Abstract
Chironomidae and Cladocera are useful palaeoindicators to describe long-term dynamics in biodiversity of freshwater aquatic communities. However, palaeoecological studies relying on these aquatic invertebrates have generally focused on post-glacial lakes, with relatively few applications to floodplain water bodies such as palaeo-oxbow lakes. In this study, zone-level (stratigraphic) trends in Chironomidae and Cladocera assemblage composition were analysed from a small palaeo-oxbow lake in a Central European river valley of Poland. Summer air temperature and hydrological conditions were found to be the main drivers of Cladocera and Chironomidae populations, causing a cascade effect in assemblage composition. At the local habitat scale, there were autecological differences in both taxonomic groups reflecting different responses to abiotic and biotic factors. Specifically, Cladocera responded distinctly to plant vegetation and fish predation during the Late Glacial lake phase, whereas Chironomidae were more sensitive to paludification processes and disappeared in the Northgrippian dry phase of the mire. Both groups indicated fluvial activity of the River Grabia, although the response was different. Also, species richness was higher in the more taxonomically diverse Chironomidae compared to Cladocera. As both groups responded to the same factors in a different way, it is recommended that they should be used in parallel in palaeoecological research studies.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Several palaeoindicators have been employed to describe the long-term dynamics in the biodiversity of freshwater aquatic communities under environmental change (Birks & Birks, 2004). Amongst these palaeoindicators are Chironomidae and Cladocera, which live mostly in lakes, ponds and streams, and are distributed up to high latitudes and altitudes (Porinchu & MacDonald, 2003; Szeroczyńska & Sarmaja-Korjonen, 2007). The ecological preferences of these two taxonomic groups are relatively well known (Armitage et al., 1995; Vallenduuk & Moller Pillot, 2007; Błędzki & Rybak, 2016) and make them good indicators to infer palaeoenvironmental conditions of lakes, including their trophic status, water-level fluctuations, pH and fish abundance (e.g. Korhola & Rautio, 2001; Brooks et al., 2007; Gąsiorowski & Kupryjanowicz, 2009; Davidson et al., 2010). Chironomidae are also very useful in the reconstruction of mean July air temperatures (Brooks, 2006), and Cladocera are an important proxy for quantitative hydroclimatic reconstructions of Late Glacial (e.g. Luoto et al., 2011; Zawiska et al., 2014) and Holocene palaeolakes (e.g. Nevalainen et al., 2012; Nevalainen & Luoto, 2017). Also, Cladocera have been used successfully in mean July air temperature transfer function and water depth inference models (Nevalainen, 2011; Nevalainen et al., 2012; Pawłowski, 2017). In this regard, the use of appropriate statistical methods for the reconstruction of palaeoenvironmental conditions has proved an important component of palaeolimnological studies (e.g. ter Braak & Juggins, 1993; Birks, 1995; Korhola et al., 2000; Birks et al., 2012; Nevalainen & Luoto, 2017).
Whilst palaeoecological studies relying on Chironomidae and Cladocera have generally focused on post-glacial lakes, relatively few investigations based on these aquatic invertebrates have dealt with floodplain water bodies such as palaeo-oxbow lakes. This is a notable knowledge gap, as paludification is a common process that in Europe has intensified since the Greenlandian (lower age of the Holocene), with river valleys in Central Europe undergoing transformation from the Late Glacial (end of Pleistocene) to the Meghalayan age (uppermost Holocene) and with valley stagnant waters turning from lakes in the Younger Dryas to mires in the Northgrippian (middle age of the Holocene) (Vandenberghe, 2003; Starkel et al., 2013).
The present study investigates the responses of Chironomidae and Cladocera in assemblage composition and abundance to regional and habitat-related environmental factors in a palaeo-habitat that turned from oxbow to mire. It is anticipated that the outcomes of the present study will contribute to the use of Chironomidae and Cladocera as proxies in palaeoenvironmental and palaeoclimatic reconstructions based on data from sediments of lakes, flood plains and valley mires. A novel analytical approach is also implemented that can be applied more widely in palaeolimnological studies.
Study area
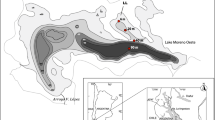
The Pawłowa site (51°30′19″ N; 19°19′59″ E) is located in the Łódź region of Central Poland (Fig. 1a) at an elevation of 185 m a.s.l. in the middle part of the course of the River Grabia (Fig. 1b) and in the marginal part of a valley floor that occupies a slight depression within the flood plain (Fig. 1c). The catchment of the Pawłowa mire is about 7.7 ha and is presently covered for the major part by an alder swamp and meadows. An area surrounding the Grabia valley consists of glacial and glaciofluvial deposits that accumulated during the Saalian glaciations, especially of the Warthanian Cold Stage (the last one to cover the Łódź region: Klatkowa, 1984), and fluvial sediments (Late Glacial and Holocene alluvium) that fill the main part of the valley. General descriptions of the geological and geomorphological characteristics of the study site, including lithology and chronology of sediments, are provided by Pawłowski et al. (2016a) (Fig. 2).
Location of the Pawłowa site: (a) Central Poland; (b) Southern part of the Łódź region; (c) Study area with indication of organic thickness of the Pawłowa mire; (d) Geological cross-section of the River Grabia valley (after Pawłowski et al., 2016a)
Lithology, chronostratigraphy and representative plant and animal macrofossils and geochemical elements from the Pawłowa site (after Pawłowski et al., 2016a). Lithology: 1 = fluvial sands; 2 = gyttja; 3 = mid-decomposed peat; 4 = well-decomposed peat
Currently, the Łódź region has a temperate climate influenced by westerly airflow from the Atlantic and by eastern Eurasian high-pressure cells (Woś, 1999). Mean annual temperature in the study area is 7.7°C, mean temperature of the warmest month (July) 18°C and of the coldest month (January) − 3.3°C. Average annual precipitation is 590 mm, ranging from 438 to 937 mm (Kłysik, 2001).
Materials and methods
Sample collection and processing
A 450-cm sediment core from Pawłowa consisting of gyttja and various types of peat was taken in the southern part of the mire (Fig. 1c, d), where the thickest organic deposits were found, using a manual Russian Corer with a diameter of 5 cm and a length of 50 cm. The chronology of the core was determined based on AMS 14C dating. Pollen biostratigraphy was based on the results of radiocarbon dating analysis of sediment deposits and on the sequence of eight local pollen assemblage zone points from the Oldest Dryas to the Subatlantic (upper Meghalayan) periods (Fig. 2).
Chironomidae subfossils were analysed at 8 cm resolution. Preparation methods for larval head capsules (HCs) followed Brooks et al. (2007), with sediments passed through a 63 µm sieve. As HC concentrations in the upper core sequence were low, kerosene flotation was used (after Rolland & Larocque, 2007). Taxon identification followed mainly keys by Klink & Moller Pillot (2003) and Brooks et al. (2007), and ecological preferences of the identified taxa were based on Brooks et al. (2007), Vallenduuk & Moller Pillot (2007) and Moller Pillot (2009, 2013). [The reference collection is deposited at the Department of Invertebrate Zoology and Hydrobiology, University of Lodz.]
Cladocera subfossils were analysed at 4 cm resolution. One cm3 of fresh sediment was prepared in the laboratory following standard procedures (Frey, 1986), with each sample boiled in a 10% KOH solution for 20 min and stirred using a magnetic stirrer to remove humic matter, and later treated with HCl to eliminate carbonates. The residue was then washed and sieved (50 µm) and diluted in 10 cm3 of distilled water. One tenth of a millilitre of solution was then used for each microscope slide. The extracted remains were identified after Szeroczyńska & Sarmaja-Korjonen (2007), Van Damme & Dumont (2008), Van Damme et al. (2010) and Faustova et al. (2011). All skeletal elements (i.e. head shield, shell, post-abdomen, claw, ephippium) in each sample were counted and used to compute the number of individuals per cm3 of fresh sediment. Classification of Cladocera habitat preferences followed Flössner (1972, 2000), Bjerring et al. (2009) and Błędzki & Rybak (2016).
