Abstract
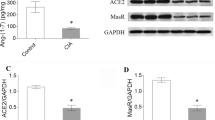
Angiotensin converting enzymes (ACE) and more recently discovered ACE-2 are important proteins involved in the renin–angiotensin system. The balance between ACE and ACE-2 is important for the regulation of blood pressure and electrolyte homeostasis. Inflammatory diseases like rheumatoid arthritis are associated with increased risk for cardiovascular complications. We studied the effect of inflammation on the expression levels of ACE and ACE-2 in two groups (n = 4/group) of adjuvant arthritis (AA) and healthy (control) rats. The AA group received 0.2 ml of 50 mg ml−1 of Mycobacterium butyricum suspended in squalene into the tail base. On day 12, rats were euthanized and their organs (hearts, liver, kidney, and intestine) were excised. The mRNA of ACE and ACE-2 were determined by real-time polymerase chain reaction. ACE and ACE-2 protein expression in rat heart was determined by Western blot. Inflammation resulted in 80% reduction of ACE-2 gene expression in rat heart. ACE-2/ACE expression ratio was significantly reduced from 0.7 ± 0.4 in control rats to 0.07 ± 0.09 in AA. Similarly, ACE-2/ACE protein expression ratio was also disrupted with a significant reduction in AA animals (6.7 ± 4.8 vs. 0.9 ± 05 in control and AA, respectively). ACE-2 has been found to provide negative feedback of renin–angiotensin system and protection of the heart and kidneys. Disruption of the balance between ACE and ACE-2 observed in inflammation may be, at least in part, involved in the cardiovascular complications seen in patients with inflammatory diseases.



Similar content being viewed by others
REFERENCES
Symmons, D.P., M.A. Jones, D.L. Scott, and P. Prior. 1998. Longterm mortality outcome in patients with rheumatoid arthritis: Early presenters continue to do well. The Journal of Rheumatology 25(6): 1072–1077.
Wallberg-Jonsson, S., H. Johansson, M.L. Ohman, and S. Rantapaa-Dahlqvist. 1999. Extent of inflammation predicts cardiovascular disease and overall mortality in seropositive rheumatoid arthritis. A retrospective cohort study from disease onset. The Journal of Rheumatology 26(12): 2562–71.
Kaplan, M.J. 2006. Cardiovascular disease in rheumatoid arthritis. Current Opinion in Rheumatology 18(3): 289–297.
Sakuta, T., Y. Morita, M. Satoh, N. Kashihara, and D.A. Fox. 2010. Involvement of the renin–angiotensin system in the development of vascular damage in a rat arthritis model: Efect of angiotensin receptor blockers. Arthritis and Rheumatism 62(5): 1319–1328.
Tipnis, S.R., N.M. Hooper, R. Hyde, E. Karran, G. Christie, and A.J. Turner. 2000. A human homolog of angiotensin-converting enzyme. Cloning and functional expression as a captopril-insensitive carboxypeptidase. The Journal of Biological Chemistry 275(43): 33238–43.
Crackower, M.A., R. Sarao, G.Y. Oudit, C. Yagil, I. Kozieradzki, S.E. Scanga, et al. 2002. Angiotensin-converting enzyme 2 is an essential regulator of heart function. Nature 417(6891): 822–828.
Dagenais, N.J., and F. Jamali. 2005. Protective effects of angiotensin II interruption: Evidence for antiinflammatory actions. Pharmacotherapy 25(9): 1213–1229.
Tikellis, C., C.I. Johnston, J.M. Forbes, W.C. Burns, M.C. Thomas, R.A. Lew, et al. 2004. Identification of angiotensin converting enzyme 2 in the rodent retina. Current Eye Research 29(6): 419–427.
Zhu, L.J., and S.W. Altmann. 2005. mRNA and 18S-RNA coapplication-reverse transcription for quantitative gene expression analysis. Analytical Biochemistry 345(1): 102–109.
Hanafy, S., N.J. Dagenais, W.F. Dryden, and F. Jamali. 2008. Effects of angiotensin II blockade on inflammation-induced alterations of pharmacokinetics and pharmacodynamics of calcium channel blockers. British Journal of Pharmacology 153(1): 90–99.
Danilczyk, U., U. Eriksson, G.Y. Oudit, and J.M. Penninger. 2004. Physiological roles of angiotensin-converting enzyme 2. Cellular and Molecular Life Sciences 61(21): 2714–2719.
Riviere, G., A. Michaud, C. Breton, G. VanCamp, C. Laborie, M. Enache, et al. 2005. Angiotensin-converting enzyme 2 (ACE2) and ACE activities display tissue-specific sensitivity to undernutrition-programmed hypertension in the adult rat. Hypertension 46(5): 1169–1174.
Gembardt, F., A. Sterner-Kock, H. Imboden, M. Spalteholz, F. Reibitz, H.P. Schultheiss, et al. 2005. Organ-specific distribution of ACE2 mRNA and correlating peptidase activity in rodents. Peptides 26(7): 1270–1277.
Lambert, D.W., M. Yarski, F.J. Warner, P. Thornhill, E.T. Parkin, A.I. Smith, et al. 2005. Tumor necrosis factor-alpha convertase (ADAM17) mediates regulated ectodomain shedding of the severe-acute respiratory syndrome-coronavirus (SARS-CoV) receptor, angiotensin-converting enzyme-2 (ACE2). The Journal of Biological Chemistry 280(34): 30113–30119.
Sakuta, T., Y. Morita, M. Satoh, D.A. Fox, and N. Kashihara. 2010. Involvement of the renin–angiotensin system in the development of vascular damage in a rat model of arthritis: Effect of angiotensin receptor blockers. Arthritis and Rheumatism 62(5): 1319–1328.
Thomas, M.C., R.J. Pickering, D. Tsorotes, A. Koitka, K. Sheehy, S. Bernardi, et al. 2010. Genetic Ace2 deficiency accentuates vascular inflammation and atherosclerosis in the ApoE knockout mouse. Circulation Research 107(7): 888–897.
Danilczyk, U., and J.M. Penninger. 2006. Angiotensin-converting enzyme II in the heart and the kidney. Circulation Research 98(4): 463–471.
Mayo, P.R., K. Skeith, A.S. Russell, and F. Jamali. 2000. Decreased dromotropic response to verapamil despite pronounced increased drug concentration in rheumatoid arthritis. British Journal of Clinical Pharmacology 50(6): 605–613.
Daneshtalab, N., R.Z. Lewanczuk, A. Russell, and F. Jamali. 2004. Rheumatoid arthritis does not reduce the pharmacodynamic response to valsartan. Journal of Clinical Pharmacology 44(3): 245–252.
Duggan, K.A., F.A. Mendelsohn, and N.R. Levens. 1989. Angiotensin receptors and angiotensin I-converting enzyme in rat intestine. The American Journal of Physiology 257(4 Pt 1): G504–G510.
Byrnes, J.J., S. Gross, C. Ellard, K. Connolly, S. Donahue, and D. Picarella. 2009. Effects of the ACE2 inhibitor GL1001 on acute dextran sodium sulfate-induced colitis in mice. Inflammation Research 58(11): 819–827.
Conflict of Interest
None
Financial Support
The study was supported by the Canadian Institute of Health Research (CIHR).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hanafy, S., Tavasoli, M. & Jamali, F. Inflammation Alters Angiotensin Converting Enzymes (ACE and ACE-2) Balance in Rat Heart. Inflammation 34, 609–613 (2011). https://doi.org/10.1007/s10753-010-9269-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-010-9269-1