Abstract
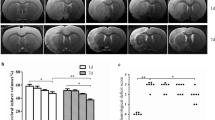
Salidroside is neuroprotective across a wide therapeutic time-window after cerebral ischemia-reperfusion injury (IRI). Here, we investigated the role of complement in mediating effects of salidroside after cerebral IRI in rats. Rats were administrated with vehicle or salidroside 50 mg/kg, given daily for either 24 or 48 h, after middle cerebral artery occlusion (MCAO) for 2 h and reperfusion for 1 h. Levels of proteins in ischemic brain were measured by immunofluorescence and western blotting. We observed early increases in the deposition of immunoglobulin M, mannose-binding lectin 2, and annexin IV on cerebral endothelial cells, induction of the complement components C3 and C3a, by 24 h after IRI, and a later significant increase in the complement component C1q by 48 h. Salidroside prevented these changes. The neuroplasticity-related early growth response proteins Egr1, Egr2, and Egr4 and activity-regulated cytoskeleton-associated protein increased transiently in the first 6 h after IRI but then decreased below baseline by 48 h after IRI. Neither salidroside nor a C3a receptor antagonist (C3aRA) affected these proteins 24 h after IRI, but both reversed their later decreases to similar and non-additive extents. Salidroside and C3aRA increased NeuN in a non-additive manner after IRI. Our results suggest that salidroside exerts neuroprotection by reducing early activation of the lectin pathway on the cerebral endothelium and inhibiting the gradual activation of the classical pathway after cerebral IRI. This prolonged neuroprotection may depend, at least in part, on increased expression of neuroplasticity-related genes driven by reduced complement activation.








Similar content being viewed by others
References
Arumugam, T.V., T.M. Woodruff, J.D. Lathia, P.K. Selvaraj, M.P. Mattson, and S.M. Taylor. 2009. Neuroprotection in stroke by complement inhibition and immunoglobulin therapy. Neuroscience 158: 1074–1089.
Cowell, R.M., J.M. Plane, and F.S. Silverstein. 2003. Complement activation contributes to hypoxic-ischemic brain injury in neonatal rats. Journal of Neuroscience 23: 9459–9468.
Mocco, J., W.J. Mack, A.F. Ducruet, S.A. Sosunov, M.E. Sughrue, B.G. Hassid, M.N. Nair, I. Laufer, R.J. Komotar, M. Claire, H. Holland, D.J. Pinsky, and E.J. Connolly. 2006. Complement component C3 mediates inflammatory injury following focal cerebral ischemia. Circulation Research 99: 209–217.
Elvington, A., C. Atkinson, L. Kulik, H. Zhu, J. Yu, M.S. Kindy, V.M. Holers, and S. Tomlinson. 2012. Pathogenic natural antibodies propagate cerebral injury following ischemic stroke in mice. Journal of Immunology 188: 1460–1468.
Ducruet, A.F., S.A. Sosunov, B.E. Zacharia, J. Gorski, M.L. Yeh, P. Derosa, G. Cohen, P.R. Gigante, and E.J. Connolly. 2011. The neuroprotective effect of genetic mannose-binding lectin deficiency is not sustained in the sub-acute phase of stroke. Translational Stroke Research 2: 588–599.
Cervera, A., A.M. Planas, C. Justicia, X. Urra, J.C. Jensenius, F. Torres, F. Lozano, and A. Chamorro. 2010. Genetically-defined deficiency of mannose-binding lectin is associated with protection after experimental stroke in mice and outcome in human stroke. PLoS One 5: e8433.
Morrison, H., J. Frye, G. Davis-Gorman, J. Funk, P. McDonagh, G. Stahl, and L. Ritter. 2011. The contribution of mannose binding lectin to reperfusion injury after ischemic stroke. Current Neurovascular Research 8: 52–63.