Statistical analysis
The number of zones (and sub-zones) in the biostratigraphic sequence was determined after Bennett (1996), using optimal sum-of-squares partitioning (Birks & Gordon, 1985; Birks, 1986) using the ZONE software (Lotter and Juggins, 1991), with testing for statistical significance based on the broken-stick model (MacArthur, 1957) using the BSTICK software (unpublished software). To identify overall trends in assemblage composition, Detrended Correspondence Analysis (DCA) was performed on percentage square-root transformed data using CANOCO 4.5 (ter Braak & Smilauer, 2002), with detrending by segments, non-linear re-scaling, and down-weighting of rare species. Stratigraphic diagrams were created using C2 (Juggins, 2007). Rarefaction with ES(n) = 15 for Chironomidae and with ES(n) = 20 for Cladocera was computed using PRIMER 6 software (Clarke & Gorley, 2006).
Trends in the relative abundance and composition of Chironomidae and Cladocera guilds (i.e. habitat indicators) and taxa were further analysed by Min–max Autocorrelation Factor Analysis (MAFA: Zuur et al., 2007) to provide a more detailed description at the guild and (representative) taxon level. MAFA is a statistical method for estimating trends in multivariate (time) series related to principal component analysis with applications in ecology (Vilizzi, 2012; Whiterod et al., 2015; Shojaei et al., 2016; Doray et al., 2018) but not yet in palaeoecological studies. In MAFA, axes have maximum autocorrelation with time lag k. The first MAF axis represents the main trend in the data associated with the highest autocorrelation at lag 1, the second MAF axis at lag 2, and so forth, in decreasing order of importance. Cross-correlations (or canonical correlations) between variables (i.e. guilds and taxa) and trends (as indicated by the MAF axes) are then computed and tested for significance. Given that in MAFA the number of ‘points’ (in the present case, the zones and sub-zones under study) is constrained to be larger than the number of variables, a maximum of five guilds or taxa (i.e. the most abundant) could be analysed for both Chironomidae and Cladocera. Following square-root transformation (Chironomidae) and double-square-root transformation (Cladocera) and centring of the relative abundance (count) data, the first two MAF axes were estimated. Canonical correlations of the individual variables (i.e. guilds and taxa within guilds) with the two MAF axes of variation were then tested for significance (α = 0.05). Analyses were carried out in Brodgar v. 2.5.7 (http://www.brodgar.com).
Results
Stratigraphy
Chironomidae
Sampling material of the Pawłowa sequence contained altogether 4460 HCs from 73 taxa of Chironomidae identified at least to the genus level, 619 HCs of unidentified Tanytarsini (excluding Corynocera ambigua Zetterstedt, 1837), 166 of Ceratopogonidae, and three of Simuliidae. Chironomidae stratigraphy was divided into six zones (Fig. 3).
Ch1 (450–408 cm): Oxbow establishment – Cold-adapted taxa were present, with Corynocera ambigua representing 60% of the community at the top of this zone. Notably, at 444 cm a singular HC of Lipiniella spp. Shilova, 1961 (a taxon usually typical of large, sandy rivers) was found.
Ch2 (408–344 cm): First well-developed lake phase – The relative abundance of Corynocera ambigua increased to 80% of the total at the top of this zone. Subdominant taxa were represented by a few mesotrophic and a wider group of warm-adapted eutrophic taxa mainly represented by Chironomus plumosus-type (sensu Brooks et al. 2007 after Chironomus (Chironomus) plumosus (Linnaeus, 1758)) and Dicrotendipes nervosus-type (sensu Brooks et al. 2007 after Dicrotendipes nervosus (Staeger, 1839)).
Ch3 (344–256 cm): Second well-developed lake phase – The relative abundance of Corynocera ambigua declined at the top of this zone from 70% to about 10% of the total and was replaced by a range of warm-adapted eutrophic taxa again mainly represented by Chironomus plumosus-type and Dicrotendipes nervosus-type.
Ch4 (256–208 cm): Water level decrease – Most of the limnetic bottom-dweller taxa dominant in zones Ch1–Ch3 disappeared and were replaced by the shallow-water phytophiles such as Lauterborniella agrayloides (Kieffer, 1911) and Paratanytarsus penicillatus-type (sensu Brooks et al., 2007 after Paratanytarsus penicillatus (Goetghebuer, 1928)), by Ceratopogonidae and by amphibious or semi-terrestrial taxa associated with fens (e.g. Limnophyes spp. Eaton, 1875 and Parametriocnemus-Paraphaenocladius (sensu Brooks et al., 2007 after Parametriocnemus Goetghebuer, 1932 and Paraphaenocladius Thienemann, 1924)). At depths of 248 cm and 216 cm, individual HCs of rheophile taxa associated with lowland streams (i.e. Euryhapsis cilium-type (after Euryhapsis cilium Oliver, 1981) and Simuliidae) were present.
Ch5 (208–152 cm): Wet semi-terrestrial conditions – The limnetic taxa in zone Ch4 were almost completely replaced by a wide range of amphibious taxa typical of fens and bogs, including Limnophyes spp., Krenopelopia Fittkau, 1962, Metriocnemus fuscipes-type (sensu Brooks et al., 2007 after Metriocnemus (Metriocnemus) fuscipes (Meigen, 1818)) and Ceratopogonidae. Subfossils of rheophile Chironomidae typical of small streams and brooks (i.e. Paracricotopus Brundin, 1956 and Simuliidae) also appeared frequently throughout this zone.
Ch6 (152–0 cm): Dry terrestrial conditions – The number of specimens was overall very low. Besides Ceratopogonidae, a few amphibious taxa were identified.
Cladocera
Twenty Cladocera taxa belonging to four families (Bosminidae, Chydoridae, Daphniidae and Sididae) were identified. Cladocera stratigraphy was divided into four zones (Fig. 4).
Cl1 (450–344 cm): Initial stage of oxbow-lake development – The most abundant taxa were represented by the macrophyte/sediment associated Alona affinis Leydig, 1860, Chydorus sphaericus (O. F. Müller, 1776) and Coronatella rectangula (Sars, 1861), and macrophyte-associated Acroperus harpae (Baird, 1835) and Eurycercus (Eurycercus) lamellatus (O. F. Müller, 1776) were also abundant. Pelagic taxa including Bosmina (Bosmina) longirostris (O. F. Müller, 1776), Daphnia (Daphnia) pulex Leydig, 1860 and Eubosmina spp. Seligo, 1900 also occurred at high densities, whereas sediment-associated taxa were mainly represented by Pleuroxus (Pleuroxus) uncinatus Baird, 1850.
Cl2 (344–232 cm): Final stage of oxbow-lake development and transformation to mire – This zone was divided into two sub-zones. The first sub-zone Cl2a (344–300 cm), was characterised by an increase (especially at the beginning) in the pelagic pelagic Cladocera Eubosmina spp., which represented > 40% of the specimens. Macrophyte/sediment-associated taxa including Alona affinis, Chydorus sphaericus and Coronatella rectangula were also quite abundant, and the macrophyte-associated Graptoleberis testudinaria (Fischer, 1848) and Sida crystalina (O. F. Müller, 1776) appeared for the first time, whereas sediment-associated taxa were still rare. The second sub-zone Cl2b (300–232 cm), was characterised by the highest frequency and diversity of Cladocera in the whole core, which exceeded more than 13 taxa and 7000 specimens per cm3. The dominant littoral macrophyte/sediment-associated taxa included Chydorus sphaericus and Coronatella rectangula, though littoral sediment-associated taxa and pelagic forms were still present, and sediment-associated Leydigia acanthocercoides (Fischer, 1854) occurred for the first time. At the end of this sub-zone, macrophyte-associated Alona guttata Sars, 1862 also occurred for the first time.
Cl3 (232–122 cm): Initial mire development – There were fluctuations in both frequency and diversity of Cladocera, with the frequency of pelagic forms such as Daphnia pulex-group Leydig, 1860 varying from 10 to 0%. There was also an increase in macrophyte/sediment-associated and macrophyte-associated taxa including Alona guttata, Alona affinis and Alonella excisa (Fischer, 1854), whereas sediment associated taxa were dominated by Pleuroxus (Pleuroxus) trigonellus (O. F. Müller, 1776) and Pleuroxus uncinatus Baird, 1850.
Cl4 (122–0 cm): Final mire development – This zone was divided into two sub-zones. The first sub-zone Cl4a (122–74 cm), included mainly Alona affinis and Chydorus sphaericus, with sporadic occurrence of Alona guttata, Coronatella rectangula and macrophyte-associated Alonella excisa, with an overall low frequency and diversity of Cladocera. The second sub-zone Cl4b (74–0 cm), was dominated by the macrophyte/sediment-associated Chydorus sphaericus.
