Abstract
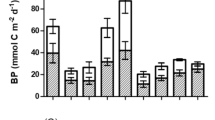
The intensity of respiratory carbon loss through respiration, assessed by respiratory electron transport system (ETS) activity, was studied in net zooplankton, microplankton, zoobenthos, macrophytes and sediment in the littoral and pelagial including profundal zone of Lake Bohinj over a complete year. The contributions of these components to the total metabolic activity in a particular zone of the lake were compared and the relationship between the contributions of littoral and pelagial including profundal respiratory carbon loss and the estimated assimilated carbon in phytoplankton and macrophytes was determined. The greatest areal (m−2) carbon loss was observed through sediments in the littoral zone which, on average, contributed 97.6% to total carbon loss in the littoral compartment of the lake. Significantly lower values were observed in this zone for zoobenthos (1.2%), microplankton (0.9%), macrophytes (0.2%) and zooplankton (0.1%). In the combined pelagial and profundal zone, sediments contributed 78.8% to the total carbon loss. Lower values were observed in microplankton (13.4%), followed by zooplankton (7.0%) and zoobenthos (0.8%). Total areal respiratory carbon loss in the littoral significantly exceeded that in the pelagial including profundal zone, but the littoral zone and the pelagial including profundal zone made similar contributions of respiratory carbon loss to whole-lake metabolism. Whole-lake respiratory carbon loss through zooplankton, microplankton, zoobenthos, sediments and macrophytes exceeded the estimated carbon productivity of phytoplankton and macrophytes. The latter was greater in littoral than in pelagial including profundal. In littoral, macrophytes contribute significantly to carbon productivity, but less to respiration, while sediment contributed the greatest part of respiration in both littoral and pelagial including profundal.



Similar content being viewed by others
References
APHA, AWWA & WEF, 1998. Standard methods for the examination of water and wastewater 20th ed. In Clesceri, L. S., A. E. Greenberg & A. D. Eaton (eds), Persulfate Method. APHA, AWWA & WEF, Washington.
ARSO, 2007. The report of quality of lakes for the year 2006. Environmental Agency of the Republic of Slovenia (ARSO), Ministry of the Environment and Spatial Planning, Slovenia: 149 (in Slovenian).
Bamstedt, U., 1980. ETS activity as an estimator of respiratory rate of zooplankton populations. The significance of variations in environmental factors. Journal of Experimental Marine Biology and Ecology 42: 267–283.
Carignan, R., D. Planas & C. Vis, 2000. Planktonic production and respiration in oligotrophic Shield lakes. Limnology and Oceanography 45: 189–199.
Cole, J. J., M. L. Pace, S. R. Carpenter & J. F. Kitchell, 2000. Persistence of net heterotrophy in lakes during nutrient addition and food web manipulations. Limnology and Oceanography 45: 1718–1730.
del Giorgio, P. A. & R. H. Peters, 1994. Patterns in planktonic P:R ratios in lakes: influence of lake trophy and dissoved organic carbon. Limnology and Oceanography 39: 772–787.
den Heyer, C. & J. Kalff, 1998. Organic matter mineralization rates in sediments: a within- and among-lake study. Limnology and Ocenaography 43: 695–705.
Devol, A. H., 1979. Zooplankton respiration and its relation to plankton dynamics in two lake of contrasting trophic state. Limnology and Oceanography 24: 893–905.
Devol, A. H. & T. T. Packard, 1978. Seasonal changes in respiratory enzyme activity and productivity in Lake Washington microplankton. Limnology and Oceanography 23: 104–117.
Duarte, C. M. & S. Agustí, 1998. The CO2 balance of unproductive aquatic ecosystems. Science 281: 234–236.
G.-Tóth, L., 1992. Respiratory electron transport system activity (ETS)-activity of the plankton and sediment in Lake Balaton (Hungary). Hydrobiologia 243/244: 157–166.
G.-Tóth, L., 1999. Aktivität des Elektronentransportsystems. In von Tümpling, W. & G. Friedrich (eds), Biologische Gewässeruntersuchung. Methoden der Biologischen Wasseruntersuchung 2. Gustav Fischer Verl, Jena, Stuttgart, Lübeck, Ulm: 465–473.
G.-Tóth, L., Z. Langó, J. Padisák & E. Varga, 1994. Terminal electron transport system (ETS)-activity in the sediment of Lake Balaton, Hungary. Hydrobiologia 281: 129–139.
G.-Tóth, L., P. Carrillo & L. Cruz-Pizarro, 1995. Respiratory electron transport system (ETS)-activity of the plankton and biofilm in the high-mountain lake La Caldera (Sierra Nevada, Spain). Archiv für Hydrobiologie 135: 65–78.
Hannan, H. H. & T. C. Dorris, 1970. Succession of a macrophyte community in a constant temperature river. Limnology and Oceanography 15: 442–453.
James, M. R., 1987. Respiratory rates in cladoceran Ceriodaphnia dubia in lake Rotiongaio, a monomictic lake. Journal of Plankton Research 9: 573–578.
Jeffrey, S. W. & G. F. Humphrey, 1975. New spectrophotometric equations for determining chlorophylls a, b, c 1 and c 2 in higher plants, algae and natural phytoplankton. Biochem. Phyriol. Pflanzen (BPP), Bd. 167: 191–194.