Orsini, F., P. Villa, S. Parrella, R. Zangari, E.R. Zanier, R. Gesuete, M. Stravalaci, S. Fumagalli, R. Ottria, J.J. Reina, A. Paladini, E. Micotti, R. Ribeiro-Viana, J. Rojo, V.I. Pavlov, G.L. Stahl, A. Bernardi, M. Gobbi, and M.G. De Simoni. 2012. Targeting mannose-binding lectin confers long-lasting protection with a surprisingly wide therapeutic window in cerebral ischemia. Circulation 126: 1484–1494.
de la Rosa, X., A. Cervera, A.K. Kristoffersen, C.P. Valdes, H.M. Varma, C. Justicia, T. Durduran, A. Chamorro, and A.M. Planas. 2014. Mannose-binding lectin promotes local microvascular thrombosis after transient brain ischemia in mice. Stroke 45: 1453–1459.
Osthoff, M., M. Katan, F. Fluri, P. Schuetz, R. Bingisser, L. Kappos, A.J. Steck, S.T. Engelter, B. Mueller, M. Christ-Crain, and M. Trendelenburg. 2011. Mannose-binding lectin deficiency is associated with smaller infarction size and favorable outcome in ischemic stroke patients. PLoS One 6: e21338.
Petry, F., P.J. McClive, M. Botto, B.J. Morley, G. Morahan, and M. Loos. 1996. The mouse C1q genes are clustered on chromosome 4 and show conservation of gene organization. Immunogenetics 43: 370–376.
De Simoni, M.G., E. Rossi, C. Storini, S. Pizzimenti, C. Echart, and L. Bergamaschini. 2004. The powerful neuroprotective action of C1-inhibitor on brain ischemia-reperfusion injury does not require C1q. American Journal of Pathology 164: 1857–1863.
Elvington, A., C. Atkinson, H. Zhu, J. Yu, K. Takahashi, G.L. Stahl, M.S. Kindy, and S. Tomlinson. 2012. The alternative complement pathway propagates inflammation and injury in murine ischemic stroke. Journal of Immunology 189: 4640–4647.
Alawieh, A., and S. Tomlinson. 2016. Injury site-specific targeting of complement inhibitors for treating stroke. Immunological Reviews 274: 270–280.
Wei, Y., H. Hong, X. Zhang, W. Lai, Y. Wang, K. Chu, J. Brown, G. Hong, and L. Chen. 2017. Salidroside inhibits inflammation through PI3K/Akt/HIF signaling after focal cerebral ischemia in rats. Inflammation 40: 1297–1309.
Shi, T.Y., S.F. Feng, J.H. Xing, Y.M. Wu, X.Q. Li, N. Zhang, Z. Tian, S.B. Liu, and M.G. Zhao. 2012. Neuroprotective effects of salidroside and its analogue tyrosol galactoside against focal cerebral ischemia in vivo and H2O2-induced neurotoxicity in vitro. Neurotoxicity Research 21: 358–367.
Lai, W., Z. Zheng, X. Zhang, Y. Wei, K. Chu, J. Brown, G. Hong, and L. Chen. 2015. Salidroside-mediated neuroprotection is associated with induction of early growth response genes (Egrs) across a wide therapeutic window. Neurotoxicity Research 28: 108–121.
Cole, A.J., D.W. Saffen, J.M. Baraban, and P.F. Worley. 1989. Rapid increase of an immediate early gene messenger RNA in hippocampal neurons by synaptic NMDA receptor activation. Nature 340: 474–476.
O'Donovan, K.J., W.G. Tourtellotte, J. Millbrandt, and J.M. Baraban. 1999. The EGR family of transcription-regulatory factors: progress at the interface of molecular and systems neuroscience. Trends in Neurosciences 22: 167–173.
Kawahara, N., Y. Wang, A. Mukasa, K. Furuya, T. Shimizu, T. Hamakubo, H. Aburatani, T. Kodama, and T. Kirino. 2004. Genome-wide gene expression analysis for induced ischemic tolerance and delayed neuronal death following transient global ischemia in rats. Journal of Cerebral Blood Flow & Metabolism 24: 212–223.
Pérez-Cadahía, B., B. Drobic, and J.R. Davie. 2011. Activation and function of immediate-early genes in the nervous. Biochemistry and Cell Biology 89: 61–73.
Longa, E.Z., P.R. Weinstein, S. Carlson, and R. Cummins. 1989. Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke 20: 84–91.