Trends in assemblage composition and relative abundance
For Chironomidae, the first DCA axis explained 17.9% of the cumulative percentage variance in the data (Fig. 3). After an increase from Ch1 to Ch2, values of the first axis of variation were high and did not change from Ch2 until the end of Ch3. At Ch4, values of the first axis decreased through to the middle of Ch6, and then sharply increased towards surface level. For Cladocera, the first DCA axis explained 30.2% of the cumulative percentage variance in the species data (Fig. 4). Along this axis, the highest values were achieved at Cl1 and Cl2, whereas at Cl2b values started to decline and were lowest at Cl3, and then increased moderately at Cl4. The first axis displayed peaks from the Oldest Dryas to the early Younger Dryas, reflecting the oxbow-lake development.
Species richness derived from rarefaction revealed different trends for Chironomidae and Cladocera. Chironomidae showed high rarefaction at Ch1, followed by a decrease to lower values at the Ch2/Ch3 boundary, and then by an increase at the Ch3/Ch4 boundary, which was followed by relatively high values at Ch4 (Fig. 3). Afterwards, at Ch5 and Ch6 the relative abundance was too small to count rarefaction for Chironomidae. Whereas, for Cladocera rarefaction could be computed for all four zones due to their higher relative abundance (Fig. 4). The highest values were identified at the bottom part of the sequence, increasing from Cl1 to Cl2b, and then decreasing at the Cl3/Cl4a boundary to reach the lowest value (0) at Cl4b and finally to increase slightly again.
For Chironomidae (noting that rheophiles were excluded from MAFA due to low relative abundances), there was a strong correlation with the first axis of variation of the oligotrophic, mesotrophic and eutrophic guilds (statistically significant for the last two) due to an overall decrease in relative abundance starting from Ch4 through to Ch6 relative to Ch1–Ch3 (Fig. 5). At the guild level, among the most abundant oligotrophic and cold-adapted morphotypes Chironomus anthracinus-type (sensu Brooks et al. 2007 after Chironomus (Chironomus) anthracinus Zetterstedt, 1860) and Corynocera ambigua were strongly correlated with the first axis (the former taxon also significantly) due to a sharp decrease in relative abundance starting from Ch4. And a similar trend was revealed for the most abundant mesotrophic and eutrophic taxa, though significantly only for the mesotrophic Microtendipes pedellus-type (sensu Brooks et al. 2007 after Microtendipes pedellus (De Geer, 1776)). Conversely, no significant and clear trends were detected across the phytophile, rheophile and eurytopic guilds (Fig. 6). However, at the guild level the phytophile Glyptotendipes severini-type (sensu Brooks et al. 2007 after Glyptotendipes subg. Caulochironomus Heyn, 1993) (significantly), Ablabesmyia Johannsen, 1905 and Paratanytarsus penicillatus-type were highly correlated with the first axis due to a peak in relative abundance between Ch3 and Ch4, whereas Monopelopia tenuicalcar (Kieffer, 1918) was significantly correlated with the second axis as a result of a peak at Ch5.
Min-max autocorrelation factor (MAF) trends (left panels; solid line for Axis 1, dashed line for Axis 2) and corresponding canonical correlations with MAF axes 1 and 2 (central and right panels, respectively) for the abundance of three Chironomidae guilds and the most representative taxa therein. Trends associated with significant (α = 0.05) canonical correlations with MAF axis 1 or 2 are highlighted in black with the corresponding guild or taxon marked with an asterisk. See also Fig. 3
Same for the abundance of three Chironomidae guilds and the most representative taxa therein. See also Fig. 3
For Cladocera (Fig. 7), all guilds were highly correlated with the first MAFA axis of variation, and significantly so for the macrophyte associated and the pelagic guilds due to an overall decreasing trend starting from Cl3. This trend was overall reflected at the guild level, except for the sediment-associated, which were mainly represented by Pleuroxus trigonellus (O. F. Müller, 1785) and Pleuroxus uncinatus that significantly decreased in relative abundance only at Cl4. Finally, the macrophyte-associated guild was mainly represented by Acroperus harpae and Alonella nana (Baird, 1850), the macrophyte/sediment-associated by Alona affinis and Coronatella rectangula, and the pelagic by Eubosmina spp. Seligo, 1900 and Daphnia pulex-group.
Same for the abundance of four Cladocera guilds and the most representative taxa therein. See also Fig. 4
Discussion
Chironomidae
Mean summer temperature and water trophic status are known to be main drivers of Chironomidae community composition and relative abundance in temperate climates (Brodersen & Quinlan, 2006; Brooks, 2006), though some taxa have specific preferences for certain mesohabitats, hence eluding general classification (Brooks et al., 2007). In the present study, the overall change in Chironomidae communities reflected the climate-related Late Glacial–Holocene transition from oxbow to mire environment of Pawłowa.
Overall, the taxonomic composition of Chironomidae communities suggests that during the Late Glacial Pawłowa was a mesotrophic water body, with all trophic groups of taxa still finding appropriate habitat conditions in the different zones of the lake. Although the trophic status of Pawłowa lake in the Late Glacial was strongly related to temperature, it also depended on climate-related variables such as denudation processes and flooding. In this respect, in-wash of nutrients and inorganic particles to Pawłowa lake has been confirmed by geochemical, palaeobotanical and Cladocera proxies (Pawłowski et al., 2016a), and similar processes have been reconstructed from the Younger Dryas sequence of Lake Świerczyna in the Grabia valley (Pawłowski et al., 2015a), where mean annual temperature was lower during the Late Glacial compared to the Holocene. This explains why the first Chironomidae generation in Pawłowa lake has been found to include cold-adapted taxa despite high summer temperatures (Brodersen & Lindegaard, 1999; Kołaczek et al., 2018b). Whereas, low annual temperatures prevented fast eutrophication of the small Pawłowa pond, when nutrients could only be assimilated by the biota during the short and warm summers, with the result that Pawłowa lake maintained rich benthic assemblages during the Late Glacial. Paludification then started at the end of the Younger Dryas and occurred in parallel with eutrophication but caused a decline in most species from the interstadial generation. Palaeobotanical proxies indicate high trophic status and slightly alkaline conditions in Pawłowa lake during the early Younger Dryas, although during its second part and at the onset of the Holocene the lake transitioned to a nutrient-poor, acidified water body (Pawłowski et al., 2016a).
Amongst the groups of taxa associated with specific mesohabitats, phytophile taxa revealed a clear pattern in assemblage composition in the present study (Fig. 7). Plant macrofossil analysis (Pawłowski et al., 2016a) indicates the development of rich aquatic vegetation (i.e. Chara spp. Linnaeus, 1753 and Potamogeton spp. Linnaeus, 1753) during the Younger Dryas and of amphibious plant vegetation (Carex spp. Linnaeus, 1753) during the Younger Dryas–Holocene transition (Fig. 2). Since the Greenlandian, the lake transformed into a mire with ample Bryales communities (Fig. 2), which could maintain abundant Chironomidae populations in the valley wetlands (Brusven et al., 1990). In this regard, Monopelopia tenuicalcar has often been recorded in peat sections of palaeolakes of the region (e.g. Płóciennik et al., 2011; Pawłowski et al., 2015a; Słowiński et al., 2016), and this is known to be a species characteristic of shallow, acid and overgrown peat pools elsewhere in Europe (Vallenduuk & Moller Pillot, 2007).
