Abstract
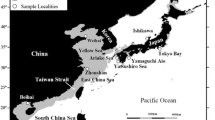
The genetic structure of spatially separated populations of the Dory snapper, Lutjanus fulviflamma, was investigated in seven areas along the East African coast and one area in the Comoros archipelago in the western Indian Ocean, using amplified fragment length polymorphism (AFLP). Phylogenetic and multidimensional scaling analyses did not show any clear clustering of individuals into the spatially separated populations. The analysis of molecular variance clearly showed that the variation was partitioned within populations and not between populations, leading to low genetic differentiation among populations. No clear relationship between genetic distance and geographic distance between populations was observed. These observations suggest that populations of Lutjanus fulviflamma have an open structure and are possibly genetically connected on a large geographic scale in the western Indian Ocean.
Similar content being viewed by others
References
Bay L. K., Choat J. H. and Robertson D. R. (2004). High genetic diversities and complex genetic structure in an Indo-Pacific tropical reef fish (Chlorurus sordidus): evidence of an unstable evolutionary past?. Marine Biology 144: 757–767
Beckman Coulter, 2002. CEQ™ 8000 Genetic Analysis System User’s Guide. Beckman Coulter Inc., Fullerton, California, USA
Bernardi G., Holbrook S. J. and Schmitt R. J. (2001). Gene flow at three spatial scales in a coral reef fish, the three-spot dascyllus, Dascyllus trimaculatus. Marine Biology 138: 457–465
Berry O., Tocher M. D. and Sarre S. D. (2004). Can assignment tests measure dispersal?. Molecular Ecology 13: 551–561
Brzustowski, J. 2002. Doh assignment test calculator. Available at http://www2.biology.ualberta.ca/jbrzusto/Doh.php
De Bruin A., Ibelings B. W. and van Donk (2003). Molecular techniques in phytoplankton research: from allozyme electrophoresis to genomics. Hydrobiologia 491: 47–63
De Roos K. (2003). CEQ™ 8000 AFLP Protocol. Beckman Coulter Netherlands BV, Mijdrecht, The Netherlands
Doherty P. J., Planes S. and Mather P. (1995). Gene flow and larval duration in seven species of fish from the Great Barrier reef. Ecology 76: 2373–2391
Excoffier L., Smouse P. E. and Quattro J. M. (1992). Analysis of molecular variance inferred from metric distances among DNA haplotypes: application to human mitochondrial DNA restriction data. Genetics 131: 479–491
Fauvelot C. and Planes S. (2002). Understanding origins of present-day genetic structures in marine fish: biologically or historically driven patterns?. Marine Biology 141: 773–788
Garcia A. A. F., Benchimol L. L., Barbosa A. M. M., Geraldi I. O. and Souza C. L. (2004). Comparison of RAPD, RFLP, AFLP and SSR markers for diversity studies in tropical maize inbred lines. Genetics and Molecular Biology 27: 579–588
Geertjes G. J., Postema J., Kamping A., van Delden J. J., Videler J. J. and van de Zande L. (2004). Allozymes and RAPDs detect little genetic population substructuring in the Caribbean stoplight parrotfish Sparisoma viride. Marine Ecology Progress Series 279: 225–235
Gold J. R. and Richardson L. R. (1994). Mitochondrial DNA variation among ‘red’ fishes from the Gulf of Mexico. Fishery Research 20: 137–150
Goudet J. (1995). FSTAT version 1.2: a computer program to calculate F-statistics. Journal of Heredity 86: 485–486
Huelsenbeck J. P. and Ronquist F. (2001). MRBAYES: Bayesian inference of phylogenetic trees. Bioinformatics 17: 754–755
Jones G. P., Milicich M.J., Emslie M. J. and Lunow C. (1999). Self-recruitment in a coral reef fish population. Nature 402: 802–804
