Abstract
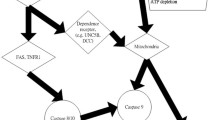
Forty-two patients with systemic lupus erythematosus (SLE), including 26 patients with renal damage and 16 without, and 20 healthy controls were included in the study. The isolated peripheral blood mononuclear cells (PBMCs) were treated with a p38 inhibitor (SB203580) or anti-tumor necrosis factor-like weak inducer of apoptosis (TWEAK) mAb, with or without phytohemagglutinin/phorbol myristate acetate (PHA/PMA) stimulation. Western blot experiments were used to evaluate the protein expression of TWEAK and p38 MAPK in PBMCs .Next, the contents of interleukin-10 (IL-10) and monocyte chemoattractant protein-1 (MCP-1) in the supernatant were measured by ELISA. The results showed that expression of TWEAK protein in PBMCs from lupus nephritis patients was significantly higher than that from SLE patients without renal damage and healthy controls. PHA/PMA simulation could upregulate the productions of TWEAK and p-p38MAPK in PBMCs from patients with SLE. Anti-TWEAK mAb treatment downregulated both TWEAK and p-p38 MAPK expression in PBMCs, as well as IL-10 and MCP-1 in the supernatant; SB203580 had the same effect on cytokine production in PBMC, but had no effect on the expression of TWEAK. Our results suggested that TWEAK–p38 MAPK–IL-10, MCP-1 signaling pathway in PBMC played an important pathogenic role in lupus nephritis.





Similar content being viewed by others
Abbreviations
- DAPI:
-
4′,6-Diamidino-2-phenylindole
- SLE:
-
Systemic lupus erythematosus
- LN:
-
Lupus nephritis
- TWEAK:
-
Tumor necrosis factor-like weak inducer of apoptosis
- p38 MAPK:
-
P38 mitogen- activated protein kinase
- IL-10:
-
Interleukin-10
- MCP-1:
-
Monocyte chemoattractant protein-1
- TNFRSF:
-
Tumor Necrosis Factor receptor superfamily
- NRSLE:
-
SLE patients without renal involvement
- PBMC:
-
Peripheral blood mononuclear cell
References
Namjou, B., J. Kilpatrick, and J.B. Harley. 2007. Genetics of clinical expression in SLE. Autoimmunity 40: 602–612.
Ka, S.M., C.W. Cheng, H.A. Shui, et al. 2007. Mesangial cells of lupus-prone mice are sensitive to chemokine production. Arthritis Research & Therapy 9: R67.
Tabibian-Keissar, H., N.S. Zuckerman, M. Barak, et al. 2008. B-cell clonal diversification and gut-lymph node trafficking in ulcerative colitis revealed using lineage tree analysis. European Journal of Immunology 38: 2600–2609.
Chan, O., and M.J. Shlomchik. 1998. A new role for B cells in systemic autoimmunity: B cells promote spontaneous T cell activation in MRL-lpr/lpr mice. Journal of Immunology 160: 51–59.
Schwartz, N., J.S. Michaelson, and C. Putterman. 2007. Lipocalin-2, TWEAK, and other cytokines as urinary biomarkers for lupus nephritis. Annals of the New York Academy of Sciences 1109: 265–274.
Li, Y., M. Tucci, S. Narain, et al. 2006. Urinary biomarkers in lupus nephritis. Autoimmunity Reviews 5: 383–388.
Goulvestre, C., F. Batteux, and J. Charreire. 2002. Chemokines modulate experimental autoimmune thyroiditis through attraction of autoreactive or regulatory T cells. European Journal of Immunology 32: 3435–3442.
Marks, S.D., S.J. Williams, K. Tullus, et al. 2008. Glomerular expression of monocyte chemoattractant protein-1 is predictive of poor renal prognosis in pediatric lupus nephritis. Nephrology, Dialysis, Transplantation 23: 3521–3526.
Zhao, Z., L.C. Burkly, S. Campbell, et al. 2007. TWEAK/Fn14 interactions are instrumental in the pathogenesis of nephritis in the chronic graft-versus-host model of systemic lupus erythematosus. Journal of Immunology 179: 7949–7958.
Chi, H., S.P. Barry, R.J. Roth, et al. 2006. Dynamic regulation of pro- and anti-inflammatory cytokines by MAPK phosphatase 1 (MKP-1) in innate immune responses. Proceedings of the National Academy of Sciences of the United States of America 103(7): 2274–2279.