Rheophile taxa, albeit rare, suggest episodes of connection of the Pawłowa lake to the main River Grabia channel (Howard et al., 2010). Thus, Lipiniella spp. included taxa typical of large rivers with a sandy bottom and low silt/clay content, and the presence of a singular head capsule of Lipiniella spp. in the lowest Pawłowa layers suggests that, during the Oldest Dryas, the River Grabia channel was large, sandy and meandering. The Lipiniella spp. head capsule detected in the present study could belong to a specimen in-washed to Pawłowa lake from the adjacent river during a flooding episode, and a small population of this taxon could have developed in the lake if habitat conditions resembled those of the river channel (Moller Pillot, 2009; Klukowska et al., 2011). The second generation of rheophile guilds appeared in Pawłowa at Younger Dryas–Holocene transition, with Simuliidae clearly associated with small–medium streams (Bass, 1998; Palmer et al., 2002). The parallel presence of a few rheophile species in the same zones indicates higher river activity and a periodical connection of the River Grabia to the Pawłowa mire during the Greenlandian. The most prominent floods were recorded in the Grabia valley during the Younger Dryas, when major flood flows are estimated to have been at least five times larger than present-day ones (Pawłowski et al., 2015a, 2016b), and higher river activity in the Younger Dryas and the Greenlandian has also been recorded from other places of the study region (Starkel et al., 2013; Kittel et al., 2016). Also, during the Greenlandian the Pawłowa mire experienced phases of substantial floods from the River Grabia, which however had different and less prominent sedimentological impact than the floods recorded from the Younger Dryas layers (Pawłowski et al., 2016a). The ‘Preboreal oscillation’ cooling then caused increased climate instability and led to a higher rate of extreme events including floods; whereas, the presence of more developed riparian and swamp vegetation might have limited the impact of floods on the oxbow lakes of central European valleys (Kołaczek et al., 2018a). Research in this regard has shown that riparian wetlands freely connected to the main channel are characterised by a higher biodiversity and greater biomass of macrobenthos (Pan et al., 2012), and that reconnection of an oxbow to the main river channel may cause an increase in Chironomidae relative abundance and benthic community diversity (Obolewski & Glińska-Lewczuk, 2011). Therefore, stronger fluvial activity of the River Grabia during the Younger Dryas and the early Greenlandian could have prolonged the presence of more diverse communities, including fully aquatic taxa even if not abundant. Starting from the late Greenlandian, when river activity decreased, aquatic species were replaced solely by semi-terrestrial Chironomidae taxa.
Despite the absence of trends in amphibious guilds (likely due to low subfossil densities), the stratigraphic data of the present study clearly revealed that amphibious Chironomidae almost exclusively dominated in the Holocene peat sequence of the Pawłowa mire. This is not surprising, as the Greenlandian oxbow paludified to a semi-terrestrial habitat, and semi-terrestrial assemblages are known to occur generally at low to very low subfossil densities (e.g. Pawłowski et al., 2015a; Kittel et al., 2016), with Chironomidae being more typical of shallow lakes (Whitehouse et al., 2008). Starting from the Northgrippian, seasonal peat desiccation and decomposition negatively influenced the preservation of Chironomidae subfossils, and even if terrestrial taxa were present, their head capsules were partly decomposed during the Northgrippian and Meghalayan.
Cladocera
Changes in Cladocera composition and relative abundance are generally related to water level fluctuations, with the formation and functioning of lakes on flood plains being dependent on the hydrological regime of the associated rivers combined with physical and chemical parameters (Amoros & Jacquet, 1987; Korhola & Rautio, 2001). There are also several environmental variables (e.g. summer water temperature, fish predation, macrophytes, nutrients, and sediment in-wash) that have been identified as important in influencing these aquatic crustacean assemblages (Medley & Havel, 2007; Schöll & Kiss, 2008; Vadadi-Fülöp, 2013; Pawłowski et al., 2015b).
Pelagic and macrophyte-associated taxa had a peak in relative abundance from the Oldest Dryas to the early Younger Dryas after which they declined. The decrease in the relative abundance of these taxa may be related to a systematic water level drop with transition of Pawłowa from lake to mire. The oligo/mesotrophic conditions during the first stage of Pawłowa lake also would have favoured the above-mentioned taxa, which however are characterised by a wide range of habitat preferences (Duigan, 1992). For example, Acroperus harpae and Alonella nana can occur in the rheopelagial as potamoplankton (Robertson, 1990; Błędzki & Rybak, 2016), and Acroperus harpae can act as a ‘pioneer’ taxon with early immigrants following ice retreat (Hofmann, 2000; Pawłowski, 2011; Błędzki & Rybak, 2016). Therefore, a gradual drop in dominance in both guilds may also indirectly reflect climate changes in the River Grabia basin with increased summer water temperatures.
Amongst the taxa associated with sediments, only two species (i.e. Pleuroxus trigonellus and Pleuroxus uncinatus) revealed a clear pattern in the assemblages of the Pawłowa palaeolake with relatively high dominance from the Oldest Dryas to the early Younger Dryas. The high dominance of these benthic taxa has been correlated to the geochemical characteristics of Pawłowa (Pawłowski et al., 2016a), and especially with an increase in the admixture of mineral matter (SiO2) and presence of detrital components (i.e. K, Al, Na, Ti, and Mg). This can be explained as the result of higher river activity and a periodical connection of the River Grabia to the Pawłowa palaeo-oxbow but also of denudation processes in the catchment. The distinct decrease in sediment-associated taxa starting from the end of the Greenlandian was then a response to the weaker fluvial activity of the River Grabia and the transformation of the oxbow-lake to mire.
Amongst the macrophyte/sediment-associated taxa revealing clear patterns, Alona affinis has highly adaptive traits, including the ability to live in lakes of various depths and trophic state and feed on detritus under extreme conditions (Flössner, 2000). Coronatella rectangula also has high ecological plasticity (Flössner, 2000) and is an important indicator of increased in-lake productivity and warm waters in the Pawłowa palaeolake, where its high relative abundance in the Younger Dryas–Greenlandian transition can be linked to higher trophic state and a gradual increase in temperature. The following sharp decrease in the relative abundance of this species is correlated with the presence of Bryales communities but could also be attributed to low water pH and the transformation of the Pawłowa lake into a semi-terrestrial habitat. Finally, it is also relevant that in zones Cl2a and Cl2b fish remains were collected from the core sediment. Fish generally affect zooplankton communities, as size-selective predation on larger Cladocera could increase the relative abundance of some other planktonic species (Davidson et al., 2007; Feniova et al., 2015).
Autecology and diversity as determinants of fossil records
Chironomidae and Cladocera have different life cycles, dispersion mechanisms and resistance to environmental factors such as temperature and hydrology. These autoecological factors were the main drivers of their different responses to the global changes that took place at Pawłowa during the Late Glacial and Holocene. The global factor driving the Grabia valley ecosystems in the long timescale was climate change, which was mainly related to temperature fluctuations and duration of ice cover. However, both Chironomidae and Cladocera also responded to local-scale abiotic factors including water trophic status, hydrology, pH and paludification, and to biotic factors related to aquatic vegetation and fish predation.
Temperature fluctuations were the main driver of freshwater biocenoses during the Late Glacial (Nevalainen et al., 2012; Luoto et al., 2019) and the onset of the Holocene (Kołaczek et al., 2018b). In temperate zones, Chironomidae are regarded as being related directly to air temperature, mostly because of their terrestrial adult stage. In this respect, adult emergence in univoltine species has been found to correlate to higher (summer taxa) or lower (species that emerge only in early spring) day temperatures (Płóciennik et al., 2018). Summer day length influences the rate of Chironomidae larval development, thereby affecting relative abundance and number of generations per year. Chironomidae are fast reproducers and their voltinism may change annually (Brooks, 2006; Armitage et al., 1995). Also, the Chironomidae fauna of glacial lakes in cold climate regions appears to be more sensitive to local continentalism, being composed of profundal, oligotrophic and cold-stenothermic taxa; whereas, floodplain assemblages of the boreal zone are closely related to July air temperatures and reveal stronger habitat influence (Nazarova et al., 2017). Cladocera, albeit generally more sensitive to changes in water trophic status, are also influenced by temperature (Lotter et al., 1997; Korhola, 1999). In this regard, parthenogenetic reproduction in Cladocera is a response to seasonal weather changes, with a quick switch to sexual reproduction and production of resting eggs if temperature conditions become unfavourable (Brendock & De Meester, 2003). As the Pawłowa oxbow was a shallow water body during the Late Glacial, summer temperatures should have acted as main driver in the assemblage composition and relative abundance of both Chironomidae and Cladocera. However, reconstructions of July air temperatures from Pawłowa show different results, with Chironomidae associated to substantially higher (≈ 2−3°C) temperatures compared to Cladocera (Pawłowski et al., 2016a; Płóciennik et al., unpublished data). Also, climatic reconstructions from Świerczyna (see above) have pointed to the importance of assemblage diversity for reliable palaeolimnological reconstructions (Pawłowski et al., 2015b), which however are often confounded by the presence of Corynocera ambigua – a common dominant species in Late Glacial lake sequences of Europe (Brodersen & Lindegaard, 1999).