Jónasson, P. M., C. Lindegaard & K. Hamburger, 1990. Energy budget of Lake Esrom. Verhandlungen Internationale Vereinigung für Limnologie 24: 632–640.
Karlsson, J., M. Jansson & A. Jonsson, 2007. Respiration of allochthonous organic carbon in unproductive forest lakes determined by the Keeling plot method. Limnology and Oceanography 52: 603–608.
Kenner, R. A. & S. I. Ahmed, 1975a. Measurements of electron transport activities in marine phytoplankton. Marine Biology 33: 119–127.
Kenner, R. A. & S. I. Ahmed, 1975b. Correlation between oxygen utilization and electron transport activity in marine phytoplankton. Marine Biology 33: 129–133.
Kurashov, E. A., 2002. The role of meiobenthos in lake ecosystems. Aquatic Ecology 36: 447–463.
Lampert, W., 1984. The measurement of respiration. In Downing, J. A. & F. H. Rigler (eds), A Manual on Methods for the Assessment of Secondary Productivity in Fresh Water. IPB Handbook 17, 2nd ed. Blackwell Scientific Publications, Oxford: 413–468.
Lindegaard, C., 1994. The role of zoobenthos in energy flow in two shallow lakes. Hydrobiologia 275(276): 313–322.
López-Archilla, A. I., S. Molla, M. C. Coleto, M. C. Guerrero & C. Montes, 2004. Ecosystem metabolism in a Mediterranean shallow lake (Laguna de Santa Olalla, Doñana National Park, SW Spain). Wetlands 24: 848–858.
Madsen, T. V., B. Olesen & J. Bagger, 2002. Carbon acquisition and carbon dynamics by aquatic isoetids. Aquatic Botany 73: 351–371.
Mazej, Z. & A. Gaberščik, 1999. ETS-activity as a measure of vitality of different macrophyte species. Phyton 39: 181–185.
Meili, M., 1992. Sources, concentrations and characteristics of organic matter in softwater lakes and streams of the Swedish forest region. Hydrobiologia 229: 23–41.
Muri, G. & T. Simčič, 2004. Respiratory activity in sediments in three mountain lakes of the Julian Alps and in subalpine Lake Bled (Slovenia): an effect of altitude and antropic influence. Aquatic Microbial Ecology 34: 291–299.
Owens, T. G. & F. D. King, 1975. The measurement of respiratory electron transport system activity in marine zooplankton. Marine Biology 30: 27–36.
Pace, M. L. & Y. T. Prairie, 2005. Respiration in lake. In Williams, P. J. L. & P. del Giorgio (eds), Respiration in Aquatic Ecosystems. Oxford University Press, Oxford: 103–121.
Packard, T. T., 1971. The measurement of respiratory electron-transport activity in marine phytoplankton. Journal of Marine Research 29: 235–244.
Packard, T. T., 1985. Measurement of electron transport activity of microplankton. In Jannash, H. & P. J. L. Williams (eds), Advances in Aquatic Microbiology, Vol. 3. Academic Press, London: 207–261.
Packard, T. T., A. H. Devol & F. D. King, 1975. The effect of temperature on the respiratory electron transport system in marine plankton. Deep Sea Research 22: 237–249.
Pålsson, C., E. S. Kritzberg, K. Christoffersen & W. Graneli, 2005. Net heterotrophy in Faroe Islands clear-water lakes: causes and consequences for bacterioplankton and phytoplankton. Freshwater Biology 50: 2011–2020.
Peñuelas, J., J. Murillo & J. Azcón-Bieto, 1988. Actual and potential dark respiration rates and different electron transport pathways in freshwater aquatic plants. Aquatic Botany 30: 353–362.
Petersen, J. E. & C.-C. Chen, 1999. A method for measuring depth-integrated community metabolism in experimental planktonic-benthic ecosystems. Hydrobiologia 319: 23–32.
Relexans, J. C., 1996. Measurement of the respiratory electron transport system (ETS) activity in marine sediments: state-of-the-art and interpretation. I. Methodology and review of literature data. Marine Ecology Progress Series 136: 277–287.
Simčič, T., 2005. The role of plankton, zoobenthos, and sediment in organic matter degradation in oligotrophic and eutrophic mountain lakes. Hydrobiologia 532: 69–79.
Simčič, T. & A. Brancelj, 1997. Electron transport system (ETS) activity in five Daphnia species at different temperatures. Hydrobiologia 360: 117–125.
Simčič, T. & A. Brancelj, 2002. Intensity of mineralization processes in mountain lakes in NW Slovenia. Aquatic Ecology 36: 345–354.
Törnblom, E. & K. Pettersson, 1998. Bacterial production and total sediment metabolism in profundal Lake Erken sediments. Archiv für Hydrobiologie Special Issues Advances in Limnology 51: 177–183.
Acknowledgements
We thank Franja Pajk and Tadej Mezek for their help in the field, and Andreja Jerebic for the chemical analysis of water samples. We thank both reviewers for constructive and helpful comments and suggestions. We also wish to thank Roger Pain for English revision of the manuscript. We acknowledge the financial support by the Slovenian Research Agency (Project J-9388).
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: Darren Bade
Rights and permissions
About this article
Cite this article
Simčič, T., Germ, M. Organic matter degradation through respiration in littoral and pelagial including profundal zones of an oligotrophic lake assessed by electron transport system activity. Hydrobiologia 635, 137–146 (2009). https://doi.org/10.1007/s10750-009-9906-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-009-9906-6