Abstract
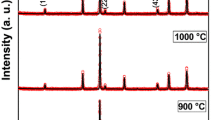
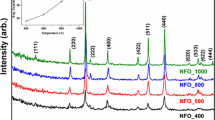
NiFe2O4 nanoparticles have been synthesized by co-precipitation method at 145°C in N2 atmosphere using ethylene glycol as solvent and capping agent. This gives the promising synthesis route for nanoparticles at low temperature. The as-synthesized NiFe2O4 is subsequently heated at 400°C, 500°C, 700°C and 800°C. Crystallite size increases with the heat treatment temperature. The heat treatment temperature has direct effect on the electron paramagnetic resonance and intrinsic magnetic properties. The room temperature Mössbauer spectrum of the 800°C heated sample shows the two sextets pattern indicating that the sample is ferrimagnetic and Fe3 + ions occupy both tetrahedral and octahedral sites of spinel structure.
Similar content being viewed by others
References
Gajbhiye, N.S., Bhattacharyya, S., Balaji, G., Ningthoujam, R.S., Das, R.K., Basak, S., Weissmüller, J.: Mössbauer and magnetic studies of MFe2O4 (M = Co, Ni) nanoparticles. Hyperfine Interact. 165, 153–159 (2005)
Gajbhiye, N.S., Ningthoujam, R.S., Weissmuller, J.: Mössbauer study of nanocrystalline ε-Fe3−x Co x N system. Hyperfine Interact. 156, 51–56 (2004)
Cao, X., Gu, L.: Spindly cobalt ferrite nanocrystals: preparation, characterization and magnetic properties. Nanotechnology 16, 180–185 (2005)
Lee, J.-G., Lee, H.M., Kim, C.S., Young-Jei, O.: Magnetic properties of CoFe2O4 powders and thin films grown by a sol-gel method. J. Magn. Magn. Mater. 177, 900 (1998)
Davies, K.J., Wells, S., Upadhyay, R.V., Charles, S.W., O’Grady, K., Hilo, M.E., Meaz, T., Mørup, S.: The observation of multi-axial anisotropy in ultrafine cobalt ferrite particles used in magnetic fluids. J. Magn. Magn. Mater. 149, 14–18 (1995)
Ding, J., Miao, W.M., McCormick, P.G., Street, R.: Mechanochemical synthesis of ultrafine Fe powder. Appl. Phys. Lett. 67, 3804–3806 (1995)
Goya, G.F., Rechenberg, H.R., Jiang, J.Z.: Structural and magnetic properties of ball milled copper ferrite. J. Appl. Phys. 84, 1101–1108 (1998)
Costa, A.C.F.M., Tortella, E., Morelli, M.R., Kaufman, M., Kiminami, R.H.G.A.: Effect of heating conditions during combustion synthesis on the characteristics of Ni0.5Zn0.5Fe2O4 nanopowders. J. Mater. Sci. 37, 3569–3572 (2002)
Prasad, S.: Solid state reactivity, characterization and anomalous magnetic behaviour of nanocrystalline spinel ferrite particles synthesized by citrate precursor technique. Dissertation, Indian Institute of Technology, Kanpur, India (1997)
Ningthoujam, R.S., Sudarsan, V., Kulshreshtha, S.K.: SnO2:Eu nanoparticles dispersed in silica: a low temperature synthesis and photoluminescence study. J. Lumin. 127, 747–756 (2007)
Ningthoujam, R.S., Sudarsan, V., Godbole, S.V., Kienle, L., Kulshreshtha, S.K., Tyagi, A.K.: SnO2:Eu3 + nanoparticles dispersed in TiO2 matrix: improved energy transfer between semiconductor host and Eu3 + ions for the low temperature synthesized samples. Appl. Phys. Lett. 90, 173113–3 (2007)
Ningthoujam, R.S., Gajbhiye, N.S., Ahmed, A., Umre, S.S., Sharma, S.J.: Re-dispersible Li + and Eu3 + co-doped ZnO nano-dumb-bell: luminescence and EPR studies. J. Nanosci. Nanotechnol. 8, 3059–30662 (2008)
Gajbhiye, N.S., Ningthoujam, R.S., Ahmed, A., Panda, D.K., Umre, S.S., Sharma, S.J.: Re-dispersible Li + and Eu3 + co-doped CdS nanowires: luminescence studies. Pramana 70, 313–321 (2008)
Pascal, C., Pascal, J.L., Farier, F., Moubtassium, M.L.E., Payen, C.: Electrochemical synthesis for the control of gamma-Fe2O3 nanoparticle size. Morphology, microstructure, and magnetic behavior. Chem. Mater. 11, 141–147 (1999)
Sidorov, S.N., Bronstein, L.M., Davankov, V.A., Tsyurupa, M.P., Solodovnikov, S.P., Valetsky, P.M., Wilder, E.A., Spontak, R.J.: Cobalt nanoparticle formation in the pores of hyper-cross-linked polystyrene: control of nanoparticle growth and morphology. Chem. Mater. 11, 3210–3215 (1999)
Koksharov, Y.A., Pankratov, D.A., Gubin, S.P., Kosobudsky, I.D., Beltran, M., Khodorkovsky, Y., Tishin, A.M.: Electron paramagnetic resonance of ferrite nanoparticles. J. Appl. Phys. 89, 2293–2298 (2001)
Cullity, B.D.: Introduction to Magnetic Materials. Addison-Wesley, London (1972)
Mitra, S., Mandal, K., Kumar, P.A.: Temperature dependence of magnetic properties of NiFe2O4 nanoparticles embedded in SiO2 matrix. J. Magn. Magn. Mater. 306, 254–259 (2006)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Umare, S.S., Ningthoujam, R.S., Sharma, S.J. et al. Mössbauer and magnetic studies on nanocrystalline NiFe2O4 particles prepared by ethylene glycol route. Hyperfine Interact 184, 235–243 (2008). https://doi.org/10.1007/s10751-008-9796-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10751-008-9796-4