Another crucial climatic aspect of Pawłowa lake during the Late Glacial–Holocene transition, and one that has been rarely accounted for, is the duration of ice cover, which can affect the phenology of both Chironomidae and Cladocera. Chironomidae seem to be more dependent on mean July temperatures than on level of continentalism, although ice cover influences the survival of their wintering larvae (Self et al., 2011). During winter, these remain in diapause and may suffer from factors indirectly related to low air temperatures and insolation level, including limited availability of food resources and duration of the ice-free period (Nazarova et al., 2017). On the other hand, Cladocera produce ephippia, which are highly resistant to freezing and desiccation (Fryer, 1996) and in cold climates their larger numbers (cf. chydorids) are generally found in lake sediments as the relative duration of the asexual reproduction period is limited in the presence of an open-water shorter season (Sarmaja-Korjonen & Seppä, 2007; Frisch et al., 2012; Pawłowski, 2017).
Although climate is a leading factor at northern latitudes, temperature combines with water trophic status and hydrology in affecting both Chironomidae and Cladocera assemblages (Brown, 2007; Schöll & Kiss, 2008; Kotrys et al., 2020). Previous research in the Grabia valley indicates that the valley’s peripheral stagnant water bodies (i.e. oxbow lakes) had a much more complex ecological profile compared to post-glacial lakes. Chironomidae and Cladocera are reliable indicators of episodic hydrological fluctuations (Pawłowski et al., 2015a, 2016b), and temporary connections of the floodplain water bodies (such as Pawłowa) with the main channel of the River Grabia had a positive influence on the life condition of these aquatic invertebrates. This is because connectivity with the river channel would favour water exchange in the oxbow, thereby increasing the diversity of plant-associated invertebrate communities. Collector-filtrators (hence, including Cladocera) are known to be dominant during stagnant phases, whereas in presence of water movement collector-gatherers (mostly, Chironomidae) play an important role. Limited water exchange between an oxbow and the main river channel would lead to unfavourable conditions for most macrobenthos and meiofauna species due to a decrease in water aeration and light penetration occurring at the onset of paludification processes (Obolewski, 2011). In this regard, aquatic insect subfossil assemblages (including mainly Chironomidae) have provided useful information regarding habitat characteristics and flow activity in palaeochannels, even in a semi-quantitative way (Gandouin et al., 2006; Howard et al., 2010). For Cladocera, the stability of Pawłowa lake was crucial for dominant macrophyte/sediment-associated taxa. In general, microcrustaceans are more exposed to the effects of floods than benthic macrofauna, with Chironomidae larvae able to migrate into the hyporheic zone. Flooding can also be critical for populations located in oxbows adjacent to the river channel or during exceptional overbank flows (Kołaczek et al., 2018a), and that is the reason why Chironomidae provided more fragmentary information about fluvial activity in the River Grabia. Well-developed microcrustacean potamoplankton is an accidental phenomenon (Illyová et al., 2008; Czerniawski & Domagała, 2012), and overbank flood episodes in Pawłowa lake have been also indirectly supported in the present study by geochemical profiles and sedimentology. During floods, there were episodic increases in sediment associated Cladocera and in fully planktonic species, and this pattern in assemblage composition occurred especially during cold climate events. Additionally, these changes were displayed by the higher rarefaction observed in the present study (Fig. 4). It is therefore argued that the increase in Cladocera species richness and relative abundance can be related to enhanced eutrophication during floods, which are considered one of the most important factors contributing to the development of aquatic biota.
Paludification, which causes pH decrease and terrestrialisation during dry phases, is also known to strongly affect macroinvertebrate communities (Verdonschot, 1992). During the Late Glacial, in Pawłowa lake there were slightly alkaline water conditions (Pawłowski et al., 2016a), similar to several shallow water bodies and mires across Poland, although at the end of the Younger Dryas and onset of the Holocene pH generally decreased (i.e. Kołaczek et al., 2015; Forysiak, 2018). Water pH below 6.0 is known to eliminate many Chironomidae taxa, with non-biting midges replaced by biting ones (i.e. Ceratopogonidae) (Henrikson et al., 1982; Walker et al., 1985; Brooks et al., 2007). Generally, paludification was one of the dominant factors in stagnant waters of mid-European lakes since the Northgrippian, and Chironomidae are regarded as a group of desiccation-resistant organisms that spend dry periods in the soil layer (Batzer & Boix, 2016). Current ecosystem studies indicate that wetland and terrestrial Chironomidae communities may be diverse and abundant (Rosenberg et al., 1988; Frouz, 1999), although species of common terrestrial genera (e.g. Limnophyes spp. or Pseudorthocladius spp. Goetghebuer, 1943) can be usually distinguished only at their adult stage. Cladocera remain far more abundant, although in the present study they were represented by few taxa of which Chydorus sphaericus is the most resistant – a species that appears in abundant populations even in ephemeral pools (Płóciennik et al., 2015a).
A biotic factor that influenced invertebrate communities in the Pawłowa palaeo-oxbow is aquatic vegetation. In the Greenlandian, phytophile Chironomidae taxa may have been predominant (Kołaczek et al., 2018a), but during the Late Glacial this role was overtaken by sediment-associated guilds (Płóciennik et al., 2011), and this was also observed in Pawłowa. Whereas, macrophyte- and macrophyte/sediment-associated Cladocera were a dominant component in Late Weichselian assemblages. As Pawłowa was a relatively shallow water body, plants including Characeae and Potamogeton spp. were abundant along the palaeochannel bank zone (Pawłowski et al., 2016a), and abundant vegetation is known to limit resources (e.g. light, nutrients) for phytoplankton and planktonic crustaceans (Kołaczek et al., 2015). A high rate of Ca:Mg has also been observed at Pawłowa (Pawłowski et al., 2016a), causing precipitation of carbonates on plant leaves thereby increasing surface roughness and strongly enhancing periphyton production (Obolewski, 2011). Chironomidae can also benefit from sediment re-suspension overload as a result of denudation processes, as observed during the Bøling-Allerød Interstadial and Younger Dryas (Pawłowski et al., 2016b) - even though Chironomidae have been shown to prefer aquatic vegetation such as Fontinalis spp. Hedwig, 1801 moss patches over inorganic sediment microhabitats (Brusven et al., 1990). In lakes with rich vegetation abundance and diversity, biotic gradients including bentho-pelagic production, fish predation, plant substratum, food availability and competition override the indirect mechanisms associated with nutrient availability (Langdon et al., 2010; Sayer et al., 2010). The quantitative and qualitative structure of macrophyte-associated invertebrate communities is driven, to a great extent, by the hydrological connectivity between an oxbow lake and the parent river channel (Obolewski, 2011), and this is why phytophile and rheophile Chironomidae may appear at the same time during the Younger Dryas–Holocene transition.
Fish predation, a second biotic factor, is considered as equally strong factor as hydrology in shaping Cladocera communities (Pawłowski et al., 2016a). A slight increase in zooplanktivorous fish density in Pawłowa lake during the late Allerød and Younger Dryas resulted in the absence of the larger taxa, especially from the Daphniidae family, paralleled by a relative increase in the relative abundance of species from the Bosminidae family (Figs. 2 and 4). Experimental studies have shown that increasing density and dominance of smaller-sized fish due to water warming conditions would augment predation pressure on large zooplankton species, favouring the development of small-sized ones (Meerhoff et al., 2007). Similarly, a shorter ice-cover period caused by increasing warming conditions in European lakes might be correlated with increasing survival of young fish during winter.
References
Brodgar v. 2.5.7: http://www.brodgar.com, Accessed 09 May 2020.
Amoros, C. & C. Jacquet, 1987. The dead-arm evolution of river systems: a comparison between the information provided by living Copepoda and Cladocera populations and by Bosminidae and Chydoridae remains. Hydrobiologia 145: 333–341.
Armitage, P. D., P. S. Cranston & L. C. V. Pinder, 1995. The Chironomidae. Biology and Ecology of Non-biting Midges. Chapman & Hall, London.
Bass, J., 1998. Last-Instar Larvae and Pupae of the Simuliidae of Britain and Ireland. A Key with Brief Ecological Notes. Freshwater Biological Association, The Ferry House, UK.
Batzer, D. & D. Boix, 2016. Invertebrates in Freshwater Wetlands. An International Perspective on their Ecology. Springer, New York City.
Bennett, K. D., 1996. Determination of the number of zones in a biostratigraphical sequence. New Phytologist 132: 155–170.