Iwata, Y., T. Wada, K. Furuichi, et al. 2003. p38 Mitogen-activated protein kinase contributes to autoimmune renal injury in MRL-Fas lpr mice. Journal of the American Society of Nephrology 14(1): 57–67.
Wang, X.S., K. Diener, C.L. Manthey, et al. 1997. Molecular cloning and characterization of a novel p38 mitogen-activated protein kinase. Journal of Biological Chemistry 272: 23668–23674.
Herlaar, E., and Z. Brown. 1999. p38 MAPK signaling cascades in inflammatory disease. Mol Medicine Today 5: 439–447.
Wang, X., and Y. Liu. 2007. Regulation of innate immune response by MAP kinase phosphatase-1. Cellular Signalling 19: 1372–1382.
Li, L., S.F. Chen, and Y. Liu. 2009. MAP kinase phosphatase-1, a critical negative regulator of the innate immune response. International Journal of Clinical and Experimental Medicine 2: 48–67.
Cook, R., C.C. Wu, Y.J. Kang, et al. 2007. The role of the p38 pathway in adaptive immunity. Cellular & Molecular Immunology 4: 253–259.
Kim, J.M., J.M. White, A.S. Shaw, et al. 2005. MAPK p38 alpha is dispensable for lymphocyte development and proliferation. Journal of Immunology 174: 1239–1244.
Li, H., A. Mittal, P.K. Paul, et al. 2009. Tumor necrosis factor-related weak inducer of apoptosis augments matrix metalloproteinase 9 (MMP-9) production in skeletal muscle through the activation of nuclear factor inducing kinase and p38 mitogen-activated protein kinase, a potential role of MMP-9 in myopathy. The Journal of Biological Chemistry 284: 4439–4450.
Blanco-Colio, L.M., J.L. Martin-Ventura, B. Munoz-Garcia, et al. 2007. TWEAK and Fn14. New players in the pathogenesis of atherosclerosis. Frontiers in Bioscience 12: 3648–3655.
Winkles, J.A., N.L. Tran, and M.E. Berens. 2006. TWEAK and Fn14: new molecular targets for cancer therapy? Cancer Letters 235: 11–17.
Leng, R.X., H.F. Pan, W.Z. Qin, et al. 2010. TWEAK as a target for therapy in systemic lupus erythematosus. Molecular Biology Reports 38: 587–592.
Clark, J., P. Vagenas, M. Panesar, et al. 2005. What does tumour necrosis factor excess do to the immune system long term? Annals of the Rheumatic Diseases 64(Suppl 4): 70–76.
Cheema, I.R., C. Hermann, S. Postell, et al. 2000. Effect of chronic excess of tumour necrosis factor-alpha on contractile proteins in rat skeletal muscle. Cytobios 103: 169–176.
Campbell, S., J. Michaelson, L. Burkly, et al. 2004. The role of TWEAK/Fn14 in the pathogenesis of inflammation and systemic autoimmunity. Frontiers in Bioscience 9: 2273–2284.
Kollias, G., and D. Kontoyiannis. 2002. Role of TNF/TNFR in autoimmunity: specific TNF receptor blockade may be advantageous to anti-TNF treatments. Cytokine & Growth Factor Reviews 13: 315–321.
Kollias, G., D. Kontoyiannis, E. Douni, et al. 2002. The role of TNF/TNFR in organ-specific and systemic autoimmunity: implications for the design of optimized ‘anti-TNF’ therapies. Current Directions in Autoimmunity 5: 30–50.
Suvannavejh, G.C., H.O. Lee, J. Padilla, et al. 2000. Divergent roles for p55 and p75 tumor necrosis factor receptors in the pathogenesis of MOG(35–55)-induced experimental autoimmune encephalomyelitis. Cellular Immunology 205: 24–33.
Ju, S.T., K. Matsui, and M. Ozdemirli. 1999. Molecular and cellular mechanisms regulating T and B cell apoptosis through Fas/FasL interaction. International Reviews of Immunology 18: 485–513.
Wong, B., and Y. Choi. 1997. Pathways leading to cell death in T cells. Current Opinion in Immunology 9: 358–364.
Qin, H.Y., P. Chaturvedi, and B. Singh. 2004. In vivo apoptosis of diabetogenic T cells in NOD mice by IFN-gamma/TNF-alpha. International Immunology 16: 1723–1732.
Mueller, A.M., X. Pedre, I. Kleiter, et al. 2005. Targeting fibroblast growth factor-inducible-14 signaling protects from chronic relapsing experimental autoimmune encephalomyelitis. Journal of Neuroimmunology 159: 55–65.
