Abstract
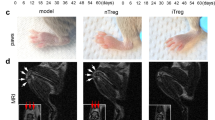
Rheumatoid arthritis (RA) is a systemic autoimmune disease that may cause bone damage and worsening disability. Manipulating antigen-specific Treg cells is a promising approach to treat autoimmune disease since the immune suppressive function of Treg cells has the feature of antigen specificity which could avoid overall immune suppression. It has been known that the function of Treg cells is impaired in RA, and adoptive transfer of Treg cells is effective in suppressing RA. Here, we designed a new approach to generate antigen-specific Treg cells by culturing CD4+ T cells from mice with RA onset, and we also proved that the adoptive transfer of these antigen-specific Treg cells reversed the collagen-induced arthritis (CIA) progression by suppressing the key inflammatory cytokine TNF-α. Further analysis showed that the transferred Treg cells were stable in vivo. These findings suggest this novel approach may have clinical applications for treatment of autoimmunity, including RA and other autoimmune disorders.




Similar content being viewed by others
References
Smolen, J.S., D. Aletaha, and I.B. McInnes. 2016. Rheumatoid arthritis. The Lancet. 388: 2023–2038.
Saag, K.G., G.G. Teng, N.M. Patkar, J. Anuntiyo, C. Finney, J.R. Curtis, et al. 2008. American College of Rheumatology 2008 recommendations for the use of nonbiologic and biologic disease-modifying antirheumatic drugs in rheumatoid arthritis. Arthritis and Rheumatism 59: 762–784.
Shi, W., Y.M. Wang, L.S. Li, M. Yan, D. Li, N.N. Chen, et al. 2004. Safety and efficacy of oral nonsteroidal anti-inflammatory drugs in patients with rheumatoid arthritis : A six-month randomised study. Clinical drug investigation. 24: 89–101.
Feldmann, M., and S.R. Maini. 2008. Role of cytokines in rheumatoid arthritis: An education in pathophysiology and therapeutics. Immunological Reviews 223: 7–19.
Tung, C.H., M.C. Lu, N.S. Lai, and S.F. Wu. 2017. Tumor necrosis factor-alpha blockade treatment decreased CD154 (CD40-ligand) expression in rheumatoid arthritis. PLoS One 12: e0183726.
Rein, P., and R.B. Mueller. 2017. Treatment with biologicals in rheumatoid arthritis: An overview. Rheumatology and Therapy.
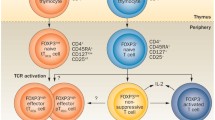
Zhang, D., E. Tu, S. Kasagi, P. Zanvit, Q. Chen, and W. Chen. 2015. Manipulating regulatory T cells: A promising strategy to treat autoimmunity. Immunotherapy 7: 1201–1211.
Li, M.O., and A.Y. Rudensky. 2016. T cell receptor signalling in the control of regulatory T cell differentiation and function. Nature reviews Immunology. 16: 220–233.
Chen, W., W. Jin, N. Hardegen, K.J. Lei, L. Li, N. Marinos, et al. 2003. Conversion of peripheral CD4+CD25-naive T cells to CD4+CD25+ regulatory T cells by TGF-beta induction of transcription factor Foxp3. The Journal of experimental medicine. 198: 1875–1886.
Malek, T.R., and A.L. Bayer. 2004. Tolerance, not immunity, crucially depends on IL-2. Nature reviews Immunology. 4: 665–674.
Mucida, D., Y. Park, G. Kim, O. Turovskaya, I. Scott, M. Kronenberg, et al. 2007. Reciprocal TH17 and regulatory T cell differentiation mediated by retinoic acid. Science 317: 256–260.
Wing, K., and S. Sakaguchi. 2010. Regulatory T cells exert checks and balances on self tolerance and autoimmunity. Nature Immunology 11: 7–13.
Leipe, J., A. Skapenko, P.E. Lipsky, and H. Schulze-Koops. 2005. Regulatory T cells in rheumatoid arthritis. Arthritis research & therapy. 7: 93.
Boissier, M.C., E. Assier, J. Biton, A. Denys, G. Falgarone, and N. Bessis. 2009. Regulatory T cells (Treg) in rheumatoid arthritis. Joint, bone, spine : revue du rhumatisme. 76: 10–14.
Morgan, M.E., R. Flierman, L.M. van Duivenvoorde, H.J. Witteveen, W. van Ewijk, J.M. van Laar, et al. 2005. Effective treatment of collagen-induced arthritis by adoptive transfer of CD25+ regulatory T cells. Arthritis and Rheumatism 52: 2212–2221.
Kong, N., Q. Lan, M. Chen, J. Wang, W. Shi, D.A. Horwitz, et al. 2012. Antigen-specific transforming growth factor beta-induced Treg cells, but not natural Treg cells, ameliorate autoimmune arthritis in mice by shifting the Th17/Treg cell balance from Th17 predominance to Treg cell predominance. Arthritis and Rheumatism 64: 2548–2558.
Hill, J.A., D.A. Bell, W. Brintnell, D. Yue, B. Wehrli, A.M. Jevnikar, et al. 2008. Arthritis induced by posttranslationally modified (citrullinated) fibrinogen in DR4-IE transgenic mice. The Journal of experimental medicine. 205: 967–979.
Inglis, J.J., E. Simelyte, F.E. McCann, G. Criado, and R.O. Williams. 2008. Protocol for the induction of arthritis in C57BL/6 mice. Nature protocols. 3: 612–618.