Birks, H. J. B., 1986. Numerical zonation, comparison and correlation of Quaternary pollen stratigraphical data. In Berglund, B. (ed.), Handbook of Holocene Palaeoecology and Palaeohydrology. Wiley-Interscience, Chichester: 743–774.
Birks, H. J. B., 1995. Quantitative palaeoenvironmental reconstructions. In Maddy, D. & J. S. Brew (eds), Statistical Modelling of Quaternary Science Data Technical Guide 5. Quaternary Research Association, Cambridge: 161–254.
Birks, H. J. B. & H. H. Birks, 2004. Quaternary Palaeoecology. The Blackburn Press, New Jersey, USA.
Birks, H. J. B. & A. D. Gordon, 1985. Numerical methods in Quaternary pollen analysis. Academic Press, London.
Birks, H. J. B., A. F. Lotter, S. Juggins & J. P. Smol, 2012. Tracking Environmental Change Using Lake Sediments. Data Handling and Numerical Techniques, Vol. 5. Springer, Dordrecht.
Bjerring, R., E. Becares, S. Declerck, E. M. Gross, L.-A. Hansson, T. Kairesalo, M. Nykanen, A. Halkiewicz, R. Kornijów, J. M. Conde-Porcuna, M. Seferlis, T. Noges, B. Moss, S. L. Amsinck, B. Vad Odgaard & E. Jeppesen, 2009. Subfossil Cladocera in relation to contemporary environmental variables in 54 Pan-European lakes. Freshwater Biology 54: 2401–2417.
Błędzki, L. A. & J. I. Rybak, 2016. Freshwater Crustacean Zooplankton of Europe. Cladocera & Copepoda (Calanoida, Cyclopoida). Key to Species Identification, with Notes on Ecology, Distribution, Methods and Introduction to Data Analysis. Springer, Basel.
Brendock, L. & L. De Meester, 2003. Egg banks in freshwater zooplankton: evolutionary and ecological archives in the sediment. Hydrobiologia 491: 65–84.
Brodersen, K. P. & C. Lindegaard, 1999. Mass occurrence and sporadic distribution of Corynocera ambigua Zetterstedt (Diptera, Chironomidae) in Danish lakes. Neo- and palaeolimnological records. Journal of Paleolimnology 22: 41–52.
Brodersen, K. P. & R. Quinlan, 2006. Midges as palaeoindicators of lake productivity, eutrophication and hypolimnetic oxygen. Quaternary Science Reviews 25: 1995–2012.
Brooks, S. J., 2006. Fossil midges (Diptera: Chironomidae) as palaeoclimatic indicators for the Eurasian region. Quaternary Science Reviews 25: 1894–1910.
Brooks, S. J., P. G. Langdon & O. Heiri, 2007. The Identification and Use of Palaearctic Chironomidae Larvae in Palaeoecology. QRA Technical Guide No. 10. Quaternary Research Association, London.
Brown, T. G., 2007. The value of long-term (palaeo) records in hydroecology and ecohydrology. In Wood, J. P., D. M. Hannah & J. P. Sadler (eds), Hydroecology and Ecohydrology: Past, Present and Future. Wiley, New York: 129–142.
Brusven, M. A., W. R. Meehan & R. C. Biggam, 1990. The role of aquatic moss on community composition and drift of fish-food organisms. Hydrobiologia 196: 39–50.
Clarke, K. R. & R. N. Gorley, 2006. PRIMER v6: User Manual/Tutorial. PRIMER-E, Plymouth.
Czerniawski, R. & J. Domagała, 2012. Potamozooplankton communities in three different outlets from mesotrophic lakes located in lake-river system. Oceanological and Hydrobiological Studies 41: 46–56.
Davidson, T. A., C. D. Sayer, M. R. Perrow, M. Bramm & E. Jeppesen, 2007. Are controls on species composition similar for contemporary and sub-fossil cladoceran assemblages? A study of 39 shallow lakes of contrasting trophic status. Journal of Paleolimnology 38: 117–134.
Davidson, T. A., C. D. Sayer, P. G. Langdon, A. Burgess & M. Jackson, 2010. Inferring past zooplanktivorous fish and macrophyte density in a shallow lake: application of a new regression tree model. Freshwater Biology 55: 584–599.
Doray, M., P. Petitgas, M. Huret, E. Duhamel, J.-B. Romagnan, M. Authier, C. Dupuy & J. Spitz, 2018. Monitoring small pelagic fish in the Bay of Biscay ecosystem, using indicators from an integrated survey. Progress in Oceanography 166: 168–188.
Duigan, C. A., 1992. The ecology and distribution of the littoral freshwater Chydoridae (Branchiopoda, Anomopoda) of Ireland with taxonomic comments on some species. Hydrobiologia 241: 1–70.
Faustova, M., V. Sacherová, J.-E. Svensson & D. J. Taylor, 2011. Radiation of European Eubosmina (Cladocera) from Bosmina (E.) longispina – concordance of multipopulation molecular data with paleolimnology. Limnology and Oceanography 56(2): 440–450.
Feniova, I., P. Dawidowicz, M. Gladyshev, I. Kostrzewska-Szlakowska, M. Rzepecki, V. Razlutskij, N. Sushchik, N. Majsak & A. Dzialowski, 2015. Experimental effects of large-bodied Daphnia, fish and zebra mussels on cladoceran community and size structure. Journal of Plankton Research 37: 611–625.
Flössner, D., 1972. Krebstiere, Crustacea (Kiemen- und Blattfüßer, Branchiopoda, Fischläuse, Branchiura). Die Tierwelt Deutschlands 60. Veb Gustav Fischer Verlag, Jena: 499.
Flössner, D., 2000. Die Haplopoda und Cladocera (ohne Bosminidae) Mitteleuropas. Backhuys Publishers, Leiden: 428.
Forysiak, J., 2018. The diversity of geomorphological features of peatland basins in Central Poland and its paleoecological significance – a review. Bulletin of the Geological Society of Finland 90: 175–184.
Frey, D. G., 1986. Cladocera analysis. In Berglund, B. E. (ed.), Handbook of Holocene Paleoecology and Paleohydrology. Wiley, Chichester: 667–692.
Frisch, D., K. Cottenie, A. Badosa & A. J. Green, 2012. Strong spatial influence on colonization rates in a pioneer zooplankton metacommunity. PLoS ONE 7: e40205.
Frouz, J., 1999. Use of soil dwelling Diptera (Insecta, Diptera) as bioindicators: a review of ecological requirements and response to disturbance. Agriculture, Ecosystems & Environment 74: 167–186.
Fryer, G., 1996. Diapause, a potent force in the evolution of freshwater crustaceans. Hydrobiologia 320: 1–14.
Gandouin, E., A. Maasri, B. Van Vliet-Lanoё & E. Franquet, 2006. Chironomid (Insecta: Diptera) assemblages from a gradient of lotic and lentic waterbodies in river floodplains of France: a methodological tool for paleoecological applications. Journal of Paleolimnology 35: 149–166.
Gąsiorowski, M. & M. Kupryjanowicz, 2009. Lake–peat bog transformation recorded in the sediments of the Stare Biele mire (Northeastern Poland). Hydrobiologia 631: 143–154.
Henrikson, L., J. B. Olafsson & H. G. Oscarson, 1982. The impact of acidification on Chironomidae (Diptera) as indicated by subfossil stratification. Hydrobiologia 86: 223–229.
Hofmann, W., 2000. Response of the chydorid faunas to rapid climatic changes in four alpine lakes at different altitudes. Palaeogeography, Palaeoclimatology, Palaeoecology 159: 281–292.
Howard, L. C., P. J. Wood, M. T. Greenwood, H. M. Rendell, S. J. Brooks, P. D. Armitage & C. A. Extence, 2010. Sub-fossil Chironomidae as indicators of palaeoflow regimes: integration into the PalaeoLIFE flow index. Journal of Quaternary Science 25: 1270–1283.
Illyová, M., K. Bukvayová & D. Némethová, 2008. Zooplankton in a Danube River Arm near Rusovce (Slovakia). Biologia 63: 566–573.
Juggins, S., 2007. C2 version 15 user guide Software for Ecological and Palaeoecological Data Analysis and Visualisation. Newcastle University, Newcastle upon Tyne.
