeiffer, T., S. Schuster, and S. Bonhoeffer. 2001. Cooperation and competition in the evolution of ATP-producing pathways. Science 292: 504–507. https://doi.org/10.1126/science.1058079.
Pober, J.S., and W.C. Sessa. 2007. Evolving functions of endothelial cells in inflammation. Nature Reviews. Immunology 7: 803–815.
Reid, M.A., X.H. Lowman, M. Pan, T.Q. Tran, M.O. Warmoes, M.B. Ishak Gabra, Y. Yang, J.W. Locasale, and M. Kong. 2016. IKKbeta promotes metabolic adaptation to glutamine deprivation via phosphorylation and inhibition of PFKFB3. Genes & Development 30: 1837–1851. https://doi.org/10.1101/gad.287235.116.
Ross, R. 1999. Atherosclerosis--an inflammatory disease. The New England Journal of Medicine 340: 115–126. https://doi.org/10.1056/NEJM199901143400207.
Sakurai, H., S. Suzuki, N. Kawasaki, H. Nakano, T. Okazaki, A. Chino, T. Doi, and I. Saiki. 2003. Tumor necrosis factor-alpha-induced IKK phosphorylation of NF-kappaB p65 on serine 536 is mediated through the TRAF2, TRAF5, and TAK1 signaling pathway. J Biol Chem 278: 36916–36923. https://doi.org/10.1074/jbc.M301598200.
Schoors, S., K. De Bock, A.R. Cantelmo, M. Georgiadou, B. Ghesquiere, S. Cauwenberghs, A. Kuchnio, et al. 2014. Partial and transient reduction of glycolysis by PFKFB3 blockade reduces pathological angiogenesis. Cell Metabolism 19: 37–48. https://doi.org/10.1016/j.cmet.2013.11.008.
Vegran, F., R. Boidot, C. Michiels, P. Sonveaux, and O. Feron. 2011. Lactate influx through the endothelial cell monocarboxylate transporter MCT1 supports an NF-kappaB/IL-8 pathway that drives tumor angiogenesis. Cancer Research 71: 2550–2560. https://doi.org/10.1158/0008-5472.CAN-10-2828.
Xu, Y., X. An, X. Guo, T.G. Habtetsion, Y. Wang, X. Xu, S. Kandala, Q. Li, H. Li, C. Zhang, R.B. Caldwell, D.J. Fulton, Y. Su, M.N. Hoda, G. Zhou, C. Wu, and Y. Huo. 2014. Endothelial PFKFB3 plays a critical role in angiogenesis. Arteriosclerosis, Thrombosis, and Vascular Biology 34: 1231–1239. https://doi.org/10.1161/ATVBAHA.113.303041.
Yalcin, A., B.F. Clem, A. Simmons, A. Lane, K. Nelson, A.L. Clem, E. Brock, D. Siow, B. Wattenberg, S. Telang, and J. Chesney. 2009. Nuclear targeting of 6-phosphofructo-2-kinase (PFKFB3) increases proliferation via cyclin-dependent kinases. The Journal of Biological Chemistry 284: 24223–24232. https://doi.org/10.1074/jbc.M109.016816.
Yamamoto, T., N. Takano, K. Ishiwata, M. Ohmura, Y. Nagahata, T. Matsuura, A. Kamata, K. Sakamoto, T. Nakanishi, A. Kubo, T. Hishiki, and M. Suematsu. 2014. Reduced methylation of PFKFB3 in cancer cells shunts glucose towards the pentose phosphate pathway. Nature Communications 5: 3480. https://doi.org/10.1038/ncomms4480.
Zhang, Q., M.J. Lenardo, and D. Baltimore. 2017. 30 years of NF-kappaB: a blossoming of relevance to human pathobiology. Cell 168: 37–57. https://doi.org/10.1016/j.cell.2016.12.012.
Acknowledgments
Part of this abstract was submitted to the 2018 Annual Critical Care Congress in San Antonio.
Funding
This work was supported by the National Natural Science Foundation of China (NSFC, Grant No. 81301614 and No.81372034).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interests
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Zhang, R., Li, R., Liu, Y. et al. The Glycolytic Enzyme PFKFB3 Controls TNF-α-Induced Endothelial Proinflammatory Responses. Inflammation 42, 146–155 (2019). https://doi.org/10.1007/s10753-018-0880-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-018-0880-x