Belayev, L., O.F. Alonso, R. Busto, W. Zhao, and M.D. Ginsberg. 1996. Middle cerebral artery occlusion in the rat by intraluminal suture. neurological and pathological evaluation of an improved model. Stroke 27: 1616–1622 discussion 1623.
Nakayama, H., M.D. Ginsberg, and W.D. Dietrich. 1988. (S)-emopamil, a novel calcium channel blocker and serotonin S2 antagonist, markedly reduces infarct size following middle cerebral artery occlusion in the rat. Neurology 38: 1667–1673.
Ducruet, A.F., B.G. Hassid, W.J. Mack, S.A. Sosunov, M.L. Otten, D.J. Fusco, Z.L. Hickman, G.H. Kim, R.J. Komotar, and J. Mocco. 2008. C3a receptor modulation of granulocyte infiltration after murine focal cerebral ischemia is reperfusion dependent. Journal of Cerebral Blood Flow & Metabolism 28: 1048–1058.
Lai, W., X. Tian, Q. Xiang, K. Chu, Y. Wei, J. Deng, S. Zhang, J. Brown, and G. Hong. 2015. 11Beta-HSD1 modulates LPS-induced innate immune responses in adipocytes by altering expression of PTEN. Molecular Endocrinology 29: 558–570.
Alawieh, A., A. Elvington, and S. Tomlinson. 2015. Complement in the homeostatic and ischemic brain. Frontiers in Immunology 6: 417.
Atkinson, C., H. Zhu, F. Qiao, J.C. Varela, J. Yu, H. Song, M.S. Kindy, and S. Tomlinson. 2006. Complement-dependent P-selectin expression and injury following ischemic stroke. Journal of Immunology 177: 7266–7274.
Mack, W.J., M.E. Sughrue, A.F. Ducruet, J. Mocco, S.A. Sosunov, B.G. Hassid, J.Z. Silverberg, V.S. Ten, D.J. Pinsky, and E.J. Connolly. 2006. Temporal pattern of C1q deposition after transient focal cerebral ischemia. Journal of Neuroscience Research 83: 883–889.
Luo, H., W. Li, F. Yang, L. Zhou, P. Wen, and J. Zhou. 2013. Expressions of complement C1q and C3c in rat brain tissues with cerebral ischemia/reperfusion injury. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi 29: 897–900.
Silverman, S.M., B.J. Kim, G.R. Howell, J. Miller, S.W. John, R.J. Wordinger, and A.F. Clark. 2016. C1q propagates microglial activation and neurodegeneration in the visual axis following retinal ischemia/reperfusion injury. Molecular Neurodegeneration 11: 24.
Lee, Y., J.C. Jung, S. Jang, J. Kim, Z. Ali, I.A. Khan, and S. Oh. 2013. Anti-inflammatory and neuroprotective effects of constituents isolated from Rhodiola rosea. Evidence-based Complementary and Alternative Medicine 2013: 514049.
Bozon, B., S. Davis, and S. Laroche. 2003. A requirement for the immediate early gene zif268 in reconsolidation of recognition memory after retrieval. Neuron 40: 695–701.
Jones, M.W., M.L. Errington, P.J. French, A. Fine, T.V. Bliss, S. Garel, P. Charnay, B. Bozon, S. Laroche, and S. Davis. 2001. A requirement for the immediate early gene Zif268 in the expression of late LTP and long-term memories. Nature Neuroscience 4: 289–296.
Lee, J.L., B.J. Everitt, and K.L. Thomas. 2004. Independent cellular processes for hippocampal memory consolidation and reconsolidation. Science 304: 839–843.
Inokuchi, K., A. Murayama, and F. Ozawa. 1996. MRNA differential display reveals Krox-20 as a neural plasticity-regulated gene in the rat hippocampus. Biochemical and Biophysical Research Communications 221: 430–436.
Williams, J., M. Dragunow, P. Lawlor, S. Mason, W.C. Abraham, J. Leah, R. Bravo, J. Demmer, and W. Tate. 1995. Krox20 may play a key role in the stabilization of long-term potentiation. Brain Research. Molecular Brain Research 28: 87–93.