Kittel, P., M. Płóciennik, R. K. Borówka, D. Okupny, D. Pawłowski, O. Peyron, R. Stachowicz-Rybka, M. Obremska & K. Cywa, 2016. Early Holocene hydrology and environments of the Ner River (Poland). Quaternary Research 85: 187–203.
Klatkowa, H., 1984. Detailed Geological Map of Poland, 1:50,000, Pabianice Sheet. Geological Institute, Warsaw.
Klink, A. G. & H. K. M. Moller Pillot, 2003. Chironomidae Larvae. Key to Higher Taxa and Species of the Lowlands of Northwestern Europe. ETI, Amsterdam, CD-ROM.
Klukowska, M., M. Płóciennik & J. Żbikowski, 2011. Lipiniella moderata (Diptera, Chironomidae) – new species for the Polish fauna. Lauterbornia 72: 1–6.
Kłysik, K., 2001. Warunki klimatyczne (Climatic conditions). In Liszewski, S. (ed.), Zarys monografii województwa łódzkiego (Monograph of the Łódź Voivodeship). Łódzkie Towarzystwo Naukowe, Lodz: 68–81. (in Polish).
Kołaczek, P., M. Gałka, M. Karpińska-Kołaczek & M. Lutyńska, 2015. Late Pleniglacial and Late Glacial lake-mire transformations in south-eastern Poland reflected in aquatic and wetland vegetation changes. Quaternary International 388: 39–50.
Kołaczek, P., M. Gałka, K. Apolinarska, M. Płóciennik, M. Gąsiorowski, S. J. Brooks, S. M. Hutchinson & M. Karpińska-Kołaczek, 2018a. A multi-proxy view of exceptionally early postglacial development of riparian woodlands with Ulmus in the Dniester River valley, western Ukraine. Review of Palaeobotany and Palynology 250: 27–43.
Kołaczek, P., M. Płóciennik, M. Gałka, K. Apolinarska, K. Tosik, M. Gąsiorowski, S. J. Brooks & M. Karpińska-Kołaczek, 2018b. Persist or take advantage of global warming: a development of Early Holocene riparian forest and oxbow lake ecosystems in Central Europe. Quaternary Science Reviews 200: 191–211.
Korhola, A., 1999. Distribution patterns of Cladocera in subarctic Fennoscandian lakes and their potential in environmental reconstruction. Ecography 22: 357–373.
Korhola, A. & M. Rautio, 2001. Cladocera and other small branchiopods. In Smol, J. P., H. J. B. Birks & W. M. Last (eds), Tracking Environmental Change Using Lake Sediments, Vol. 4., Zoological Indicators Kluwer Academic Press, Dordrecht: 5–41.
Korhola, A., H. Olander & T. Blom, 2000. Cladoceran and chironomid assemblages as qualitative indicators of water depth in subarctic Fennoscandian lakes. Journal of Paleolimnology 24: 43–54.
Kotrys, B., M. Płóciennik, P. Sydor & S. J. Brooks, 2020. Expanding the Swiss-Norwegian chironomid training set by incorporating Polish data and assessing its transfer-function capability using temperature reconstructions from Lake Żabieniec. Boreas 49: 89–107.
Langdon, P. G., Z. Ruiz, S. Wynne, C. D. Sayer & T. A. Davidson, 2010. Ecological influences on larval chironomid communities in shallow lakes: implications for palaeolimnological interpretations. Freshwater Biology 55: 531–545.
Lotter, A. F. & S. Juggins, 1991. POLPROF, TRAN and ZONE. Programmes for plotting, editing and zoning of pollen and diatom data. INQUA Commission for the study of the Holocene. Working Group on Data Handing Methods. Newsletter 6: 4–6.
Lotter, A. F., H. J. B. Birks, W. Hofmann & A. Marchetto, 1997. Modern diatom, cladocera, chironomid, and chrysophyte cyst assemblages as quantitative indicators for the reconstruction of past environmental conditions in the Alps. I. Climate. Journal of Paleolimnology 18: 395–420.
Luoto, T. P., L. Nevalainen, S. Kultti & K. Sarmaja-Korjonen, 2011. An evaluation of the influence of water depth and river inflow on quantitative Cladocera-based temperature and lake level inferences in a shallow boreal lake. Hydrobiologia 676: 143–154.
Luoto, T. P., B. Kotrys & M. Płóciennik, 2019. East European chironomid-based calibration model for past summer temperature reconstructions. Climate Research 77: 63–76.
MacArthur, R. H., 1957. On the relative abundance of bird species. The Proceedings of the National Academy of Sciences USA 43: 293–295.
Medley, K. A. & J. E. Havel, 2007. Hydrology and local environmental factors influencing zooplankton communities in floodplain ponds. Wetlands 27: 864–872.
Meerhoff, M., J. M. Clemente, F. Teixeira-de Mello, C. Iglesias, C. R. Pedersen & E. Jeppesen, 2007. Can warm climate-related structure of littoral predator assemblies weaken clear water state in shallow lakes? Global Change Biology 13: 1888–1897.
Moller Pillot, H. K. M., 2009. Chironomidae Larvae of the Netherlands and Adjacent Lowlands. Biology and Ecology of the Chironomini. KNNV Publishing, Zeist.
Moller Pillot, H. K. M., 2013. Chironomidae Larvae of the Netherlands and Adjacent Lowlands. Biology and Ecology of the Aquatic Orthocladiinae. KNNV Publishing, Zeist.
Nazarova, L. B., A. E. Self, S. J. Brooks, N. Solovieva, L. S. Syrykh & V. A. Dauvalter, 2017. Chironomid fauna of the lakes from the Pechora River Basin (East of European part of Russian Arctic): ecology and reconstruction of recent ecological changes in the region. Contemporary Problems of Ecology 10: 350–362.
Nevalainen, L., 2011. Intra-lake heterogeneity of sedimentary cladoceran (Crustacea) assemblages forced by local hydrology. Hydrobiologia 676: 9–22.
Nevalainen, L. & T. P. Luoto, 2017. Relationship between cladoceran (Crustacea) functional diversity and lake trophic gradients. Functional Ecology 31: 488–498.
Nevalainen, L., T. P. Luoto, S. Kultti & K. Sarmaja-Korjonen, 2012. Do subfossil Cladocera and chydorid ephippia disentangle Holocene climate trends? Holocene 22: 291–299.
Obolewski, K., 2011. Composition and density of plant-associated invertebrates in relation to environmental gradients and hydrological connectivity of wetlands. Oceanological and Hydrobiological Studies 40: 52–63.
Obolewski, K. & K. Glińska-Lewczuk, 2011. Effects of oxbow reconnection based on the distribution and structure of benthic macroinvertebrates. Clean – Soil. Air, Water 39: 853–862.
Palmer, S., I. Walker, M. Heinrichs, R. Hebda & G. Scudder, 2002. Postglacial midge community change and Holocene palaeotemperature reconstructions near treeline, southern British Columbia (Canada). Journal of Paleolimnology 28: 469–490.
Pan, B.-Z., Z.-Y. Wang & M.-Z. Xu, 2012. Macroinvertebrates in abandoned channels: assemblage characteristics and their indications for channel management. River Research and Applications 28: 1149–1160.
Pawłowski, D., 2011. Evolution of an Eemian lake based on Cladocera analysis (Konin area, Central Poland). Acta Geologica Polonica 61: 441–450.
Pawłowski, D., 2017. The usefulness of subfossil Cladocera remains in Younger Dryas climatic reconstructions in central Poland. Acta Geologica Polonica 67: 567–584.
Pawłowski, D., G. Kowalewski, K. Milecka, M. Płóciennik, M. Woszczyk, T. Zieliński, D. Okupny, W. Włodarski & J. Forysiak, 2015a. A reconstruction of the palaeohydrological conditions of a flood-plain: a multi-proxy study from the Grabia River valley mire, central Poland. Boreas 44: 543–562.
Pawłowski, D., M. Płóciennik, S. J. Brooks, T. P. Luoto, K. Milecka, L. Nevalainen, O. Peyron, A. Self & T. Zieliński, 2015b. A multiproxy study of Younger Dryas and Early Holocene climatic conditions from the Grabia River palaeo-oxbow lake (central Poland). Palaeogeography, Palaeoclimatology, Palaeoecology 438: 34–50.