Kingsford M. J., Leis J. M., Shanks A., Lindeman K. C., Morgan S. G. and Pineda J. (2002). Sensory environments, larval abilities and local self-recruitment. Bulletin of Marine Science 70: 309–340
Leis J. M. (2002). Pacific coral-reef fishes: the implications of behaviour and ecology of larvae for biodiversity and conservation and a reassessment of the open population paradigm. Environmental Biology of Fishes 65: 199–208
Leis J. M. and Carson-Ewart B. M. (2000). The Larvae of Indo-Pacific Coastal Fishes. An Identification Guide to Marine Fish Larvae. Fauna Malesiana Handbook 2, Brill, Leiden
Leis J. M. and Rennis D. S. (2000). Lutjanidae. In: Leis, J. M. and Carson-Ewart, B.M. (eds) The Larvae of Indo-Pacific Coastal Fishes. An Identification Guide to Marine Fish Larvae, pp 329–337. Fauna Malesiana Handbook 2, Brill, Leiden
Manel S., Gaggiotti O. E. and Waples R. S. (2005). Assignment methods: matching biological questions with appropriate techniques. Trends in Ecology & Evolution 20: 136–142
McCartney M. A., Acevedo J., Heredia C., Rico C., Quenoville B., Bermingham E. and McMillan W. O. (2003). Genetic mosaic in a marine species flock. Molecular Ecology 12: 2963–2973
McMillen-Jackson A. L., Bert T. M., Cruz-Lopez H., Seyoum S., Orsoy T. and Crabtree R. E. (2005). Molecular genetic variation in tarpon (Megalops atlanticus Valenciennes) in the northern Atlantic Ocean. Marine Biology 146: 253–261
Mickett K., Morton C., Feng J., Li P., Simmons M., Cao D., Dunham R. A. and Liu Z. (2003). Assessing genetic diversity of domestic populations of channel catfish (Ictalurus punctatus) in Alabama using AFLP markers. Aquaculture 228: 91–105
Miller, M. P., 1998. AMOVA-PREP: a program for the preparation of analysis of molecular variance input files from dominant-markers raw data, version 1.01. Available at http://www.marksgeneticsoftware.net/amovaprep.htm
Miller, M. P., 1999. Tools for population genetic analyses (TFPGA): a windows ™ program for the analysis of allozyme and molecular population genetic data. Available at http://www.marksgeneticsoftware.net/_vti_bin/shtml.exe/tfpga.htm
Mora C. and Sale P. F. (2002). Are populations of coral reef fish open or closed?. Trends in Ecology & Evolution 17: 422–428
Nei M. and Li W. H. (1979). Mathematical model for studying genetic variation in terms of restriction endonucleases. Proceedings of the National Academy of Science USA 76: 5269–5273
Nzioka R. M. (1979). Observations on the spawning seasons of East African reef fishes. Journal of Fish Biology 14: 329–342
Paetkau D., Calvert W., Sterling I. and Strobeck C. (1995). Microsatellite analysis of population structure in Canadian polar bears. Molecular Ecology 4: 347–354
Planes S., Galzin R. and Bonhomme F. (1996). A genetic metapopulation model for reef fishes in oceanic islands: the case of the surgeonfish, Acanthurus triostegus. Journal of Evolutionay Biology 9: 103–117
Planes S. (1993). Genetic differentiation in relation to restricted larval dispersal of the convict surgeonfish Acanthurus triostegus in French Polynesia. Marine Ecology Progress Series 98: 237–246
Planes S., Romans P. and Lecomte-Finiger R. (1998). Genetic evidence of closed life cycles for some coral reef fishes within Taiaro Lagoon (Tuamotu Archipelago, French Polynesia). Coral Reefs 17: 9–14
Planes S. and Fauvelot C. (2002). Isolation by distance and vicariance drive genetic structure of a coral reef fish in the Pacific Ocean. Evolution 56: 378–399