Brand, D.D., K.A. Latham, and E.F. Rosloniec. 2007. Collagen-induced arthritis. Nature protocols. 2: 1269–1275.
Zhang, D., C. Chia, X. Jiao, W. Jin, S. Kasagi, R. Wu, et al. 2017. D-mannose induces regulatory T cells and suppresses immunopathology. Nature Medicine.
Placek, K., G. Hu, K. Cui, D. Zhang, Y. Ding, J.E. Lee, et al. 2017. MLL4 prepares the enhancer landscape for Foxp3 induction via chromatin looping. Nature Immunology 18: 1035–1045.
Fang, Z.Z., D. Zhang, Y.F. Cao, C. Xie, D. Lu, D.X. Sun, et al. 2016. Irinotecan (CPT-11)-induced elevation of bile acids potentiates suppression of IL-10 expression. Toxicology and applied pharmacology. 291: 21–27.
La Paglia, G.M.C., M.C. Leone, G. Lepri, R. Vagelli, E. Valentini, A. Alunno, et al. 2017. One year in review 2017: Systemic lupus erythematosus. Clinical and experimental rheumatology. 35: 551–561.
Konkel, J.E., D. Zhang, P. Zanvit, C. Chia, T. Zangarle-Murray, W. Jin, et al. 2017. Transforming growth factor-beta signaling in regulatory T cells controls T helper-17 cells and tissue-specific immune responses. Immunity 46: 660–674.
Kasagi, S., P. Zhang, L. Che, B. Abbatiello, T. Maruyama, H. Nakatsukasa, et al. 2014. In vivo-generated antigen-specific regulatory T cells treat autoimmunity without compromising antibacterial immune response. Science Translational Medicine 6: 241ra78.
Zanvit, P., J.E. Konkel, X. Jiao, S. Kasagi, D. Zhang, R. Wu, et al. 2015. Antibiotics in neonatal life increase murine susceptibility to experimental psoriasis. Nature Communications 6: 8424.
Zhang, D., J. Wang, Z. Li, M. Zhou, Q. Chen, X. Zeng, et al. 2015. The activation of NF-kappaB in infiltrated mononuclear cells negatively correlates with Treg cell frequency in oral lichen planus. Inflammation 38: 1683–1689.
Tang, Q., K.J. Henriksen, M. Bi, E.B. Finger, G. Szot, J. Ye, et al. 2004. In vitro-expanded antigen-specific regulatory T cells suppress autoimmune diabetes. The Journal of experimental medicine. 199: 1455–1465.
Tarbell, K.V., L. Petit, X. Zuo, P. Toy, X. Luo, A. Mqadmi, et al. 2007. Dendritic cell-expanded, islet-specific CD4+ CD25+ CD62L+ regulatory T cells restore normoglycemia in diabetic NOD mice. The Journal of experimental medicine. 204: 191–201.
Nguyen, T.L., N.L. Sullivan, M. Ebel, R.M. Teague, and R.J. DiPaolo. 2011. Antigen-specific TGF-beta-induced regulatory T cells secrete chemokines, regulate T cell trafficking, and suppress ongoing autoimmunity. Journal of immunology. 187: 1745–1753.
Stephens, L.A., K.H. Malpass, and S.M. Anderton. 2009. Curing CNS autoimmune disease with myelin-reactive Foxp3+ Treg. European Journal of Immunology 39: 1108–1117.
Jaeckel, E., H. von Boehmer, and M.P. Manns. 2005. Antigen-specific FoxP3-transduced T-cells can control established type 1 diabetes. Diabetes 54: 306–310.
Tu, E., D. Bourges, P.A. Gleeson, D.K. Ang, and I.R. van Driel. 2013. Pathogenic T cells persist after reversal of autoimmune disease by immunosuppression with regulatory T cells. European Journal of Immunology 43: 1286–1296.
Thomas, R. 2013. Dendritic cells and the promise of antigen-specific therapy in rheumatoid arthritis. Arthritis research & therapy. 15: 204.
Corrigall, V.M., and G.S. Panayi. 2002. Autoantigens and immune pathways in rheumatoid arthritis. Critical Reviews in Immunology. 22: 281–293.
Funding
This work was supported by grants from the National Natural Science Foundations of China (NO.81600876), the Promotive Research Fund for Excellent Young and Middle-Aged Scientists of Shandong Province (BS2010YY054), the Science Foundation for The Youth Scholars of Sichuan University (NO.2016SCU11048), Shandong Medical and Health Technology Development (2014WSB04020), Shandong Provincial Natural Science Foundation (ZR2015HM060), and Shandong Provincial Science and Technology Development Projects Foundation (2013GSF31805).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
All animal studies were approved by the Subcommittee on Research and Animal Care (SRAC) of Shandong University and Sichuan University, and were performed by following the guidelines of Experimental Animal Center of Shandong University and Sichuan University for the use and care of live animals.
Conflict of Interest
The authors declare that there is no conflict of interest.
Rights and permissions
About this article
Cite this article
Sun, G., Hou, Y., Gong, W. et al. Adoptive Induced Antigen-Specific Treg Cells Reverse Inflammation in Collagen-Induced Arthritis Mouse Model. Inflammation 41, 485–495 (2018). https://doi.org/10.1007/s10753-017-0704-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-017-0704-4