Abstract
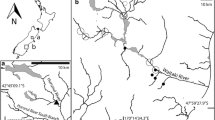
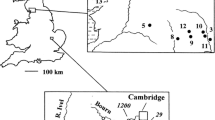
A study was made of the aquatic environment, tissue nutrient composition and surface phosphatase activities of the aquatic moss Warnstorfia fluitans in Brandon Pithouse Stream, a small acidic stream in N-E England. The water, which originates from an underground spring, had been pH 2.6 for at least 30 years, but about 3.9 during the present study. The moss was by far the most abundant phototroph during all this period. Seasonal changes in aqueous nitrogen and phosphorus fractions were measured over a 2-year period near the source. Most of the filtrable N and P were at times organic, but the very high N:P ratio (even if organic N is excluded) suggests that only organic phosphate is likely to be important for the moss. There was a high peak in organic phosphate in late spring in both study years. Surface phosphomonoesterase (PMEase) and phosphodiesterase (PDEase) activities were highly correlated in the field and in axenic culture, though there were some differences in response to environmental factors. Axenic material showed higher PMEase and PDEase activities when grown with organic P than with inorganic P. Although the data suggest that internal P content is an important factor influencing phosphatase activities, PDEase activity was especially marked when the moss was grown with the diester, DNA, as P source, indicating that at least one of its surface phosphatases can also respond directly to the environment.






Similar content being viewed by others
Abbreviations
- bis-pNPP:
-
bis-para-nitrophenyl phosphate
- DMG:
-
3-dimethylglutaric acid
- FON:
-
filtrable organic nitrogen
- FOP:
-
filtrable organic phosphorus
- FRP:
-
filtrable reactive phosphorus
- HEPES:
-
N-(2-hydroxyethyl)-piperazine-N′-2-ethanesulfonic acid
- MU:
-
methylumbelliferone
- MUP:
-
methylumbelliferyl phosphate
- pNP:
-
para-nitrophenol
- pNPP:
-
para-nitrophenyl phosphate
- PMEase:
-
phosphomonoesterase
- PDEase:
-
phosphodiesterase
- Pi:
-
inorganic phosphate-P
- TIN:
-
total inorganic nitrogen
- TN:
-
total nitrogen
- TP:
-
total phosphorus
References
Bates, J. W., 1994. Responses of the mosses Brachythecium rutabulum and Pseudoscleropodium purum to a mineral nutrient pulse. Functional Ecology 8: 686–692.
Bates, J. W., 2000. Mineral nutrition, substratum ecology, and pollution. In Shaw A. J. & B. Goffinet (eds), Bryophyte Biology. Cambridge University Press, UK: 248–311.
Brock, T. D., 1978. The habitats. In Brock T. D. (eds), Thermophilic Microorganisms and Life at High Temperatures. Springer-Verlag, New York, 12–38.
Brown, D. H., 1984. Uptake of mineral elements and their use in pollution monitoring. In Dyer A. F. & J. G. Duckett (eds), The Experiment Biology of Bryophytes. Academic Press, London: 229–255.
Christmas, M. & B. A. Whitton, 1998a. Phosphorus and aquatic bryophytes in the Swale–Ouse river system, north-east England. 1. Relationship between ambient phosphorus, internal N:P ratio and surface phosphatase activity. Science of the Total Environment 210/211: 389–399.
Christmas, M. & B. A. Whitton, 1998b. Phosphorus and aquatic bryophytes in the Swale–Ouse river system, north-east England. 2. Phosphomonoesterase and phosphodiesterase activities of Fontinalis antipyretica. Science of the Total Environment 210/211: 401–409.
Chu, S. P., 1942. The influence of the mineral composition of the media on the growth of planktonic algae. 1. Methods and culture media. Journal of Ecology 30: 284–325.
Eckstein, R. L. & P. S. Karlsson, 1999. Recycling of nitrogen among segments of Hylocomium splendens as compared with Polytrichum commune: implications for clonal integration in an ectohydric bryophyte. Oikos 86: 87–96.
Ellwood, N. T. W., 2002. Factors Influencing Phosphatase Activities of Mosses in Upland Streams. Ph.D. Thesis. University of Newcastle-upon-Tyne, UK.
Ellwood, N. T. W., S. M. Haile & B. A. Whitton, 2002. Surface phosphatase activity of the moss Warnstorfia fluitans as an indicator of the nutrient status of an acidic stream. Verhandlung Internationale Vereinigung Limnologie 28: 620–623.
Fedde, K. N. & M. P. Whyte, 1990. Alkaline phosphatase (tissue-non specific isoenzyme) is a phosphoethanolamine and pyridoxal-5′phosphate ectophosphatase: normal and hypophosphatasia fibroblast study. American Journal of Human Genetics 47: 767–775.
Garcia-Alvaro, M. A., J. Martinez-Abaigar, E. Nuñez–Olivera & N. Beaucourt, 2000. Element concentrations and enrichment ratios in the aquatic moss Rhynchostegium riparioides along the River Iregua (La Rioja, Northern Spain). Bryologist 103: 518–533.
Gibson, M. T. & B. A. Whitton, 1987. Hairs, phosphatase activity and environmental chemistry in Stigeoclonium, Chaetophora and Draparnaldia (Chaetophorales). British phycological Journal 22: 11–22.