Pawłowski, D., R. K. Borówka, G. Kowalewski, T. P. Luoto, K. Milecka, L. Nevalainen, D. Okupny, T. Zieliński & J. Tomkowiak, 2016a. Late Weichselian and Holocene record of the paleoenvironmental changes in a small river valley in Central Poland. Quaternary Science Reviews 135: 24–40.
Pawłowski, D., R. K. Borówka, G. Kowalewski, T. P. Luoto, K. Milecka, L. Nevalainen, D. Okupny, M. Płóciennik, M. Woszczyk, J. Tomkowiak & T. Zieliński, 2016b. The response of flood-plain ecosystems to the Late Glacial and Early Holocene hydrological changes: a case study from a small Central European river valley. Catena 147: 411–428.
Płóciennik, M., A. Self, H. J. B. Birks & S. J. Brooks, 2011. Chironomidae (Insecta: Diptera) succession in Żabieniec bog and its palaeo-lake (central Poland) through the Late Weichselian and Holocene. Palaeogeography, Palaeoclimatology, Palaeoecology 307: 150–167.
Płóciennik, M., A. Kruk, J. Forysiak, D. Pawłowski, K. Mianowicz, S. Elias, R. K. Borówka, M. Kloss, M. Obremska, R. Coope, M. Krąpiec, P. Kittel & S. Żurek, 2015. Fen ecosystem responses to water-level fluctuations during the early and middle Holocene in central Europe: a case study from Wilczków, Poland. Boreas 44: 721–740.
Płóciennik, M., M. Skonieczka, O. Antczak & J. Siciński, 2018. Phenology of non-biting midges (Diptera Chironomidae) in peatland ponds, Central Poland. Entomologica Fennica 29: 61–74.
Porinchu, D. F. & G. M. MacDonald, 2003. The use and application of freshwater midges (Chironomidae: Insecta: Diptera) in geographical research. Progress in Geography 27: 378–422.
Robertson, A. L., 1990. The population dynamics of Chydoridae and Macrothricidae (Cladocera:Crustacea) from the River Thames, U.K. Freshwater Biology 24: 375–389.
Rolland, N. & I. Larocque, 2007. The efficiency of kerosene flotation for extraction of chironomid head capsules from lake sediments samples. Journal of Paleolimnology 37: 565–572.
Rosenberg, M. D., A. P. Wiens & B. Bilyj, 1988. Chironomidae (Diptera) of peatlands in northwestern Ontario, Canada. Ecogeography 11: 19–31.
Sarmaja-Korjonen, K. & H. Seppä, 2007. Abrupt and consistent responses of aquatic and terrestrial ecosystems to the 8200 cal. yr cold event: a lacustrine record from Lake Arapisto. Finland. The Holocene 17: 457–467.
Sayer, C. D., T. A. Davidson, J. I. Jones & P. G. Langdon, 2010. Combining contemporary ecology and palaeolimnology to understand shallow lake ecosystem change. Freshwater Biology 55: 487–499.
Schöll, K. & A. Kiss, 2008. Spatial and temporal distribution patterns of zooplankton assemblages (Rotifera, Cladocera, Copepoda) in the water bodies of the Gemenc Floodplain (Duna-Dráva National Park, Hungary). Opuscula Zoologica Budapest 39: 65–76.
Self, A., S. J. Brooks, H. J. B. Birks, L. B. Nazarova, D. Porinchu, A. Odland, H. Yang & V. J. Jones, 2011. The influence of temperature and continentality on modern chironomid assemblages in high-latitude Eurasian lakes: development and application of new chironomid-based climate-inference models in northern Russia. Quaternary Science Reviews 30: 1122–1141.
Shojaei, M. G., L. Gutov, J. Dannheim, E. Rachor, A. Schröder & T. Brey, 2016. Common trends in German Bight benthic macrofaunal communities: assessing temporal variability and the relative importance of environmental variables. Journal of Sea Research 107: 25–33.
Słowiński, M., K. Marcisz, M. Płóciennik, M. Obremska, D. Pawłowski, D. Okupny, S. Słowińska, R. K. Borówka, P. Kittel, J. Forysiak, D. J. Michczyńska & M. Lamentowicz, 2016. Drought as a stress driver of ecological changes in peatland – A palaeoecological study of peatland development between 3500 BCE and 200 BCE in central Poland. Palaeogeography Palaeoclimatology Palaeoecology 461: 272–291.
Starkel, L., J. D. Michczyńska, M. Krąpiec, W. Margielewski, D. Nalepka & A. Pazdur, 2013. Progress in the Holocene chrono-climatostratigraphy of Polish territory. Geochronometria 40: 1–21.
Szeroczyńska, K. & K. Sarmaja-Korjonen, 2007. Atlas of subfossil Cladocera from central and northern Europe. Friends of Lower Vistula Society, Świecie, Poland.
ter Braak, C. J. F. & S. Juggins, 1993. Weighted averaging partial least squares regression (WA-PLS): an improved method for reconstructing environmental variables from species assemblages. Hydrobiologia 269: 485–502.
ter Braak, C. J. F. & P. Šmilauer, 2002. CANOCO Reference Manual and CanoDraw for Windows User’s Guide: Software for Canonical Community Ordination (Version 4.5), Microcomputer Power, Ithaca, NY, USA.
Vadadi-Fülöp, C., 2013. Microcrustacean assemblages in a large river: on the importance of the flow regime. Hydrobiologia 702: 129–140.
Vallenduuk, H. J. & H. K. M. Moller Pillot, 2007. Chironomidae Larvae of the Netherlands and Adjacent Lowlands. General Ecology and Tanypodinae. KNNV Publishing, Zeist.
Van Damme, K. & H. J. Dumont, 2008. Further division of Alona Baird, 1843: separation and position of Coronatella Dybowski & Grochowski and Ovalona gen. n. (Crustacea: Cladocera). Zootaxa 1960: 1–44.
Van Damme, K., A. A. Kotov & H. J. Dumont, 2010. A checklist of names in Alona Baird 1843 (Crustacea: Cladocera: Chydoridae) and their current status: an analysis of the taxonomy of a lump genus. Zootaxa 2330: 1–63.
Vandenberghe, J., 2003. Climate forcing of fluvial system development: an evolution of ideas. Quaternary Science Reviews 22: 2053–2060.
Verdonschot, P. F. M., 1992. Macrofaunal community types in ponds and small lakes (Overijssel, The Netherlands). Hydrobiologia 232: 111–132.
Vilizzi, L., 2012. Abundance trends in floodplain fish larvae: the role of annual flow characteristics in the absence of overbank flooding. Fundamental and Applied Limnology 181: 215–227.
Walker, I. R., C. H. Fernando & C. G. Paterson, 1985. Association of Chironomidae (Diptera) of shallow, acid, humic lakes and bog pools in Atlantic Canada, and a comparison with an earlier paleoecological investigation. Hydrobiologia 120: 11–22.
Whitehouse, N. E., P. G. Langdon, R. Bustin & S. Galsworthy, 2008. Fossil insects and ecosystem dynamics in wetlands: implications for biodiversity and conservation. Biodiversity and Conservation 17: 2055–2078.
Whiterod, N. S., M. P. Hammer & L. Vilizzi, 2015. Spatial and temporal variability in fish community structure in Mediterranean climate temporary streams. Fundamental and Applied Limnology 187: 135–150.
Woś, A., 1999. Klimat Polski (Climate of Poland). PWN, Warsaw. (in Polish).
Zawiska, I., M. Słowiński, A. Correa-Metrio, M. Obremska, T. Luoto, L. Nevalainen, M. Woszczyk & K. Milecka, 2014. The response of a shallow lake and its catchment to Late Glacial climate changes – A case study from eastern Poland. Catena 126: 1–10.
Zuur, A. F., E. N. Ieno & G. M. Smith, 2007. Analysing Ecological Data. Springer, The Netherlands.
Acknowledgements
We thank MSc Ewa Janowska from the University of Lodz for support Chironomidae laboratory work. The current research project was financed through a grant from the National Science Centre (No. 2011/01/B/ST10/04905: ‘The role of valley mires in reconstructing palaeohydrological events in the light of selected palaeoecological studies’).
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: Jasmine Saros
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Płóciennik, M., Pawłowski, D., Vilizzi, L. et al. From oxbow to mire: Chironomidae and Cladocera as habitat palaeoindicators. Hydrobiologia 847, 3257–3275 (2020). https://doi.org/10.1007/s10750-020-04327-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-020-04327-6