Kaplan, M.J., E.E. Lewis, E.A. Shelden, et al. 2002. The apoptotic ligands TRAIL, TWEAK, and Fas ligand mediate monocyte death induced by autologous lupus T cells. Journal of Immunology 169: 6020–6029.
Schwartz, N., L. Su, L.C. Burkly, et al. 2006. Urinary TWEAK and the activity of lupus nephritis. Journal of Autoimmunity 27: 242–250.
Molano, A., P. Lakhani, A. Aran, et al. 2009. TWEAK stimulation of kidney resident cells in the pathogenesis of graft versus host induced lupus nephritis. Immunology Letters 125: 119–128.
Xu, H., A. Okamoto, J. Ichikawa, et al. 2004. TWEAK/Fn14 interaction stimulates human bronchial epithelial cells to produce IL-8 and GM-CSF. Biochemical and Biophysical Research Communications 318: 422–427.
Gao, H.X., S.R. Campbell, L.C. Burkly, et al. 2009. TNF-like weak inducer of apoptosis (TWEAK) induces inflammatory and proliferative effects in human kidney cells. Cytokine 46: 24–35.
Lichtner, M., C. Maranon, P.O. Vidalain, et al. 2004. HIV type 1-infected dendritic cells induce apoptotic death in infected and uninfected primary CD4 T lymphocytes. AIDS Research and Human Retroviruses 20: 175–182.
Campbell, S., L.C. Burkly, H.X. Gao, et al. 2006. Proinflammatory effects of TWEAK/Fn14 interactions in glomerular mesangial cells. Journal of Immunology 176: 1889–1898.
Jakubowski, A., B. Browning, M. Lukashev, et al. 2002. Dual role for TWEAK in angiogenic regulation. Journal of Cell Science 115: 267–274.
Miyagawa, H., M. Yamai, D. Sakaguchi, et al. 2008. Association of polymorphisms in complement component C3 gene with susceptibility to systemic lupus erythematosus. Rheumatology (Oxford, England) 47: 158–164.
Chicheportiche, Y., L. Fossati-Jimack, S. Moll, et al. 2000. Down-regulated expression of TWEAK mRNA in acute and chronic inflammatory pathologies. Biochemical and Biophysical Research Communications 279: 162–165.
Baxter, F.O., P.J. Came, K. Abell, et al. 2006. IKKbeta/2 induces TWEAK and apoptosis in mammary epithelial cells. Development 133: 3485–3494.
Desplat-Jego, S., R. Creidy, S. Varriale, et al. 2005. Anti-TWEAK monoclonal antibodies reduce immune cell infiltration in the central nervous system and severity of experimental autoimmune encephalomyelitis. Clinical Immunology 117: 15–23.
Zheng, T.S., and L.C. Burkly. 2008. No end in site: TWEAK/Fn14 activation and autoimmunity associated-end-organ pathologies. Journal of Leukocyte Biology 84: 338–347.
Winkles, J.A. 2008. The TWEAK-Fn14 cytokine–receptor axis: discovery, biology and therapeutic targeting. Nature Reviews. Drug Discovery 7: 411–425.
Pradet-Balade, B., J.P. Medema, M. Lopez-Fraga, et al. 2002. An endogenous hybrid mRNA encodes TWE-PRIL, a functional cell surface TWEAK-APRIL fusion protein. EMBO Journal 21: 5711–5720.
Desplat-Jego, S., L. Feuillet, R. Creidy, et al. 2009. TWEAK is expressed at the cell surface of monocytes during multiple sclerosis. Journal of Leukocyte Biology 85: 132–135.
Nakayama, M., N. Kayagaki, N. Yamaguchi, et al. 2000. Involvement of TWEAK in interferon gamma-stimulated monocyte cytotoxicity. The Journal of Experimental Medicine 192: 1373–1380.
Kaplan, M.J., D. Ray, R.R. Mo, et al. 2000. TRAIL (Apo2 ligand) and TWEAK (Apo3 ligand) mediate CD4+ T cell killing of antigen-presenting macrophages. Journal of Immunology 164: 2897–2904.
Desplat-Je go, S., L. Feuillet, R. Creidy, et al. 2009. TWEAK is expressed at the cell surface of monocytes during multiple sclerosis. Journal of Leukocyte Biology 85: 132–135.
Chan, F.K., and M.J. Lenardo. 2002. Tumor necrosis factor family ligands and receptors in the immune system: targets for future pharmaceuticals. Drug News & Perspectives 15: 483–490.