Gimeno, C., F. Puche & B. A. Whitton, 1998. Effect of pH on shoots and protonema of Warnstorfia fluitans (Hedw.) Loeske. Boletin Sociedad Español Briologia 13: 13–17.
Glime, J. M. & D. H. Vitt, 1984. The physiological adaptations of aquatic musci. Lindbergia 10: 41–52.
Grainger, S. L. J., A. Peat, D. N. Tiwari & B. A. Whitton, 1989. Phosphomonoesterase activity of the cyanobacterium (blue-green alga) Calothrix parietina. Microbios 59: 7–17.
Gross, W., 2001. Ecophysiology of algae living in highly acidic environments. Hydrobiologia 433: 173–180.
Hargreaves, J. W., E. J. H. Lloyd & B. A. Whitton, 1975. Chemistry and vegetation of highly acidic streams. Freshwater Biology 5: 563–576.
Hargreaves, J. W. & B. A. Whitton, 1976. Effect of pH on growth of acid stream algae. British Phycological Journal 11: 215–223.
Houba V., W. van Vark, I. Walinga & J.J. van der Lee, 1989. Plant Analysis Procedures (Part 7, Chapter 2.2). Department of Soil Science and Plant Analysis. Wageningen, The Netherlands.
Lambert, D. & W. Maher, 1994. An evaluation of the efficiency of the alkaline persulphate digestion method for the determination of total phosphorus in turbid waters. Water Research 29: 7–9.
Langer, C. L. & P. F. Hendrix, 1982. Evaluation of a persulphate digestion method for particulate nitrogen and phosphorus. Water Research 16: 1451–1454.
Livingstone, D. & B. A. Whitton, 1984. Water chemistry and phosphatase activity of the blue-green alga Rivularia in Upper Teesdale streams. Journal of Ecology 72: 405–421.
Novozamsky, I., V. J. G. Houba, R. van Eck & W. van Vark, 1983. A novel digestion technique for multi-element analysis. Communications in Soil Science and Plant Analysis 14: 239–249.
Press, M. C. & J. A. Lee, 1983. Acid phosphatase activity in Sphagnum species in relation to phosphate nutrition. New Phytologist 93: 567–573.
Proctor MCF. 2000. Physiological ecology. In Shaw A. J. & B. Goffinet (eds), Bryophyte Biology. Cambridge University Press, Cambridge, 225–247.
Skalar, 1995. The Sanplus Segmented Flow analyser: Soil and Plant Analysis. Instruction Manual: Publ. No. 0102003. Skalar Analytical, Breda, The Netherlands.
Steinman, A. D., 1994. The influence of phosphorus enrichment on lotic bryophytes. Freshwater Biology 31: 53–63.
Steinman, A. D. & H. L. Boston, 1993. The ecological role of aquatic bryophytes in a woodland stream. Journal of the North American Benthological Society 12: 17–26.
Turner, B. L., R. Baxter, N. T. W. Ellwood & B. A. Whitton, 2001. Characterization of the phosphatase activities of mosses in relation to their environment. Plant Cell and Environment 24: 1165–1176.
Turner, B. L., R. Baxter, N. T. W. Ellwood & B. A. Whitton, 2003. Seasonal phosphatase activities of mosses from Upper Teesdale, northern England. Journal of Bryology 25: 203–214.
Walinga, I., W. van Vark, V. J. G. Houba & L. L. van der Lee, 1989. Plant Analysis Procedures, Part 7. Department of Soil Science and Plant Nutrition, Wageningen Agricultural University, The Netherlands, 138–141 pp.
Wells, J. M. & D. H. Brown, 1996. Mineral nutrient recycling within shoots of the moss Rhytidiadelphus squarrosus in relation to growth. Journal of Bryology 19: 1–17.
Whitton, B. A., E. Clegg, M., Christmas, J. J. Gemmell & P.J. Robinson, 2002 Development of Phosphastase Assay for Monitroing Nutrients in Rivers – Methodology Manual for Measurement of Phosphatase Activity in Mosses and Green Algae. Environment Agency of England and Wales STRE106-E-P 53 pp. (Distributed by WRc, Frankland Road, Swindon, Wilts SN5 8YF, UK).
Whitton, B. A., A. M. Al-Shehri, N. T. W. Ellwood & B. L. Turner, 2005. Ecological aspects of phosphatase activity in cyanobacteria, eukaryotic algae and bryophytes. In Turner B. L., E. Frossard & D. S. Baldwin, (eds), Organic Phosphorus in the Environment. CAB International, Wallingford, UK: 205–241.
Acknowledgements
N.T.W.E. was supported by a studentship from the UK Engineering and Physical Sciences Research Council. The authors are also most grateful to H.J. Banks Ltd for other financial support and Mr C. Harle for access to the site.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: S. M. Thomaz
Rights and permissions
About this article
Cite this article
Ellwood, N.T.W., Whitton, B.A. Phosphatase activities of the aquatic moss Warnstorfia fluitans (Hedw.) Loeske from an acidic stream in North-East England. Hydrobiologia 575, 95–107 (2007). https://doi.org/10.1007/s10750-006-0283-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-006-0283-0