DeSteno, D.A., and C. Schmauss. 2008. Induction of early growth response gene 2 expression in the forebrain of mice performing an attention-set-shifting task. Neuroscience 152: 417–428.
Li, L., J. Carter, X. Gao, J. Whitehead, and W.G. Tourtellotte. 2005. The neuroplasticity-associated arc gene is a direct transcriptional target of early growth response (Egr) transcription factors. Molecular & Cellular Biology 25: 10286–10300.
Honkaniemi, J., B.A. States, P.R. Weinstein, J. Espinoza, and F.R. Sharp. 1997. Expression of zinc finger immediate early genes in rat brain after permanent middle cerebral artery occlusion. Journal of Cerebral Blood Flow & Metabolism 17: 636–646.
Honkaniemi, J., and F.R. Sharp. 1996. Global ischemia induces immediate-early genes encoding zinc finger transcription factors. Journal of Cerebral Blood Flow & Metabolism 16: 557–565.
Ducruet, A.F., S.A. Sosunov, S.H. Visovatti, D. Petrovic-Djergovic, W.J. Mack, E.J. Connolly, and D.J. Pinsky. 2011. Paradoxical exacerbation of neuronal injury in reperfused stroke despite improved blood flow and reduced inflammation in early growth response-1 gene-deleted mice. Neurological Research 33: 717–725.
Tan, C.B., M. Gao, W.R. Xu, X.Y. Yang, X.M. Zhu, and G.H. Du. 2009. Protective effects of salidroside on endothelial cell apoptosis induced by cobalt chloride. Biological & Pharmaceutical Bulletin 32: 1359–1363.
Xu, M.C., H.M. Shi, H. Wang, and X.F. Gao. 2013. Salidroside protects against hydrogen peroxide-induced injury in HUVECs via the regulation of REDD1 and mTOR activation. Molecular Medicine Reports 8: 147–153.
Shi, K., X. Wang, J. Zhu, G. Cao, K. Zhang, and Z. Su. 2015. Salidroside protects retinal endothelial cells against hydrogen peroxide-induced injury via modulating oxidative status and apoptosis. Bioscience, Biotechnology, and Biochemistry 79: 1406–1413.
Chaitanya, G.V., A. Minagar, and J.S. Alexander. 2014. Neuronal and astrocytic interactions modulate brain endothelial properties during metabolic stresses of in vitro cerebral ischemia. Cell Communication and Signaling 12: 7.
Chaitanya, G.V., W.E. Cromer, S.R. Wells, M.H. Jennings, P.O. Couraud, I.A. Romero, B. Weksler, A. Erdreich-Epstein, J.M. Mathis, A. Minagar, and J.S. Alexander. 2011. Gliovascular and cytokine interactions modulate brain endothelial barrier in vitro. Journal of Neuroinflammation 8: 162.
Chen, S.F., H.J. Tsai, T.H. Hung, C.C. Chen, C.Y. Lee, C.H. Wu, P.Y. Wang, and N.C. Liao. 2012. Salidroside improves behavioral and histological outcomes and reduces apoptosis via PI3K/Akt signaling after experimental traumatic brain injury. PLoS One 7: e45763.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Projects 81473382 and 81603323), the Department of Technology and Science of Fujian Province (Projects 2014Y4004, 2015J01328, and 2015J01685), the Collaborative Innovation Center for Rehabilitation Technology of Fujian University of Traditional Chinese Medicine (TCM), and the Rehabilitation Research of State Administration of TCM.
The authors would like to thank the staff in the Animal Center of the Fujian University of TCM for their technical supports.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
No competing financial interests are associated with this paper.
Additional information
Wenfang Lai and XiuLi Xie contributed equally to this work.
Electronic Supplementary Material
ESM 1
(DOCX 3266 kb)
Rights and permissions
About this article
Cite this article
Lai, W., Xie, X., Zhang, X. et al. Inhibition of Complement Drives Increase in Early Growth Response Proteins and Neuroprotection Mediated by Salidroside After Cerebral Ischemia. Inflammation 41, 449–463 (2018). https://doi.org/10.1007/s10753-017-0701-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-017-0701-7