Trouche, N., S. Wieckowski, W. Sun, et al. 2007. Small multivalent architectures mimicking homotrimers of the TNF superfamily member CD40L: delineating the relationship between structure and effector function. Journal of the American Chemical Society 129: 13480–13492.
Idriss, H.T., and J.H. Naismith. 2000. TNF alpha and the TNF receptor superfamily: structure–function relationship(s). Microscopy Research and Technique 50: 184–195.
Yamana, J., L. Santos, and E. Morand. 2009. Enhanced induction of LPS-induced fibroblast MCP-1 by interferon-gamma: involvement of JNK and MAPK phosphatase-1. Cellular Immunology 255: 26–32.
Martin, M., R.E. Schifferle, N. Cuesta, et al. 2003. Role of the phosphatidylinositol 3 kinase-Akt pathway in the regulation of IL-10 and IL-12 by Porphyromonas gingivalis lipopolysaccharide. Journal of Immunology 171: 717–725.
Richardson, C.E., T. Kooistra, and D.H. Kim. 2010. An essential role for XBP-1 in host protection against immune activation in C. elegans. Nature 463: 1092–1095.
Ruland, J. 2008. CARD9 signaling in the innate immune response. Annals of the New York Academy of Sciences 1143: 35–44.
Thornton, T.M., and M. Rincon. 2009. Non-classical p38 map kinase functions: cell cycle checkpoints and survival. International Journal of Biological Sciences 5: 44–51.
Colombara, M., V. Antonini, A.P. Riviera, et al. 2005. Constitutive activation of p38 and ERK1/2 MAPKs in epithelial cells of myasthenic thymus leads to IL-6 and RANTES overexpression: effects on survival and migration of peripheral T and B cells. Journal of Immunology 175: 7021–7028.
Khiem, D., J.G. Cyster, J.J. Schwarz, et al. 2008. A p38 MAPK-MEF2C pathway regulates B-cell proliferation. Proceedings of the National Academy of Sciences of the United States of America 105: 17067–17072.
Alvarado-Kristensson, M., and T. Andersson. 2005. Protein phosphatase 2A regulates apoptosis in neutrophils by dephosphorylating both p38 MAPK and its substrate caspase 3. Journal of Biological Chemistry 280: 6238–62344.
Stambe, C., D.J. Nikolic-Paterson, P.A. Hill, et al. 2004. p38 Mitogen-activated protein kinase activation and cell localization in human glomerulonephritis: correlation with renal injury. Journal of the American Society of Nephrology 15: 326–336.
Ohashi, R., T. Nakagawa, S. Watanabe, et al. 2004. Inhibition of p38 mitogen-activated protein kinase augments progression of remnant kidney model by activating the ERK pathway. American Journal of Pathology 164: 477–485.
Sallusto, F., C.R. Mackay, and A. Lanzavecchia. 2000. The role of chemokine receptors in primary, effector, and memory immune responses. Annual Review of Immunology 18: 593–620.
Nagasawa, M., Z. Yi, S. Imashuku, et al. 2008. Soluble TWEAK is markedly elevated in hemophagocytic lymphohistiocytosis. American Journal of Hematology 83: 222–225.
Garcia, G.E., Y. Xia, J. Harrison, et al. 2003. Mononuclear cell-infiltrate inhibition by blocking macrophage-derived chemokine results in attenuation of developing crescentic glomerulonephritis. American Journal of Pathology 162: 1061–1073.
Marks, S.D., S.J. Williams, and K. Tullus. 2008. Glomerular expression of monocyte chemoattractant protein-1 is predictive of poor renal prognosis in pediatric lupus nephritis. Nephrology, Dialysis, Transplantation 23: 3521–3526.
Shimizu, S., H. Nakashima, K. Masutani, et al. 2004. Anti-monocyte chemoattractant protein-1 gene therapy attenuates nephritis in MRL/lpr mice. Rheumatology (Oxford, England) 43: 1121–1128.
Ishida, H., T. Muchamuel, S. Sakaguchi, et al. 1994. Continuous administration of anti-interleukin 10 antibodies delays onset of autoimmunity in NZB/W F1 mice. The Journal of Experimental Medicine 179: 305–310.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhi-Chun, L., Qiao-Ling, Z., Zhi-Qin, L. et al. Tumor Necrosis Factor-Like Weak Inducer of Apoptosis (TWEAK) Mediates p38 Mitogen-Activated Protein Kinase Activation and Signal Transduction in Peripheral Blood Mononuclear Cells from Patients with Lupus Nephritis. Inflammation 35, 935–943 (2012). https://doi.org/10.1007/s10753-011-9396-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-011-9396-3