Ramon M. L., Lobel P. S. and Sorenson M. D. (2003). Lack of mitochondrial genetic structure in hamlets (Hypoplectrus spp.): recent speciation or ongoing hybridization?. Molecular Ecology 12: 2975–2980
Raymond M. and Rousset F. (1995). GENEPOP version 1.2: population genetics software for exact tests and ecumenicism. Journal of Heredity 86: 248–249
Richmond M. D. (2002). A Field Guide to the Seashores of Eastern Africa and the Western Indian Ocean Islands. Sida/SAREC-UDSM, Dar es Salaam, Tanzania
Rivera M. A. J., Kelley C. D. and Roderick G. K. (2004). Subtle population genetic structure in the Hawaiian grouper, Epinephelus quernus (Serranidae) as revealed by mitochondrial DNA analysis. Biological Journal of the Linnean Society 81: 449–468
Rhodes K. L., Lewis R. I., Chapman R. W. and Sadovy Y. (2003). Genetic structure of the camouflage grouper, Ephinephelus polyphekadion (Pisces: Serranidae), in the western central Pacific. Marine Biology 142: 771–776
Roberts C. M. (1997). Connectivity and management of Caribbean coral reefs. Science 278: 1454–1457
Saitou N. and Nei M. (1987). The neighbour-joining method: a new method for reconstructing phylogenetic trees. Molecular Biology and Evolution 4: 406–425
Schmitt R. J. and Holbrook S. J. (2002). Correlates of spatial variation in settlement of two tropical damselfishes. Marine and Freshwater Research 53: 329–337
Shulman M. J. and Bermingham E. (1995). Early life histories, ocean currents and the population genetics of Caribbean reef fishes. Evolution 49: 897–910
Simpson S. D., Meekan M. G., McCauley R. D. and Jeffs A. (2004). Attraction of settlement-stage coral reef fishes to reef noise. Marine Ecology Progress Series 276: 263–268
Stepien C. A., Randall J. E. and Rosenblatt R. H. (1994). Genetic and morphological divergence of a circumtropical complex of goatfishes: Mulloidichthys vanicolensis, M. dentatus, and M. martinicus. Pacific Science 48: 44–56
Swearer S. E., Caselle J. E., Lea D. W. and Warner R. R. (1999). Larval retention and recruitment in an island population of a coral-reef fish. Nature 402: 799–802
Swofford, D. L., 2002. PAUP*: phylogenetic analysis using parsimony (and other methods). Version 4.0 Beta. Sinauer Associates, Sunderland, Massachusetts, USA
Taylor M. S. and Hellberg M. E. (2003). Genetic evidence for local retention of pelagic larvae in a Caribbean reef fish. Science 299: 107–109
van Herwerden L., Benzie J. and Davies C. (2003). Microsatellite variation and population genetic structure of the red throat emperor on the Great Barrier Reef. Journal of Fish Biology 62: 987–999
Vos P., Hogers R. and Bleeker M. (1995). AFLP: a new technique for DNA fingerprinting. Nucleic Acids Research 23: 4407–4414
Warner R. R. (1997). Evolutionary ecology: how to reconcile pelagic dispersal with local adaptation. Coral Reefs 16: S115–S120
Wright S. (1931). Evolution in Mendelian populations. Genetics 16: 97–159
Yeh, F. C. & T. J. B. Boyle, 1999. POPGENE Version 1.31. Microsoft Window-based Freeware for Population Genetic Analysis. Available at http://www.ualberta.ca/~fyeh/
Zatcoff M. S., Ball A. O. and Sedberry G. R. (2004). Population genetic analysis of red grouper, Epinephelus morio, and scamp, Mycteroperca phenax, from the southeastern US Atlantic and Gulf of Mexico. Marine Biology 144: 769–777
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dorenbosch, M., Pollux, B.J.A., Pustjens, A.Z. et al. Population structure of the Dory snapper, Lutjanus fulviflamma, in the western Indian Ocean revealed by means of AFLP fingerprinting. Hydrobiologia 568, 43–53 (2006). https://doi.org/10.1007/s10750-006-0020-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-006-0020-8